Unit 3 Practice Questions
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
Oxidation of which of the following bonds result in energy production by candles, cars, mice, and us?
C-H and C-C
The symbol delta-Go represents _____ while the symbol delta-G represents _____.
a standard reference point; actual conditions
Which of the following statements regarding Gibbs free energy is correct?
If a reaction has a large and negative standard free energy change (∆G°), product formation will be favored in actual conditions unless the ratio of products to reactants is high enough to drive the reaction in the reverse direction.
Consider the reaction A → B. If, at equilibrium, the concentration of B is greater than the concentration of A, is the change in standard free energy (deltaG'o) of the reaction:
Negative
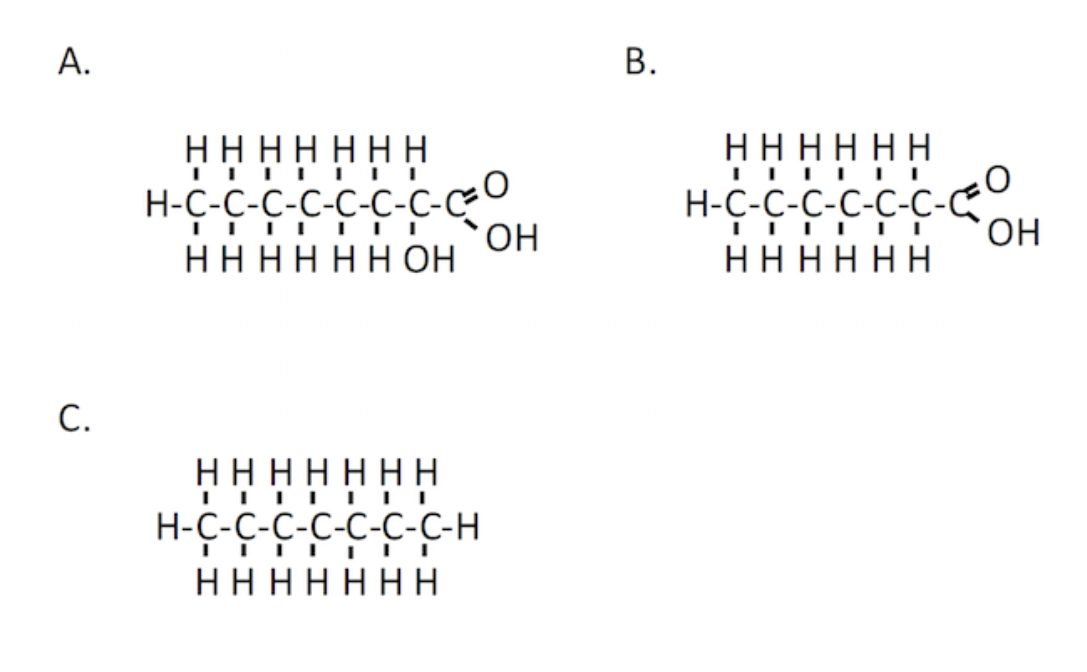
Oxidation of which of the following molecules will produce the largest amount of energy?
C
ATP is a substrate for phosphofructokinase, yet ATP inhibits enzyme activity. A possible model for this is:
ATP binds to both the active site of the enzyme and to a separate allosteric site.
Which of the following molecules allosterically inhibits phosphofructokinase (the third enzyme in glycolysis)? Think about what makes metabolic sense.
ATP
Steps 1, 3 and 10 of glycolysis are irreversible. Which of the following statements regarding glycolysis is incorrect?
The standard free energy changes for steps 1, 3 and 10 are large and positive.
How many pyruvate molecules are formed from one molecule of glucose?
2
How many net ATP are formed in glycolysis per glucose molecule?
2
Thiamine is necessary for pyruvate dehydrogenase activity. In a thiamine deficient person, which of the following compounds might you expect to be elevated?
pyruvate
Which of the following statements regarding fermentation is correct?
The lactate dehydrogenase reaction regenerates NAD+ for glycolysis.
Where is the pyruvate dehydrogenase complex localized?
mitochondria
Consider the pyruvate dehydrogenase complex. When is carbon dioxide formed in the pyruvate dehydrogenase reaction mechanism?
E1 reactions
Which of the following is a product of the pyruvate dehydrogenase complex?
NADH
The E3 subunit can be the same in PDH and alpha-keto DH because:
the substrates for E3 are identical in both reactions
How many reduced bonds does acetate have? In other words, how many reduced cofactors are formed when acetate is fully oxidized in the citric acid cycle?
4
How many molecules of CO2 are formed in the citric acid cycle per acetyl-CoA?
2
Where does the citric acid cycle take place?
mitochondria
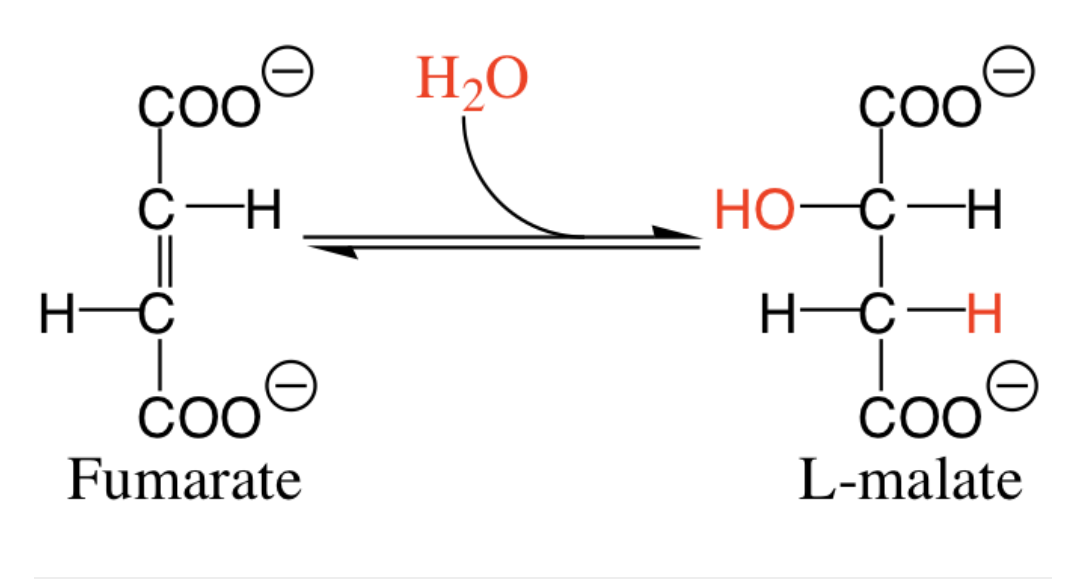
How many reduced cofactors (NADH or FADH2) are formed in this reaction?
0
Which of the following types of enzymes typically catalyzes redox reactions?
dehydrogenases
Where does beta-oxidation of fatty acids occur?
mitochondria
Palmitoyl-CoA is a saturated, 16-carbon long fatty acid. How many acetyl-CoA, NADH and FADH2 will be formed from the full oxidation of this molecule?
8, 7, 7
Why do fatty acids bond with carnitine prior to beta-oxidation?
Fatty acyl-carnitine can be transported across the inner mitochondrial membrane while fatty acyl-CoA cannot.
What was concluded from the Knoop experiment?
Fatty acids are oxidized 2-carbon units at a time.
Glutamine synthetase adds NH3 to ____ to generate glutamine.
Glutamate
Nitrogen enters the mitochondria from the liver cytosol in which of the following molecules?
Glutamate and glutamine
Two nitrogen-containing groups are used to make urea in liver cells. What two molecules of the urea cycle contribute these two nitrogen?
Carbamoyl phosphate and Aspartate
Which of the following statements regarding the urea cycle is incorrect?
All steps of the urea cycle occur in the cytosol.
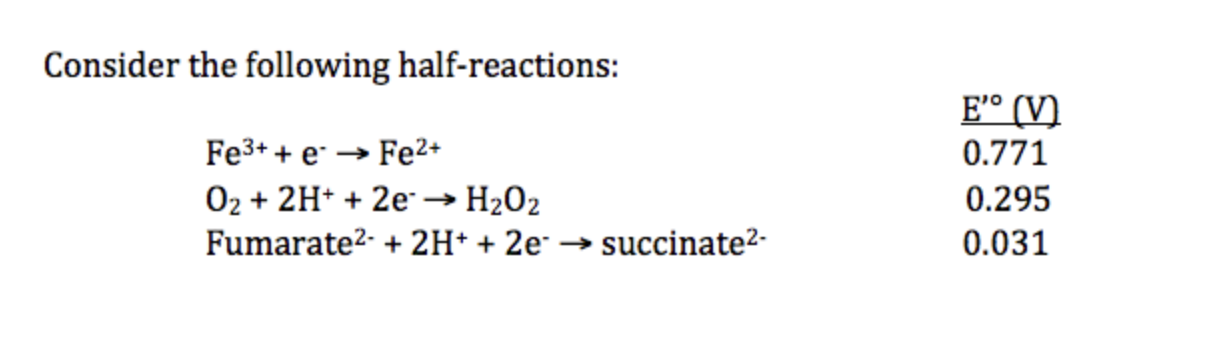
____ has the highest affinity for electrons while _____ has the lowest affinity for electrons.
Fe3+; fumarate
Reactions that have positive ∆E°’ values will proceed to the ______.
right
Which of the following complexes in the electron transport chain does not pump hydrogen ions into the inner membrane space of the mitochondria?
II
For every one NADH molecule oxidized at complex I, how many TOTAL hydrogen ions are pumped across all of the complexes from the mitochondrial matrix to the inner membrane space?
10
The proton motive force is required to release the ATP that is bound to ATP synthase.
True
Natural uncoupler proteins dissipate the proton gradient. Which of the following is a consequence of uncoupler proteins?
All of the above
Which of the following will allosterically increase the activity of phosphofructokinase-1?
AMP