ANS 8- Drug design - simple substitutions
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
How does structure affect function?
there is a link between the chemical group - and function - can result in different responses
What is a structure activity relationships ?
-It describes the interaction /relationship between the structure and its function
-what functional groups are actually needed or part of the functions are relevant
What do the interactions at the receptor depend on ?
Types of functional groups
number of functional groups
relative arrangement of functional groups
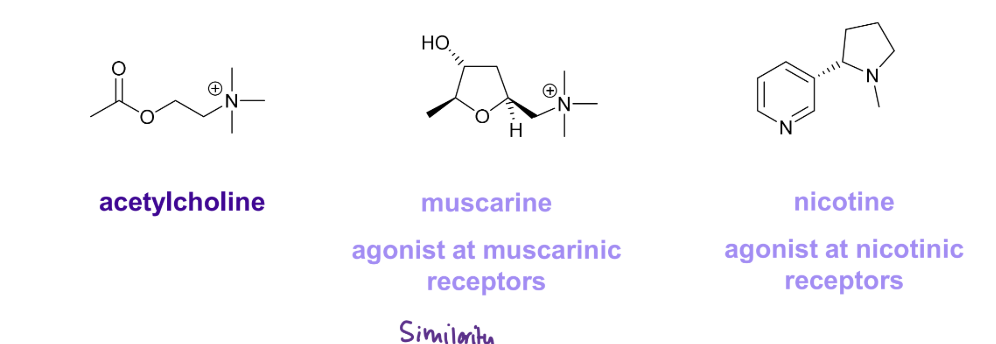
How do we design a new drug?
design a new drug from a natural ligand- new lead compound
5 steps :
identify the types of interactions possible- How could it bind
• identify the important structural features (simplification)- remove some interactions, see if it works
• develop a structure-activity relationship- written
• identify a pharmacophore- a diagram ( the similarities)
• modify its pharmaceutical properties - is it soluble, suitable for a patient, etc
What are the drug properties for the lead/natural ligand?
Usually, the lead is not selective between different receptor subtypes with different biological activities
• Usually not good ‘drug-like’ properties leading to difficult delivery
need to make it selective to receptor types
How do you determine the interactions ?
look at what can be protonated or deprotonated - results in charges +,- = ionic bond interactions
look at hba,hbd = hydrogen bond
aromatic - pi-pi stacking
van der Waals interactions- from any alkyl chains throughout molecules
What are the 3 changes to enhance binding activity and drug-like characteristics?
simple substitutions- removing, changing groups
isosteres,biosteres
Altering 3d shape - stereochemistry,rigidification
Chain extension - exploit additional interactions - making it more complex- better binding
NOTE- You can only change one at a time
Describe the simple substitutions-Ionic interactions
At physiological pH, it is generally only from
Quaternary ammoniums
Protonated amines
Deprotonated carboxylic acids
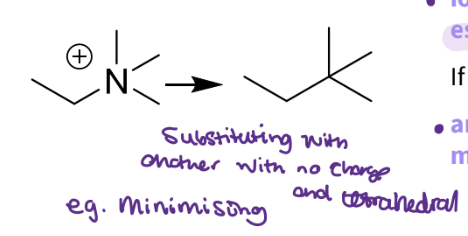
What substitution would you do for a Quaternary ammoniums?
Replace with a carbon
Same shape – tetrahedral
No charge
No activity - the ionic was essential
active- not needed
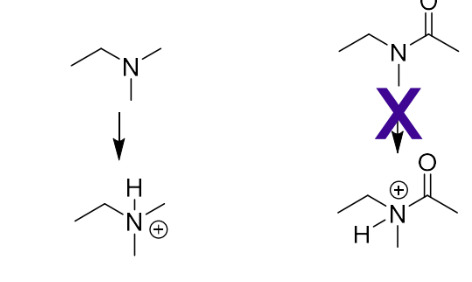
What substitution would you do for a tertiary amine?
Replace with an amide
Nitrogen in an amide is not able to become charged
no longer shows activity- amine is essential to activity
The molecule is still active -the amine is not interacting through an ionic interaction
What substitution would you do for a carboxylic acid?
Replace with
an aldehyde- cannot be deprotonated at physiological ph
an aliphatic alcohol- not deprotonated at physiological pH
an ester- worst option, methyl larger than h
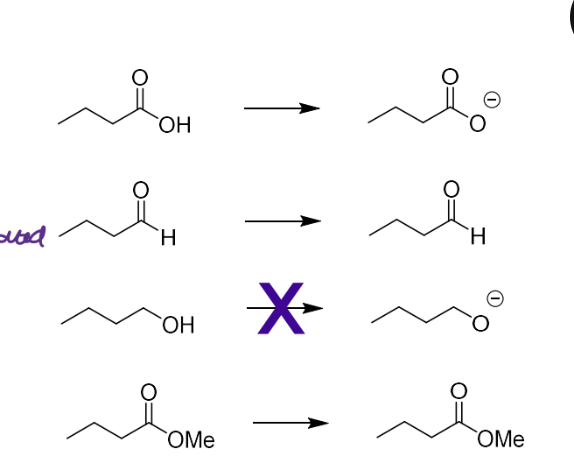
Describe the simple substitution - hydrogen bonding interactions
Groups that are BOTH Hydrogen Bond Donors and Acceptors
Groups that are acceptors
Groups that are donors
Note - usually make a series of changes due to Hba,hbd
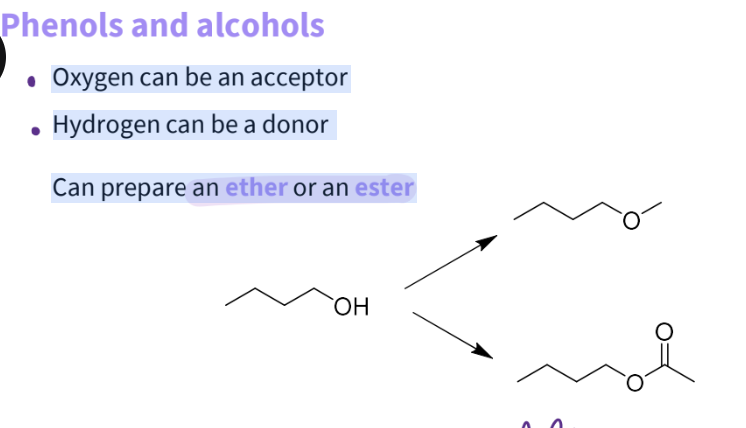
What substitutions would you do for a phenol and alcohol?
Oxygen can be an acceptor
Hydrogen can be a donor
Can prepare an ether- gets rid of hbd , reduces hba activity - better change
an ester- gets rid of hbd,more likely to reduce h bond activity
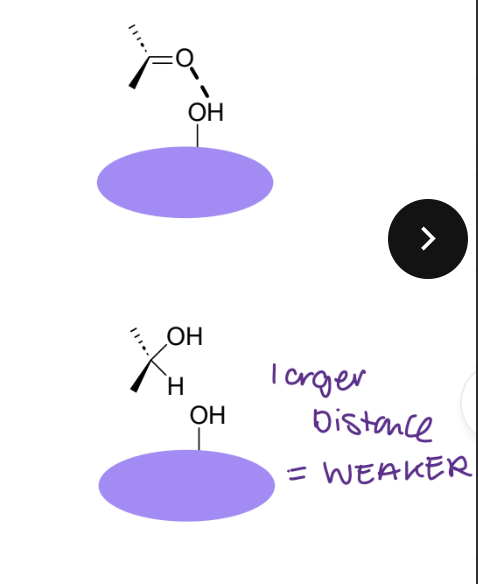
What changes would you make for a ketone and an aldehyde ?
Oxygen in carbonyl can be an acceptor
Two possible interactions
Can reduce to alcohol- larger distance = weaker
Still keeps H-bonding potential, but changes the shape of the molecule
Carbonyl is planar and sp2
Alcohol is tetrahedral and sp3
Likely to weaken any H-bonding interaction
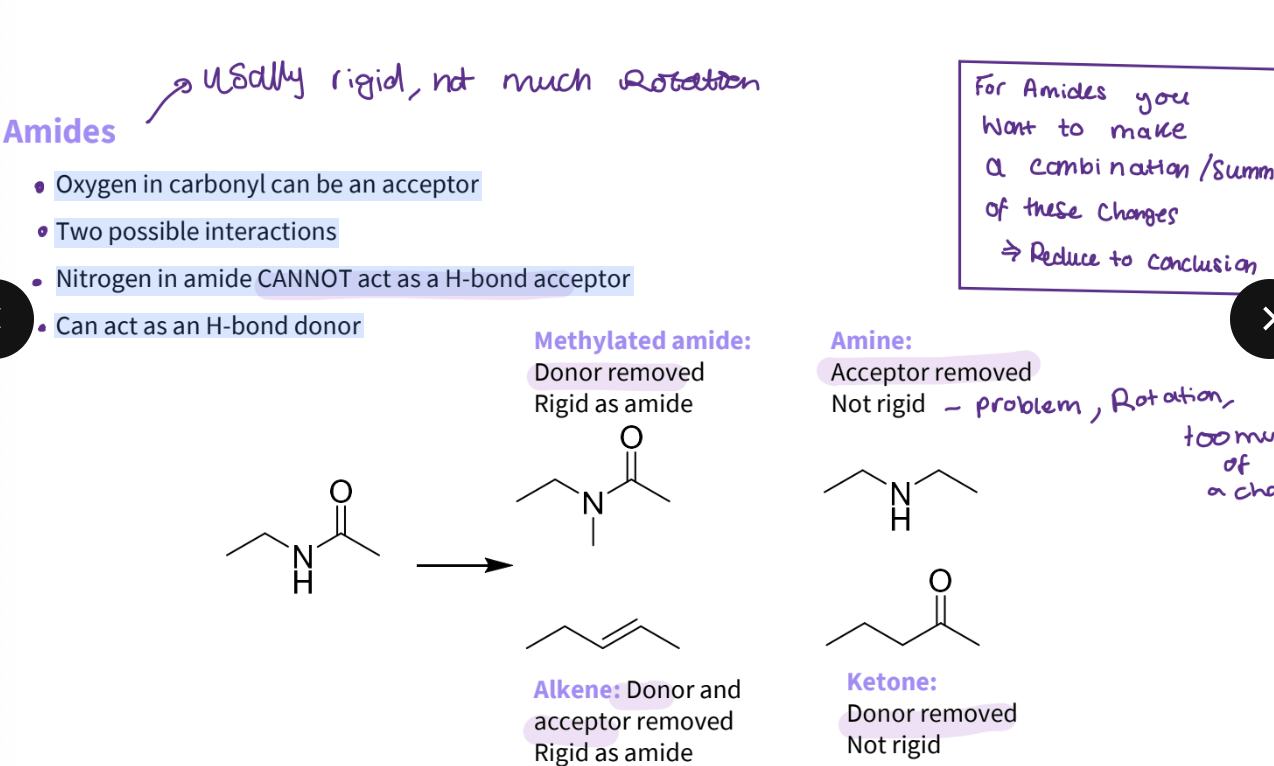
What substitutions would you make for an amide?
Oxygen in carbonyl can be an acceptor
Two possible interactions
Nitrogen in amide CANNOT act as a H-bond acceptor
Can act as an H-bond donor- remove donor methylated -rigid as an amine
remove acceptor - amine - not rigid - rotation too much of a change
alkene- donor and acceptor removes the rigidity
ketone - donor removed - not rigid
Note- for amides, you make a combination of changes
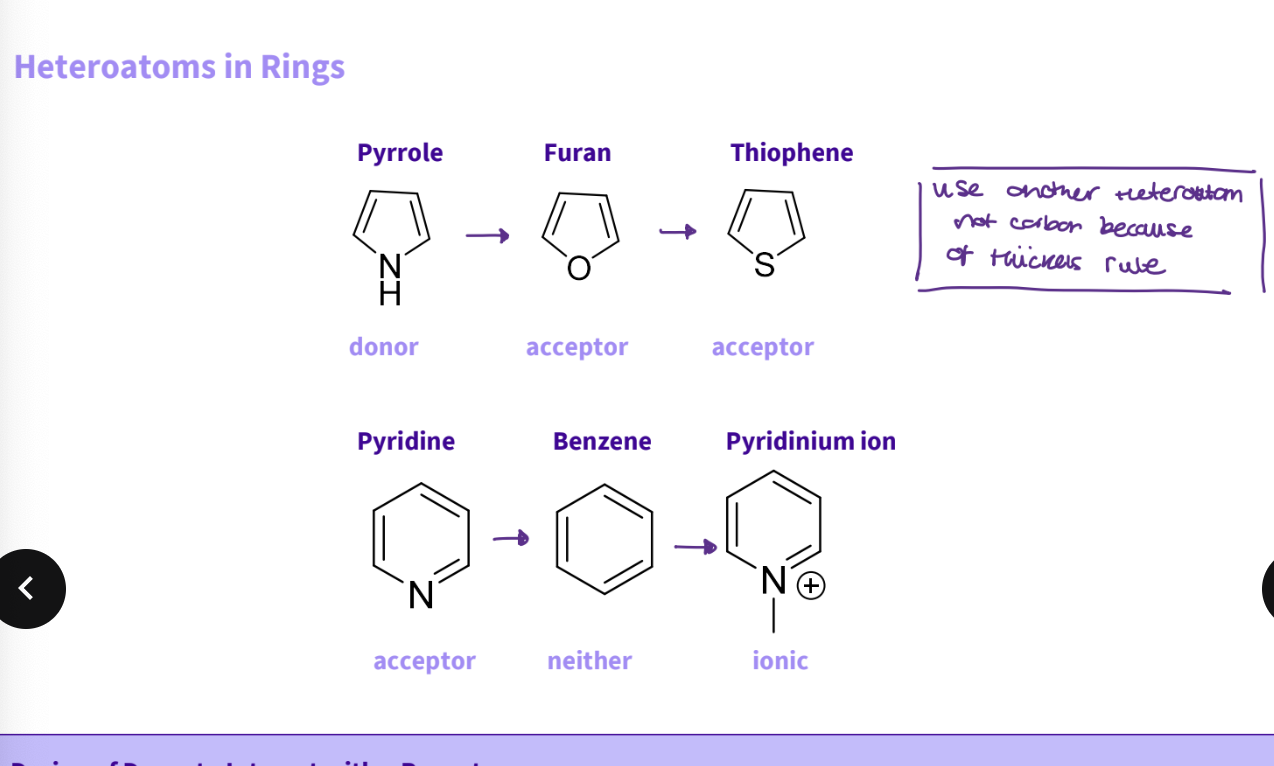
What substitutions do you do for a rings ?
use another atom - for pyrrole goes from donor - acceptor - heteroatoms, e.g., furan, thiophene
for pyridine acceptor - benzene neither - pyridinium ion ionic
What substitutions would you do for esters ?
Oxygen can act as an acceptor
Carbonyl more likely-more exposed,higher electron density
Could replace with an ether - removes carbonyl
What is the bigger issue when substituting for esters ?
Bigger issue:
lack of stability
easily enzymatically hydrolysed in the body
easily chemically hydrolysed on storage
may need to be replaced by a bioisostere
What is an isostere ?
Isostere: Atoms or groups that have the same valency and similar chemical and physical properties
eg - ether to a carbon chain
alcohol - to a carbon chain
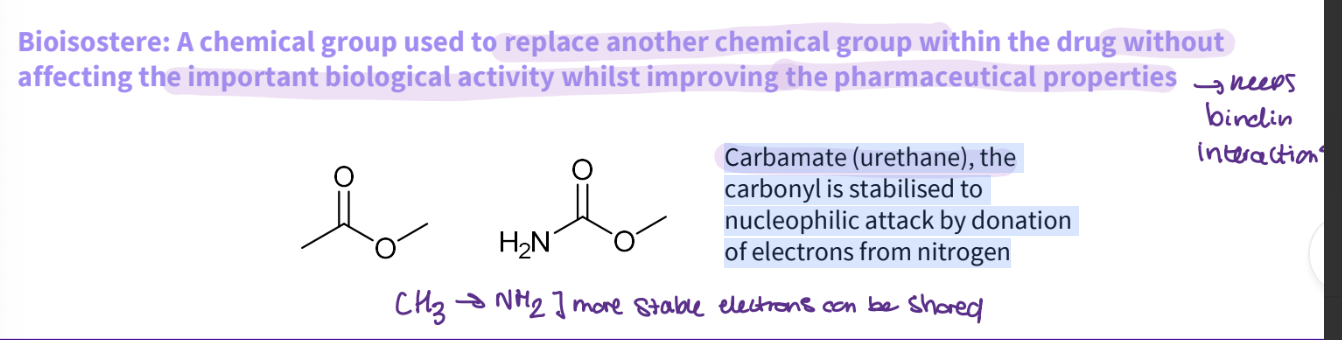
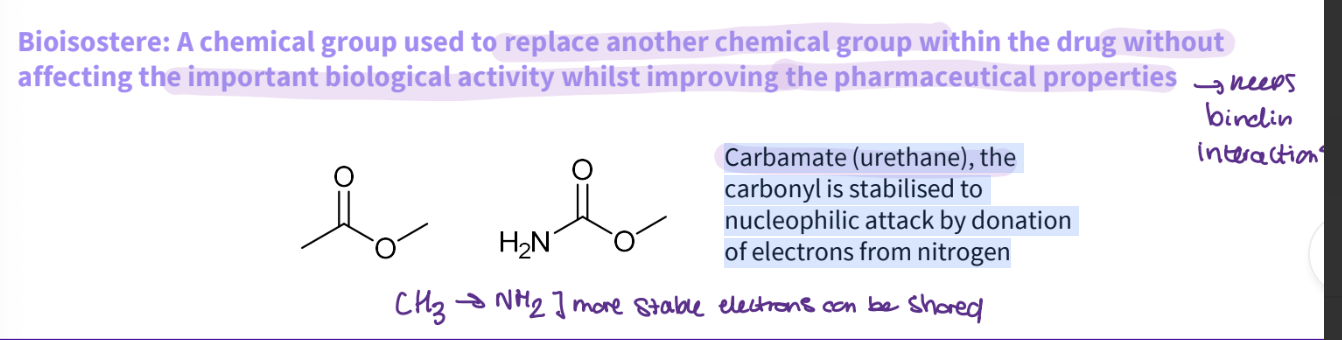
What is a bioisostere ?
Bioisostere: A chemical group used to replace another chemical group within the drug without affecting the important biological activity whilst improving the pharmaceutical properties- keeps the binding interactions
What would you do for an ester?
Carbamate (urethane), the carbonyl is stabilised to nucleophilic attack by donation of electrons from nitrogen
more stable - electrons can be shared
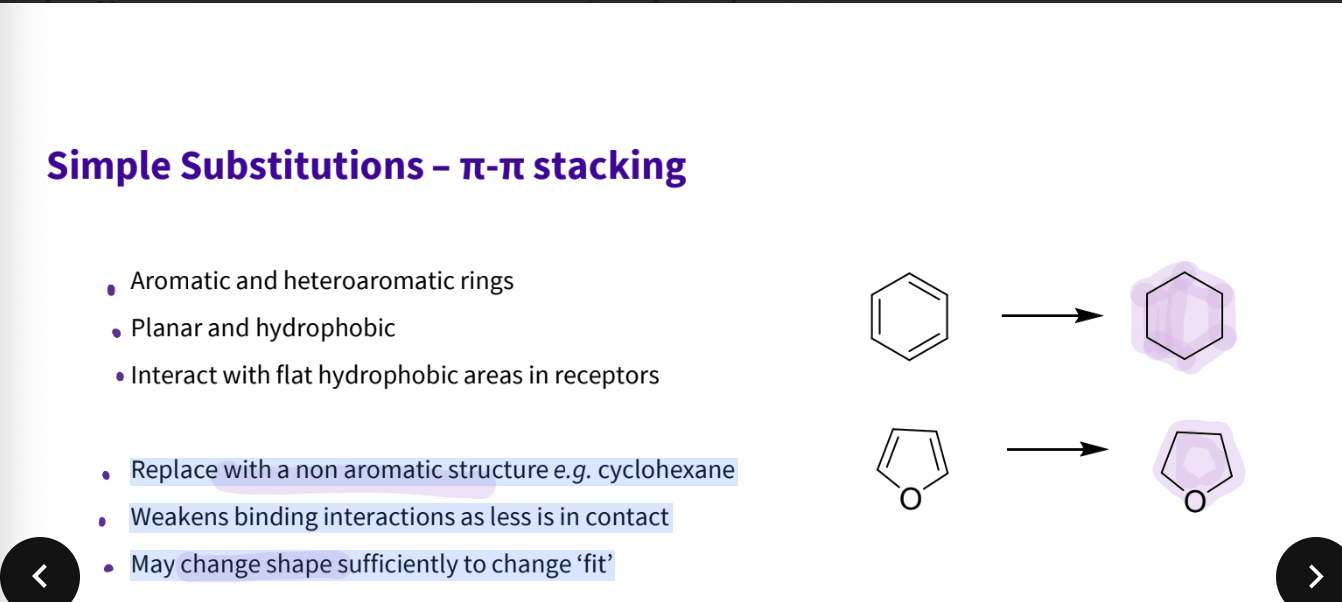
Describe simple substitutions - pi pi stacking
Aromatic and heteroaromatic rings
Planar and hydrophobic
Interact with flat hydrophobic areas in receptors
What substitution would you do for aromatics?
Replace with a non-aromatic structure, e.g. cyclohexane
Weakens binding interactions as less is in contact
May change shape sufficiently to change ‘fit’