2.1 - Introduction to Energy
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Thermodynamics
study of the flow of energy in systems
First Law of Thermodynamics
energy cannot be created or destroyed, but can be converted from one form to another
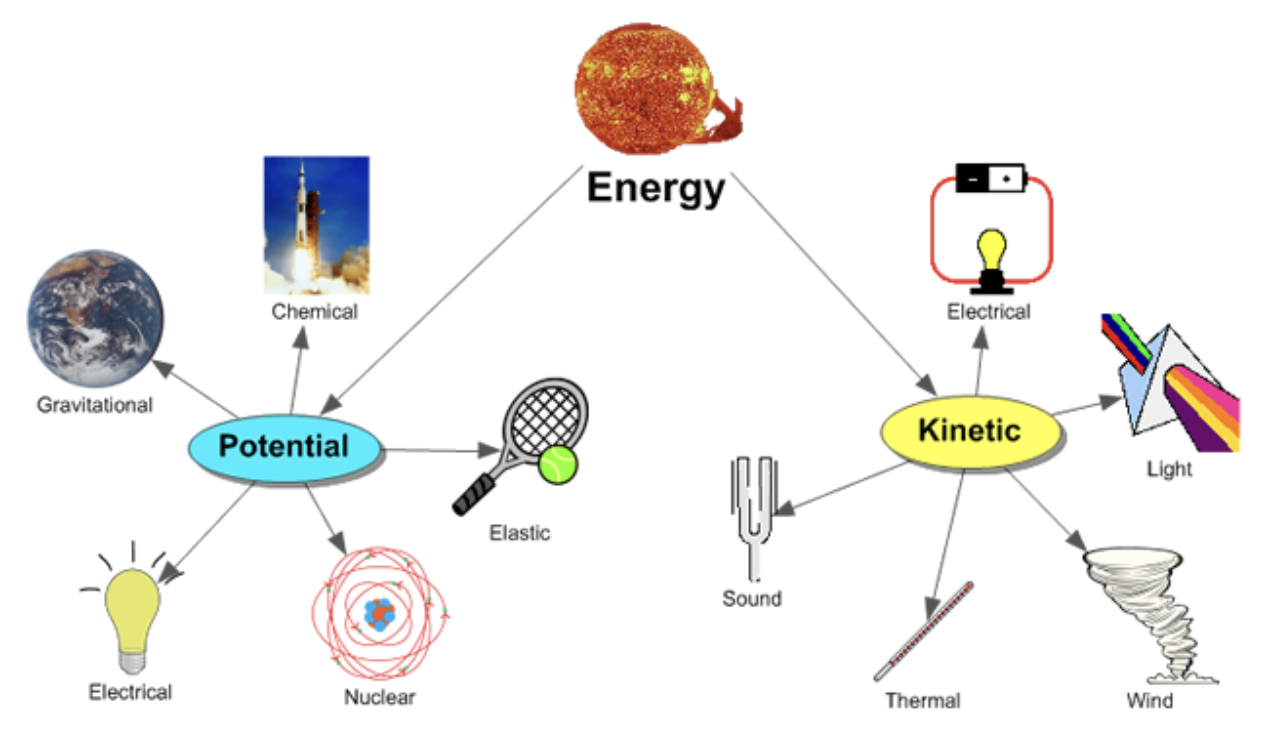
Energy
the ability to:
do “work”
bring about a change
2 states (of energy)
kinetic:
energy of motion
potential:
stored energy
Different Forms of Energy…
thermal - heat
electrical - flow of charged particles
nuclear - radioactive decay
light - waves of photons
sound - mechanical wave moves through air
chemical - potential energy stored in chemical bonds
Only type of energy organisms can use
Chemical Energy
Ex. glucose
contains chemical potential energy
(stored in its covalent bonds)
Types of energy
Organisms Need Energy For...
Organisms need energy every second for:
active transport
nerve transmission
homeostasis
muscle contraction
anabolic reactions
synthesis reactions → making bigger molecules
condensation/dehydration rxn’s
making bonds
endothermic (energy needed)
energy provided by hydrolysis of ATP