Chemistry - Organic
1/136
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
137 Terms
What is a disproportionation reaction
Where both oxidation and reduction take place .
Problems with Chlorine reacting with water in sunlight
The chlorine is regularly lost from water so has to be regularly topped up.
What are the properties of Catalytic Cracking?
High temperature and normal pressure but with a zeolite catalyst to produce aromatic compounds with carbon rings
Equation for Catalytic converters?
2CO + 2NO → 2CO2 + N2
Equation for flue gas desulphurization?
CaO + SO2 → CaSO3
What is the name of the reaction and what conditions are needed to form haloalkanes from alkanes?
Free radical substitution under UV light.
What are the problems with CFC’s?
The C-Cl bonds in CFCs are broken by ultraviolet radiation, releasing chlorine radicals. These act as catalysts in ozone depletion.
What are the safer alternatives to CFCs?
HFCs
What is the name of the mechanism and conditions needed to form an alkene from a haloalkane?
Elimination under alcoholic conditions with KOH
What mechanism do alkenes undergo?
Electrophilic Addition
Why are halogenoalkanes more reactive than normal alkanes?
The carbon halogen bond becomes a dipole. Reactivity of the halogenoalkanes increases as the bond gets weaker down the group.
What is the name of the mechanism of the production of alcohols from alkenes?
Electrophilic addition with H2SO4 catalyst.
Why are the formation of major products favoured than minor products?
Major products are made from a more stable carbocation. This stability arises from a greater positive inductive effect.
How to identify alkenes?
Use bromine water, if a double bond is present the solution will go from orange-brown to colourless.
How are branched chain polymers formed?
High pressures and temperatures produce branched chain polymers with weak intermolecular forces
How are straight chain polymers formed?
Low temperatures and pressures produce these with strong intermolecular forces.
What are some properties of polymers that make it good for the production of plastics?
They have multiple strong, non polar covalent bonds.
What is observed when an alcohol is oxidised?
Colour change from orange to green
What is the name of the mechanism and conditions needed to form an alkene from an alcohol?
Elimination with an excess of concentrated H2SO4
Why is flue gas desulphurisation needed?
To remove sulphur impurities that react to form a weak H2SO4.
Properties of thermal cracking
High Temperature and pressure to produce more alkenes than alkanes
What is formed when primary alcohols are heated with acidified potassium dichromate?
Aldehydes
How can primary alcohols oxidise to form caboxylic acids?
When their aldehyde is heated further under reflux
Why are some polymers unreactive?
They have multiple non-polar covalent bonds
What is formed when secondary alcohols are heated with acidified potassium dichromate?
A ketone
What does reacting an acyl chloride/acid anhydride with water make?
Carboxylic acid + HCl/Carboxylic acid
What does reacting an acyl chloride/acid anhydride with alcohol make?
Ester + HCl/Carboxylic acid
What does reacting an acyl chloride/acid anhydride with Ammonia make?
Amide + HCl/Carboxylic Acid
What does reacting an acyl chloride/acid anhydride with a primary amine make?
N-substituted amide + HCl/Carboxylic acid
What is stereoisomerism?
Two molecules with the same molecular and structural formula but the atoms are arranged differently in 3D space
Equation for the decomposition of ozone.
cl. + o3 → clo. + o2
clo. + o3 → 2o2 + cl.
Why are catalytic converters used?
They remove CO, NO and unburned hydrocarbons. They are lined with a platinum catalyst to increase surface area.
How can you distinguish acyl chlorides?
Add silver nitrate solution and white ppt of silver chloride should form
How does the shape of an organic molecule affect bp and mp?
Straight chain alkanes have a higher bp and mp because a greater surface area is in contanct with many more molecules so many more vdw forces can be made.
Why is heating under reflux used?
The reaction can run at the solvents boiling point for longer to ensure the reaction goes to completion, ensuring a higher yield without losing vapour materials.
Why is KCN used instead of HCN in cyanide mechanisms?
HCN is toxic and hard to store as a gas. KCN will completely ionise and HCN only partially ionises.
How are racemic mixtures produced?
The planar carbonyl group (C=O) means that the nucleophile can attack from either side with equal probability. The aldehyde/ketone must also be unsymmetrical.
Why are acid anyhdrides preferred to acyl chlorides?
Cheaper, less corrosive, no corrosive hcl produced, less exothermic
How are triglyceride esters formed?
From the esterification of glycerol
What is another name for glycerol?
propane-1,2,3-triol
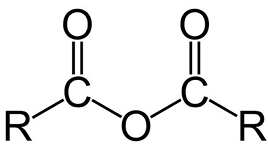
Draw the structure of an acid anhydride (use R for any other groups)
What is the name of the mechanism that acyl chlorides/acid anhydrides undergo?
Nucleophilic Addition - Elimination
What happens in acidic hydrolysis?
Acidic conditions and a H+ ion catalyst so it is a slow process. It happens in equilibrium so yeild of the alcohol and carboxylic acid are always low.
What catalyst is used to form esters from alcohols and carboxylic acids?
concentrated sulfuric acid
Test for aldehydes with fehlings solution
Changes colour from blue to a red ppt.
What happens in basic hydrolysis?
Ester will react with a base (NaOH) forming an alcohol and a carboxylate salt. The reaction goes to completion so has a higher percentage yield. You can regenerate the carboxylic acid by reacting the salt with a strong acid.
How are soaps formed?
When a triglyceride ester reacts with a base like NaOH, forming glycerol and 3 moles of a sodium soap.
How are triglyceride esters formed?
When glycerol reacts with 3 moles of a carboxylic acid.
How is biodeisel formed?
When a triglyceride ester reacts with methanol, forming glycerol and 3 moles of a methyl ester.
How does infrared spectroscopy work?
The bond that joins a pair of atoms is always vibrating. The stronger bonds vibrate faster at a higher frequency. This bond, will absorb a frequency of IR radiation equal to the natural frequency of the bond. The radiation emerged can be passed through an instrument where it plots a graph of intensity against frequency. The dips represent frequencies of specific bonds in the sample being absorbed.
How do you test for an alcohol?
Add acidified potassium dichromate, primary and secondary alcohols will change from orange dichromate ions to green chromium ions. Tertiary alcohols experience no change
Test for an aldehyde?
Tollen’s Reagent will form a silver mirror.
What is the mechanism and reagent needed for the reduction of aldehydes/ketones to their alcohols?
Nucleophilic addition, NaBH4
What is the name of the mechanism and reagents needed to form hydroxynitriles from aldehydes/ketones?
Nucleophilic Addition and KCN
Test for a halogenoalkane?
Sodium hydroxide solution warmed then add silver nitrate and dilute nitric acid. Add the haloalkane and a silver halide ppt should form.
How can a PVC be made flexible?
Adding a plasticiser
What is the structure of the benzene molecule?
A planar hexagonal molecule with 6 carbon atoms. It has a 120 degree bond angle with all C-C bonds equal in length.
What is the bonding in the benzene molecule?
Each Carbon makes three covalent bonds, 2 to carbon and 1 to hydrogen. Delocalised electrons in the p orbital overlap to form an electron ring above and below the plane of the molecule.
What was the original structure of benzene?
A hexagonal ring with alternating double and single bonds.
Why is the new model of benzene more thermodynamically stable than the old structure of benzene?
It has a delocalised electron orbital
What is the difference in stability between benzene and the hypothetical cyclohexatriene?
Benzene is less exothermic and more stable by 152 kJ mol^-1
What is the order of the bonds of increasing bond length in cyclohexatriene and benzene?
c=c, c-c bond in benzene, normal c-c bond
What is the name for aromatic compounds that contain a benzene ring?
Arenes
What reactions does benzene undergo?
Electrophilic substitution
In nitration of benzene, give the reagents needed to produce the electrophile NO2+
Concentrated H2SO4 and HNO3
Give the equation for the generation of the electrophile NO2+.
H2SO4 + HNO3 → HSO4- + H2O + NO2+
Which reagent in making the NO2+ electrophile is the base?
HNO3 as it accepts the H+ proton
What is formed in the acylation of benzene?
A phenyl ketone
What is the electrophile in the acylation of benzene and how is it formed?
RCO+, formed by reacting an acyl chloride/anhydride with aluminium chloride which is the catalyst.
What is the equation to form the RCO+ electrophile?
AlCl3 + RCOCl → AlCl4- + RCO+
Why is aluminium chloride a good choice for forming the RCO+ electrophile?
It polarises the electron cloud around the chloride ion, causing it to be removed so the catalyst gains it.
How is the catalyst regenerated from AlCl4-?
AlCl4- + H+ → AlCl3 + HCl
In what complexes does cis(Z) - trans (E) isomerism occur?
Octahedral and square planar
What would a cis (Z) isomer look like?
2 ligands which are next to each other
What would a trans (E) isomer look like?
2 ligands opposite to each other
In what complexes does optical isomerism occur?
Octahedral complexes
Give an example of a linear complex.
[Ag(NH3)2]+
Give an example of a square planar complex.
[PtCl4]2-
Give an example of a tetrahedral complex.
[CuCl4]2-
Give an example of an octahedral complex.
[Cu(H2O)6]2+
Why does NMR sepctroscopy work?
Different bond environments absorb different amounts of energy, therefore they show as different peaks on a spectrum.
Why is TMS used in NMR spectroscopy?
It is added along with the organic molecule to be used as a standard. It will show as one peak at 0ppm on the x axis. It is inert and non-toxic and can be removed from the sample easily as it has a low boiling point.
In what solvent should samples that are being analysed in Proton NMR be dissolved in, and why are these solvents used?
CCl4 or CDCl4 as they are non polar and they do not contain any hydrogen atoms so will not produce any peaks on the spectrum.
How can you classify amines in terms of them being 1o, 2o or 3o ?
The number of R groups bonded to the nitrogen is equal to the class.
For amines, what do you name the benzene group attached to the nitrogen?
phenyl
If other functional groups of higher priority are present, what is used instead of amine?
amino at the beginning of the name
How can ammonia and amines behave as nucleophiles?
They have an electron pair to donate
How can ammonia and amines behave as a base?
They have a lone pair to accept a H+ ion.
Why is phenylamine not a good base?
The lone pair on the nitrogen become part of the electron ring in the benzene making them less available for protonation
How can primary amines be formed in a 1 step process?
Nucleophilic substitution with excess ammonia
How are some amines a stronger base than others?
The R group increases electron density on the nitrogen lone pair. It becomes more available for protonation.
What is the 2 step process to form primary amines?
Nucleophilic substitution reaction (ethanol) with NaCN and then reduction with 2H2 under a nickel catalyst.
Why are the advantages and disadvantages of using the 1 step process to form primary amines?
Yield is higher, no toxic or flammable substances. Primary amine produced is impure and can react further with more of the haloalkane
In the further substitution reactions, how do you form the amine as the major product?
React the haloalkane with excess ammonia
In the further substitution reactions, how do you form the QAS as the major product?
React ammonia with excess haloalkane
How is phenylbenzene formed?
Reduction of nitrobenzene by HCl with a tin catalyst followed by NaOH
What is the equation for the formation of phenylamine from nitrobenzene?
C6H5NO2 + 6[H] → C6H5NH2 + 2H2O
What are phenylamines commonly used for?
Dyes
What can happen to the phenylamine after its production?
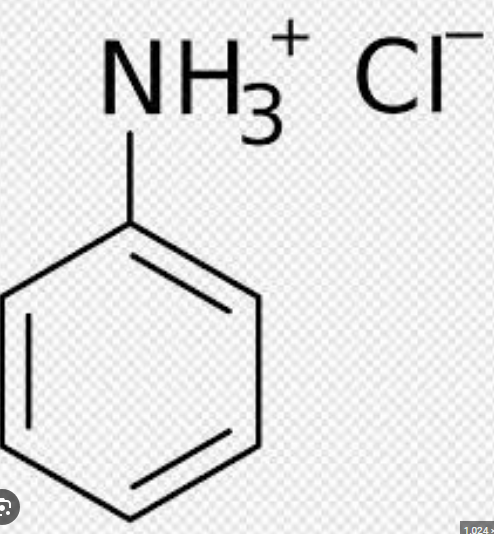
It can react with excess HCl and become protonated to form phenylammonium chloride which is soluble in water
Draw the structure of phenylammonium chloride.