AP Biology Chapter 5
1/47
Earn XP
Description and Tags
The Structure and Function of Large Biological Molecules
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
Macromolecules
A giant molecule formed by the joining of smaller molecules. Polysaccharides, proteins, and nucleic acids are macromolecules.
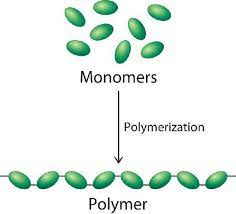
Polymer
A long molecule consisting of many similar or identical monomers linked together by covalent bonds.
Monomer
The subunit that serves as the building block of a polymer.
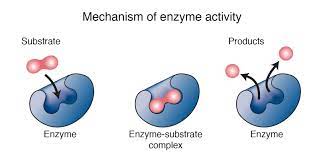
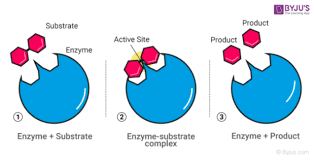
Enzymes
A macromolecule serving as a catalyst, a chemical agent that increases the rate of a reaction without being consumed by the reaction. Most enzymes are proteins.
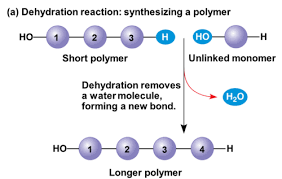
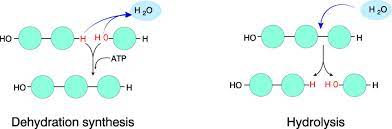
Dehydration Reaction
A chemical reaction in which two molecules become covalently bonded to each other with the removal of a water molecule.
Hydrolysis
A chemical reaction that breaks bonds between two molecules by the addition of water; functions in disassembly of polymers to monomers.
Carbohydrates
A sugar (monosaccharide) or one of its dimers (disaccharides) or polymers (polysaccharides).
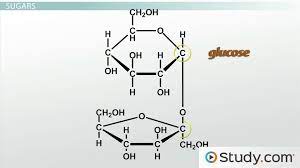
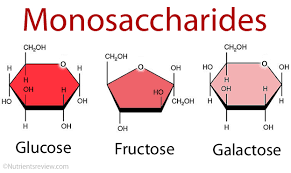
Monosaccharide
The simplest carbohydrate, active alone or serving as a monomer for disaccharides and polysaccharides. Also called simple sugars, monosaccharides have molecular formulas that are generally some multiple of C-H-subscript-2-O.
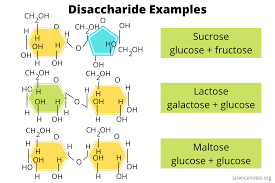
Disaccharide
A double sugar, consisting of two monosaccharides joined by a glycosidic linkage formed by a dehydration reaction.
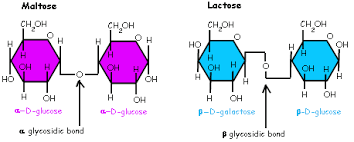
Glycosidic Linkage
A covalent bond formed between two monosaccharides by a dehydration reaction.
Polysaccharide
A polymer of many monosaccharides, formed by dehydration reactions.
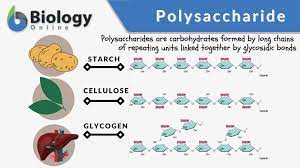
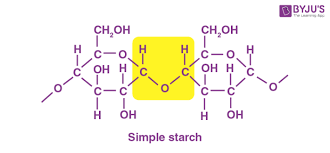
Starch
A storage polysaccharide in plants, consisting of entirely glucose monomers joined by glycosidic linkages.
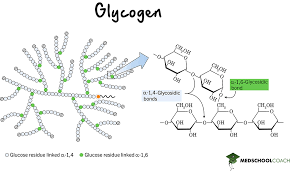
Glycogen
An extensively branched glucose storage polysaccharide found in the liver and muscle of animals; the animal equivalent of starch.
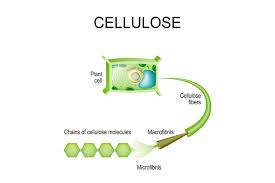
Cellulose
A structural polysaccharide of planting cell walls, consisting of glucose monomers joined by beta glycosidic linkages.
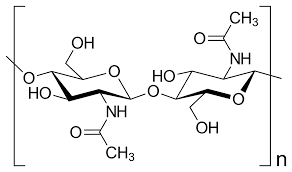
Chitin
A structural polysaccharide, consisting of amino sugar monomers, found in many fungal cell walls and in the exoskeletons of all arthropods.
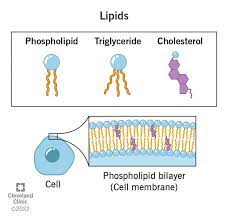
Lipids
Any large group of biological molecules, including fats, phospholipids, and steroids, that mix poorly, if at all, with water.
Fat
A lipid consisting of three fatty acids linked to one glycerol molecule; also called a triaglycerol or triglyceride.
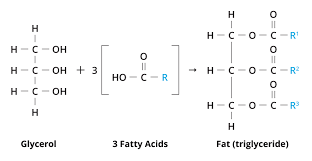
Fatty Acid
A carboxylic acid with a long carbon chain. Fatty acids vary in length and in the number and location of double bonds; three fatty acids linked to a glycerol molecule form a fat molecule, also called a triaglycerol or triacylglyceride.
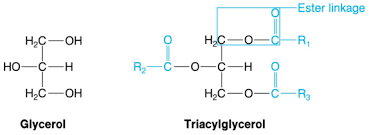
Triacylglycerol
A lipid consisting of three fatty acids linked to one glycerol molecule; also called a fat of triglyceride.
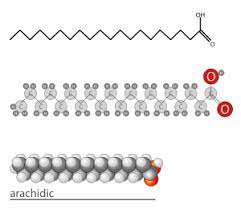
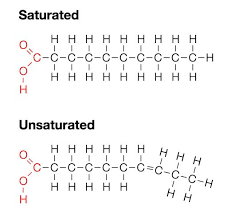
Saturated Fatty Acid
A fatty acid in which all carbons in the hydrocarbon tail are connected by single bonds, thus maximizing the number of hydrogen atoms that are attached to the carbon skeleton.
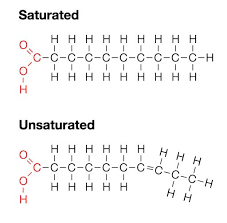
Unsaturated Fatty Acid
A fatty acid that has one or more double bonds between carbons in the hydrocarbon tail. Such bonding reduces the number of hydrogen atoms attached to the carbon skeleton.
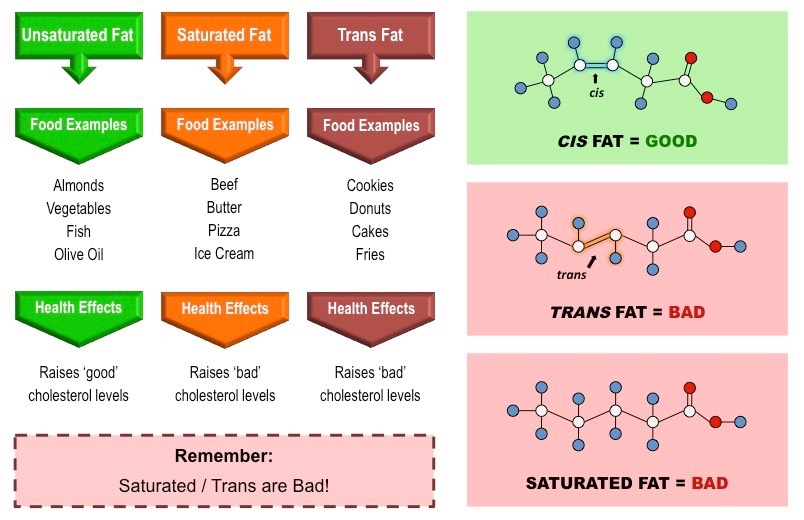
Trans Fat
An unsaturated fat, formed artificially during hydrogenation of oils, containing one or more trans double bonds.
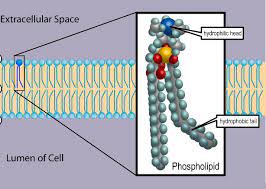
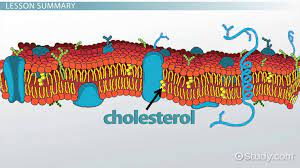
Phospholipid
A lipid made up of glycerol joined to two fatty acids and a phosphate group. The hydrocarbon chains of the fatty acids act as nonpolar, hydrophobic tails, while the rest of the molecule acts as polar, hydrophilic head. Phospholipids form bilayers that function as biological membranes.
Steroids
A type of lipid characterized by a carbon skeleton consisting of four fused rings with various rings attached.
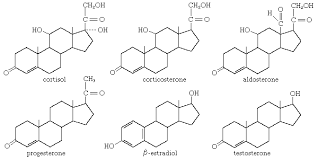
Cholesterol
A steroid that forms an essential component of animal cell membranes and acts as a precursor molecule for the synthesis of other biologically important steroids, such as many hormones.
Catalysts
A chemical element that selectively increases the rate of a reaction without being consumed by the reaction.
Polypeptide
A polymer of many amino acids linked together by peptide bonds.
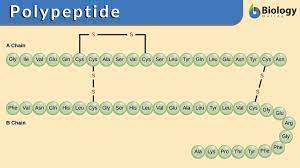
Protein
A biologically functional molecule consisting of one or more polypeptides folded and coiled into a specific three-dimensional structure.
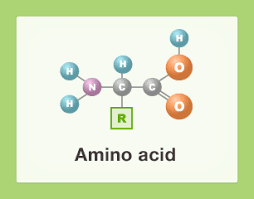
Amino Acid
An organic molecule processing both a carboxyl group and an amino group. Amino acids serve as the monomers of polypeptides.
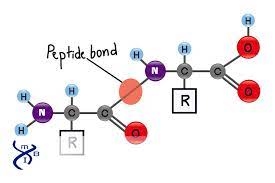
Peptide Bond
The covalent bond between the carboxyl group on one amino acid and the amino group on another, formed by a dehydration reaction.
Primary Structure
The level of protein structure referring to the specific linear sequence of amino acids.
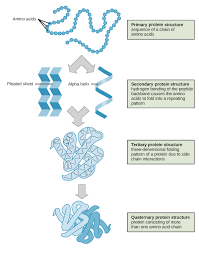
Secondary Structure
Regions of repetitive coiling or folding of the polypeptide backbone of a protein due to hydrogen bonding between constituents of the backbone (not the side chains).
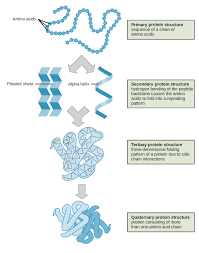
Tertiary Structure
The overall shape of a protein molecule due to interactions of amino acid side chains, including hydrophobic interactions, ionic bonds, hydrogen bonds, and disulfide bridges.
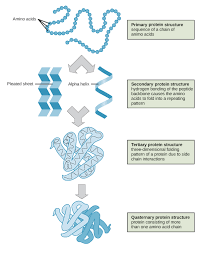
Sickle-Cell Disease
A recessively inherited human blood disease in which a single nucleotide change in the a-globin gene causes hemoglobin to aggregate, changing red blood cell shape and causing multiple symptoms in affected individuals.
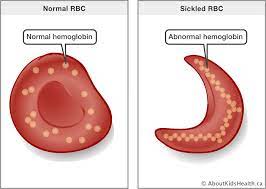
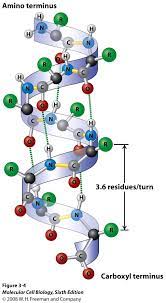
Alpha Helix
A coiled region constituting one form of the secondary structure of proteins, arising from a specific pattern of hydrogen bonding between atoms of the polypeptide backbone (not the side chains).
Beta Pleated Sheets
One form of the secondary structure of proteins in which the polypeptide chains fold back and forth. Two regions of the chain lie parallel to each other and are held together by hydrogen bonds between atoms of the polypeptide backbone (not the side chains).
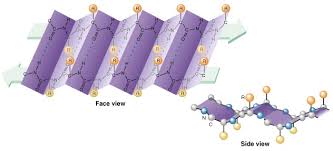
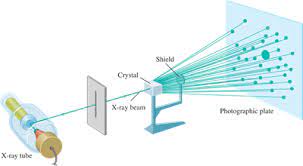
X-Ray Crystallography
A technique used to study the three-dimensional structure of molecules. It depends on the diffraction of an X-ray beam by the individual atoms of a crystallized molecule.
Nucleic Acids
A polymer (polynucleotide) consisting of many nucleotide monomers; serves as a blueprint for proteins and, through actions of proteins, for all cellular activities. The two types are DNA and RNA.
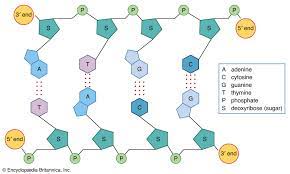
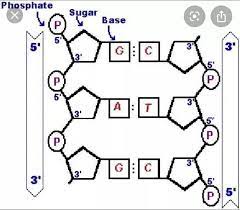
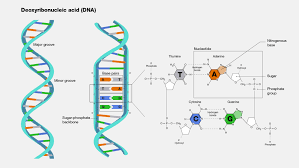
Deoxyribonucleic Acid
A nucleic acid molecule, usually a double-stranded helix, in which each polynucleotide strand consists of nucleotide monomers with a deoxyribose sugar and the nitrogenous bases Adenine (A), Cytosine (C), Guanine (G), and Thymine (T); capable of being replicated and determining the inherited structure of a cell’s proteins.
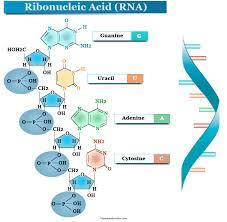
Ribonucleic Acid
A type of nucleic acid consisting of a polynucleotide made up of nucleotide monomers with a ribose sugar and the nitrogenous bases Adenine (A), Cytosine (C), Guanine (G), and Uracil (U); usually single-stranded; functions in protein synthesis, gene regulation, and as the genome of some viruses.
Polynucleotide
A polymer consisting of many nucleotide monomers in a chain. The nucleotides can be those of DNA and RNA.
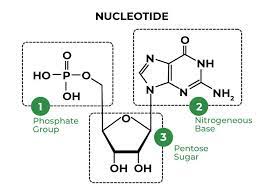
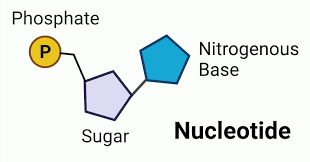
Nucleotide
The building block of a nucleic acid, consisting of a five-carbon sugar covalently bonded to a nitrogenous base and one to three phosphate groups.
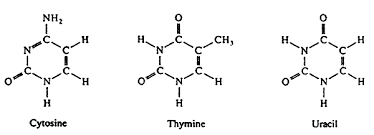
Pyrimidine
One of two types of nitrogenous bases found in nucleotides, characterized by a six-membered ring. Cytosine (C), Thymine (T), and Uracil (U) are pyrimidines.
Purine
One of two types of nitrogenous bases found in nucleotides, characterized by a six-membered ring fused to a five-membered ring. Adenine (A) and Guanine (G) are purines.
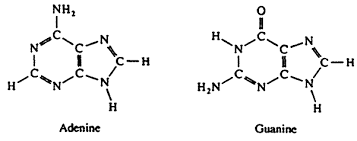
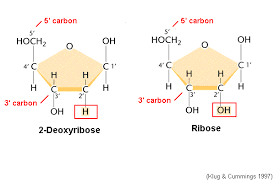
Deoxyribose
The sugar components of DNA nucleotides, having one fewer hydroxyl group than ribose, the sugar component of RNA nucleotides.
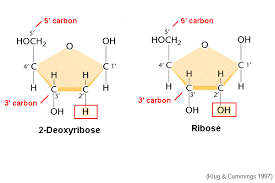
Ribose
The sugar components of RNA nucleotides.
Double Helix
The form of native DNA, referring to its two adjacent antiparallel polynucleotide strands wound around an imaginary axis into a spiral shape.
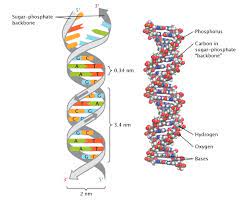
Antiparallel
Referring to the arrangement of sugar-phosphate backbones in a DNA double helix (they run in opposite 5’ to 3’ directions).