VSEPR "Electron Geometry"
0.0(0)
Studied by 0 peopleCard Sorting
1/6
Earn XP
Description and Tags
Electron Geometry
Last updated 3:52 PM on 3/24/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
1
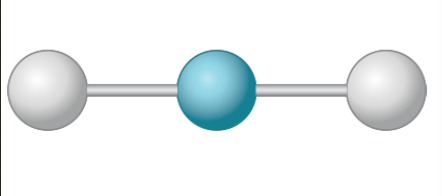
New cards
Liner
180°
2
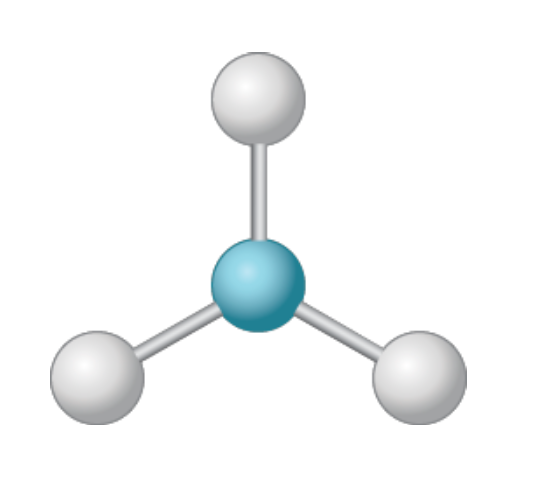
New cards
Trigonal Planar
120°
3
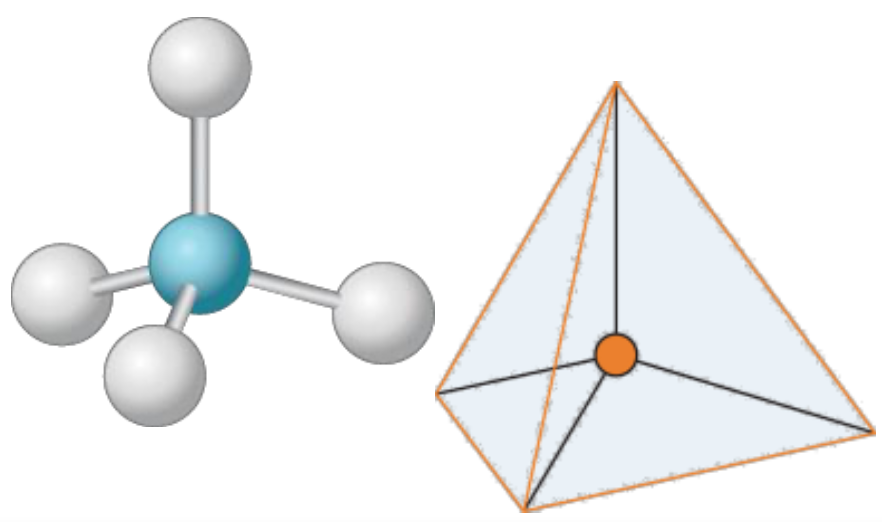
New cards
Tetrahedral
109.5°
4
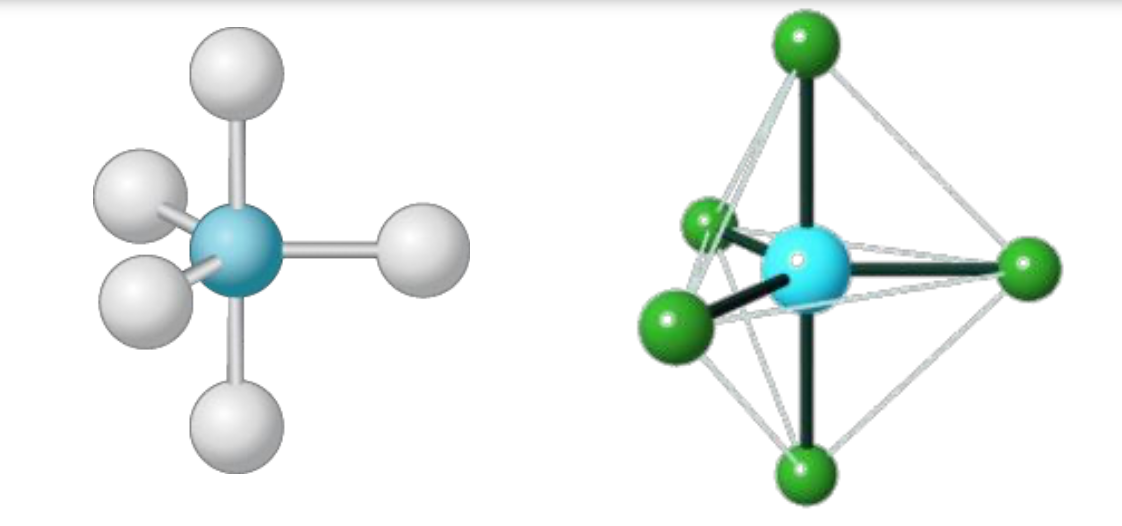
New cards
Trigonal Bipyramidal
90° and 120°
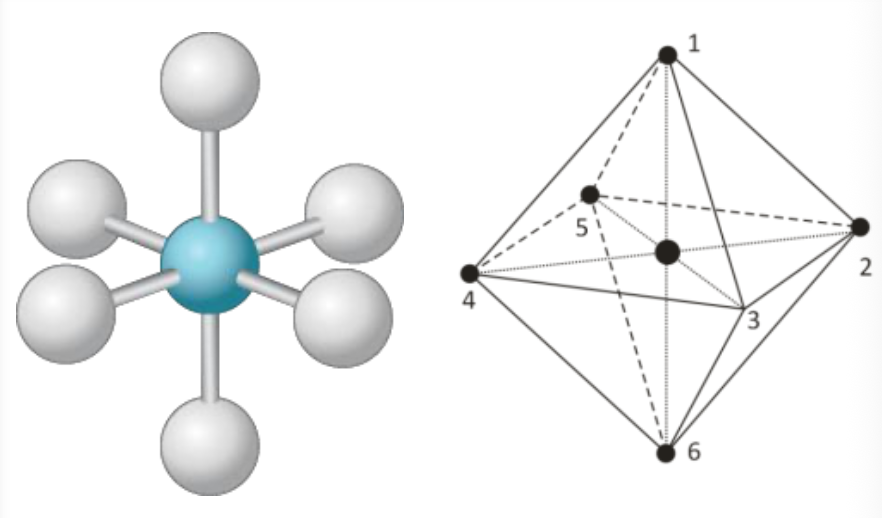
5
New cards
Octahedral
90°
6
New cards
how does Lone Pair affect the angles
They push the bonded atoms closer together squishing the bond angle “<“ making it smaller
7
New cards
Double/Triple bond
Contains way more negative charge, than a single bond.
Push the single bonds away from them, making them “>“ Larger
Explore top notes
Ecology Notes (Intro, Population Ecology, Community Ecology, Ecosystem Ecology)
Updated 1032d ago0.0(0)
Ecology Notes (Intro, Population Ecology, Community Ecology, Ecosystem Ecology)
Updated 1032d ago0.0(0)