MIMM 214 - Intro Immunology Midterm
1/117
Earn XP
Description and Tags
Not all encompassing, but hopefully useful
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
118 Terms
4 Categories of Pathogens
Virus, Bacteria, Parasite, Fungi
Primary Lymphoid Organs
Where immune cells are generated.
Bone Marrow and Thymus
Secondary Lymphoid Organs
Where immune cells are activated.
Lymph Nodes, Spleen, and Muscosal Associated Lymphoid Tissue
Leukocytes
White blood cells that arise from Hematopoietic Stem Cells in the bone marrow.
Hematopoiesis
The process by which HSCs differentiate. Occurs in bone marrow.
Common Lymphoid Progenitor
A type of hematopoietic stem cell that gives rise to lymphocytes, including B cells, T cells, and natural killer (NK) cells. Also produces Dendritic Cells (DCs)
Common Myeloid Progenitor
A type of HSC that gives rise to myeloid cells, including red blood cells, platelets, and various types of white blood cells such as granulocytes, monocytes, and mast cells. Also produces DCs.
Cluster of Differentiation (CD)
Molecules present on the surface of lymphocytes. Denoted as CD#.
B-Cell Receptor (BCR)
Can be membrane-bound or secreted as an antibody. Can recognize antigens.
T-Cell Receptor (TCR)
Membrane-bound protein that recognizes antigens presented by an MHC.
Antigen (Ag)
Molecule that an immune response is generated against. Contains an epitope.
Epitope
The fragment of the antigen that is recognized and presented to T-Cells.
Pattern Recognition Receptors (PRRs)
Proteins that recognize PAMPs to differentiate between self and non-self cells.
Pathogen Associated Molecular Patterns (PAMPs)
Common foreign structures that characterize whole groups of pathogens.
Innate Immunity
Fast, nonspecific, first line of defense. Uses germline encoded receptors to recognize pathogens with identical responses each time.
Adaptive Immunity
Slower, specific reaction that incorporates B- and T-cells to clear infection and form “memory”.
Cytokine
Secreted messanger proteins.
Chemokine
A type of cytokine that specifically attracts immune cells to sites of infection or inflammation.
Hallmarks of Inflammatory Response
Heat, swelling, redness, and pain
Antigen Presenting Cells (APCs)
Cells that display antigens on their surface to activate T cells. Includes dendritic cells, macrophages, B cells, and most nucleated cells.
Major Histocompatibility Complex (MHC)
Presents a processed antigen epitope to TCRs.
Antibody
Secreted Immunoglobulin molecule that recognizes and binds to the epitope of antigens. Produced by B cells and circulates in the serum.
Difference Between B and T cells Origin
Both arise in bone marrow, but T cells mature inside of the thymus.
How many antigens can a receptor be specific to?
1
Humoral Immunity
Production of antibodies by B cells to clear and/or neutralize antibody.
Cell-mediated Immunity
Meditated by T cells that may boost activity of macrophages, attract and activate B cells, or directly kill infected cells.
Active Immunization
The process of inducing immunity in an individual by exposing them to a pathogen or its components, leading to the production of antibodies and memory cells. Can happen naturally through infection or be induced with vaccines.
Passive Immunization
The transfer of pre-formed antibodies to an individual, providing immediate but temporary immunity. This can occur naturally, such as through maternal antibodies, or artificially via antibody-containing serum.
1st Level of Innate Immunity
Mucosal and epithelial surfaces. Tend to be acidic and produce antimicrobials.
2nd Level of Innate Immunity
Cellular elements such as neutrophils, granulocytes, monocytes, macrophages, and dendritic cells.
Phagocytosis
Engulfment and internalization of pathogens. Conducted by macrophages, DCs, granulocytes, and neutrophils.
Antimicrobial Enzyme vs Peptide
Antimicrobial enzymes are proteins that inhibit microbial growth, while antimicrobial peptides are short sequences of amino acids that disrupt microbial membranes. ex: Lysozymes (enzyme) digest peptidoglycan and defensins (peptide) disrupt the cell membrane.
Opsonins
Soluble proteins bound to microbe surfaces that promote phagocytosis (opsonization).
Phagolysosome
Formed by the fusion of pathogen containing phagosome with lysosome. Contains an acidic environment, hydrolytic enzymes, ROS and RNS, and antimicrobial peptides.
Reactive Oxygen Species (ROS)
Conducts oxidative attack, generated by NADPH oxidase enzyme complex.
Which cell type contains primary and secondary granules?
Neutrophils
Damage-Associated Molecular Patterns (DAMPs)
Molecules released by damaged or dying cells that alert the immune system to tissue injury and promote inflammation. Bind to PRRs.
CD47
“Don’t Eat Me” Signal, prevents phagocytosis of healthy cells
Macrophage
Tissue specific resident phagocyte. Called microglia in CNS.
Neutrophil
Non-tissue resident (circulating) phagocyte. 20-60% form neutrophil extracellular traps that trap microorganisms aand prevent spread.
Disease caused by loss of microglia
Multiple Sclerosis
Complements
Proteins in the blood that enhance both immune responses, aiding in the opsonization and destruction of pathogens. Most composed of proteasomes.
3 Key Complement Mechanisms
Promote vascular permeability and chemotaxis
Destroying pathogen cell membrane (MAC)
Increase pathogen recognition (opsonization)
3 Complement Activation Pathways
Classical
Alternative
Lectin
Classical Pathway
C1q binds to pathogen surface OR antibodies bind pathogen
C1q binding generates C3 convertase
Alternative Pathway
Version 1: C3b produced by Lectin/Classical pathway used in amplification loop with factors B and D.
Version 2: When C3 concentration is high, it undergoes spontaneous hydrolysis with factors B and D.
Alternative pathway C3 convertase is unstable, can be stabilized with factor P from neutrophils
Lectin Pathway
Triggered by soluble PRRs called lectins that bind to specific carbohydrates on pathogens, leading to the activation of complement proteins and formation of C3 convertase.
C3
Converted into C3a and C3b. C3a increases inflammation, and C3b increases opsonization and C5 convertases. All pathways converge on C3
C3 Convertases
C4b2a and C3bBb
C5
Converted into C5a and C5b. C5a is a chemoattractant while C5b initiates the formation of the membrane attack complex.
C5 Convertase
C3b
Toll-Like Receptors (TLRs)
Membrane bound PRRs that recognize both intracellular and extracellular PAMPs. Binding activates a signal cascade that leads to the production of transcription factors NF-kB, IRF, and AP-1.
C-type Lectin Receptors (CLRs)
Membrane bound PRR that binds to carbohydrates on pathogens and allergens. Activated tyrosine kinases trigger signalling cascade.
Rig-I-Like Receptors (RLRs)
Cytosolic PRRs that recognize dsRNA and some ssRNA. Mitochondrial antiviral signallers that active IRFs and NF-kB.
Nod-Like Receptors (NLRs)
Cytosolic PRRs that detect bacterial peptidoglycans , activates caspase-1-protease. Triggers NF-kB, AP-1, and IRF.
Signal Transduction
Ligand induced receptor dimerization
Recruitment/Activation of kinases and adaptors
Secondary messangers
Activation/Nuclear translocation of transcription factors
Changes in gene expression
Post-translational modifications
Functional response
Effects from PRR Signaling
Cytokine production
Chemokine production
Increased expression of costimulatory molecules
Enhanced DC migration to secondary lymphoid
Steps of Innate Cell Migration
Rolling adhesion
Tight binding
Diapedesis
Migration
Adhesion Molecules
Integrins, Selectins, Immunoglobulin superfamily
Cytokines
Small glycoproteins that provide cellular communication on an auto, para, and endocrine level. Can cascade and induce the production of more cytokines. May lead to activation activation, proliferation, death, differentiation, and expression.
5 Groups of Cytokines
Interleukins, Interferons, Tumor Necrosis Factors, Chemokines, and Hematopeoitins (growth factors)
Pleiotropy
One cytokine producing multiple effects
Redundancy
More than one cytokine inducing the same effect
Synergy
Two+ cytokines work together to induce an effect
Antagonism
One cytokine inactivates the effect of another
Cell-Mediated Response
Targets viruses and intracellular parasites, characterized by T-helper cells and cytotoxic T cells.
Humoral Response
Targets extracellular pathogens, characterized by B cell activation and antibody production.
Tumor Necrosis Factors (TNF)
A group of cytokines involved in systemic inflammation and the regulation of immune cells.
TNF-α
A pro-inflammatory cytokine produced mainly by macrophages. Stimulates migration of innate cells and dilates blood vessels. Can cause local or systemic effects.
Acute Phase Response
Change in proteins present in blood during the early phases of infection. Includes increase synthesis and secretion of antimicrobial proteins (complements, mannose-binding lectin, and c-reactive proteins).
Which cytokines induce fever?
TNF-α, IL-1, and IL-6 signal the hypothalamus to increase body temperature.
Natural Killer Cells (NK)
Innate lymphoid cell (ILC) that can be found in tissue or circulation. Directly kills altered self cells (infected or tumors).
NK Cell Receptors
Germline encoded, activating and inhibitory receptors. Recognizes MHC 1 and MHC 1-like molecules that prevent the NK from killing the cell.
Conventional Dendritic Cell (cDC)
“Classic” APC that travels to the the lymph to activate T-cells.
Plasmocytoid Dendritic Cell (pDC)
Very high levels of PRRs, stays at site of infection to secrete type 1 IFNs and cytokines.
Lymphoid Tissues
Lymph nodes, spleen, and Peyer’s patches
Afferent vs. Efferent Lymphatics
Activated DCs enter via afferent lymphatics.
T and B cells exit via efferent lymphatics to travel to infected tissue.
How do T and B cells enter lymph nodes?
High Endothelial Venules (post capillary blood vessels)
T-Cell Entry Into Lymph Nodes
Rolling (selectin mediated)
Activation (chemokine mediated)
Arrest and Tight Binding (integrin mediated)
Diapedesis
Fibroblastic Reticular Cells
Secrete CCL19/21 chemokines that help attract T-Cells and DCs to lymph nodes.
T-Cell Activation Pathway
APC activation
Antigen entry to lymph node
TCR:MHC interaction
T-Cell proliferation
CD8+
Becomes Cytotoxic T Lymphocytes (CTLs). Recognizes Ag on MHC1
CD4+
Becomes Helper T cells. Recognizes Ag in MHC2. Five subcategories:
TH1
TH2
TH17
TREG
TFH
TCR Complex
TCR, DC3, Zeta Chain, and ITAMs (immunoreceptor tyrosine-based activation motifs)
TCR Recognition Subunit
Membrane bound, clonotypic heterodimer of α and β chains
α chain: Variable (V), Joining (J), and Constant (C) domains
β chain, V, Diversity (D), J, and C domains
Where does TCR rearrangement occur?
In the Thymus
Professional APC
Cells that express MHC 1 and 2 molecules and costimulatory molecules. DCs, macrophages, and activated B-Cells.
Non-Professional APC
Cells that do not express MHC class II molecules or provide costimulatory signals. Almost all nucleated cells in the body.
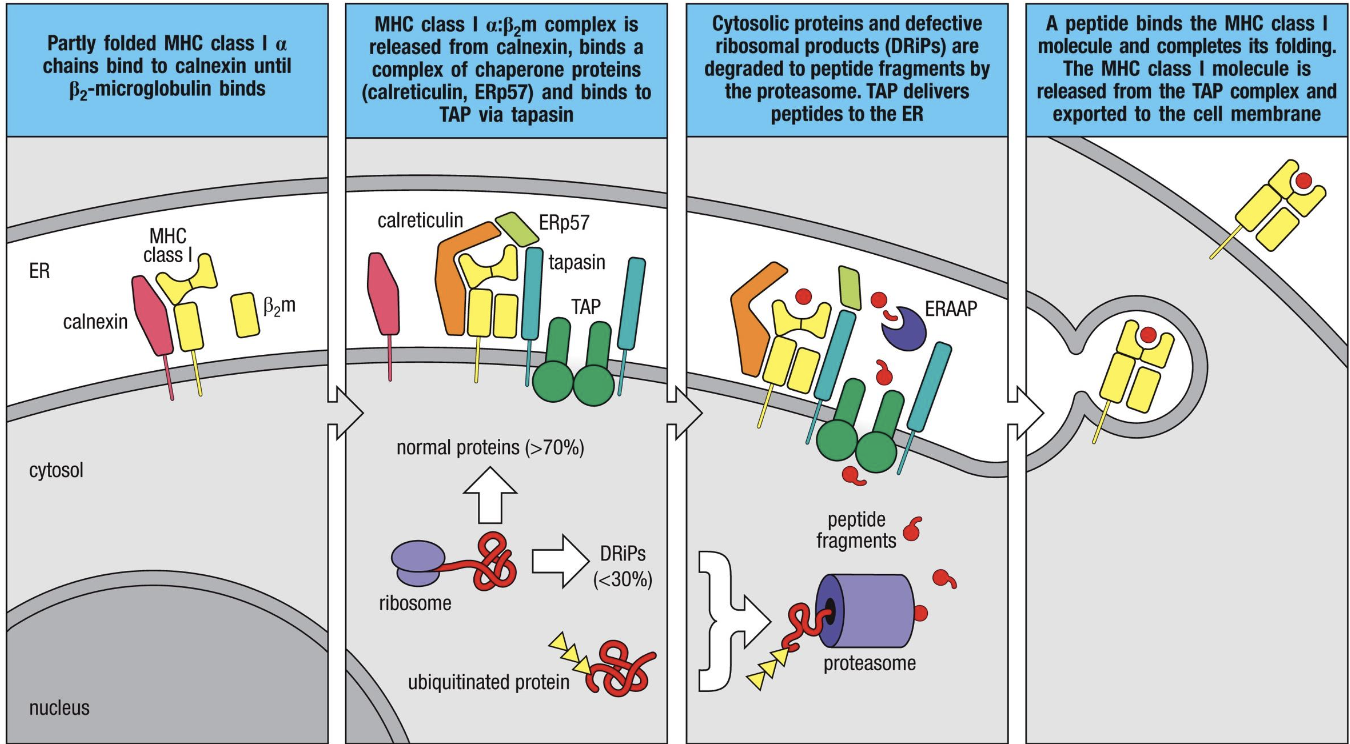
MCH Class 1
Binds and presents peptides generated within the cell (including self). Made of α chain (transmembrane) and β2-microglobulin. It is recognized by CD8+ T cells.
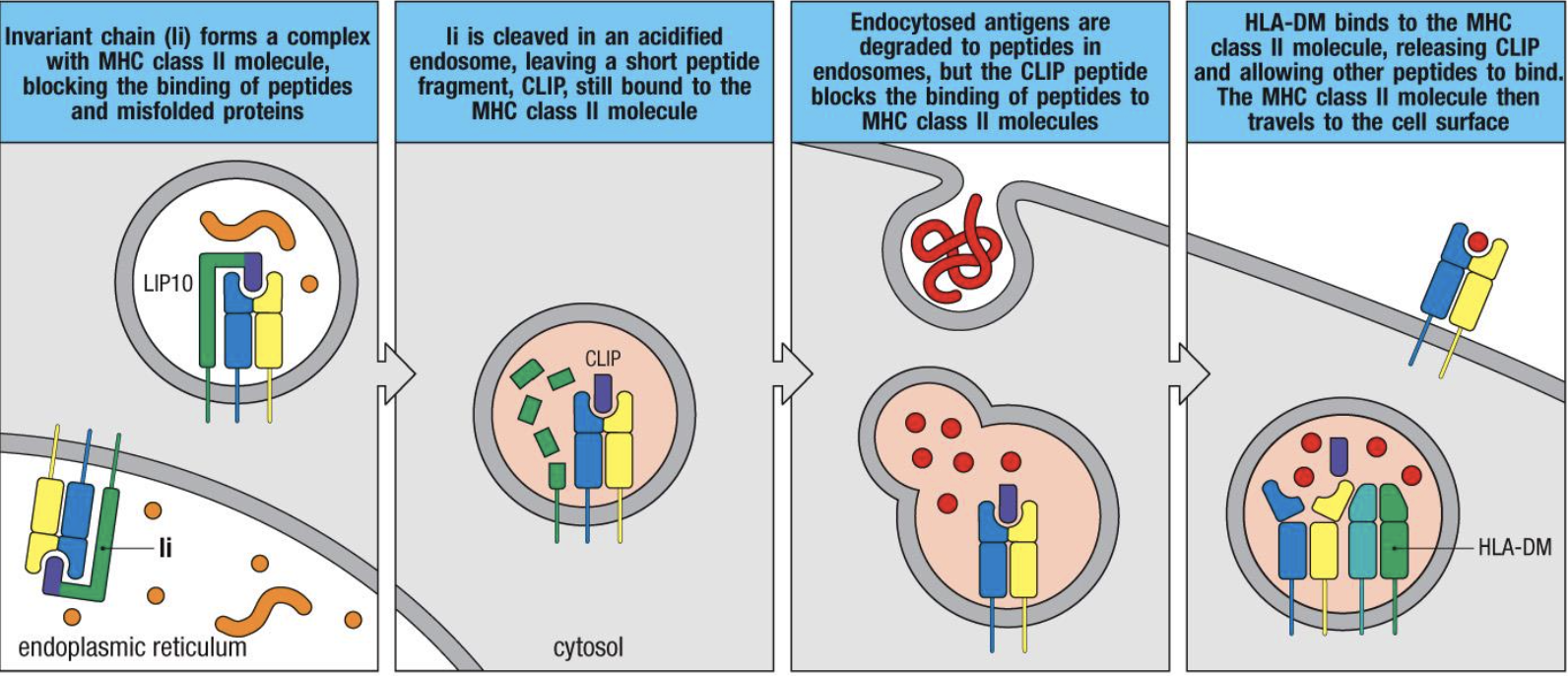
MHC Class 2
Binds and present extracellular peptides. Made of α and β chains that both have transmembrane elements. Recognized by CD4+ T cells.
Immunoglobulin (Ig) Like Domains
Secondary structure in each chain consisting of 100+ amino acids stabilized by intrachain disulfide bonds.
Size of Peptides Bound by MHC 1
8-10 AAs
Size of Peptides Bound by MHC2
13+ AAs
Endogenous Pathway (MHC1)
Entry of pathogen is pathogen mediated (virus, intracellular pathogen)
Exogenous Pathway (MHC2)
Entry of pathogen is meditated by immune cell (phagocytosis)
Cross-Presentation
Requires a “license” from CD4+ helper T cells, and can only be done by DCs. Allows for presentation of peptides on MHC1 to CD8+.
MHC2 Cytosolic Peptide Presentation
Requires breakdown of cell’s own organelles in autophagosome that allows endogenous peptides to be expressed through the exogenous pathway (MHC2). Helpful against resistant intracellular pathogen and certain viruses.
Allorecognition
Recognition of non-self MHC by up to 10% of T cells. May be direct (recipient T cell recognizes MHC on donor/transplant APC) or indirect (recipient APC processes donor peptides).
MHC1 Genes
Human Leukocyte Antigen (HLA) - A, -B, and -C
MHC2 Genes
HLA-DR, -DQ, -DP, -DM, -DOA, -DOB