Chem 1: Molecular + Electron Geometry, Polarity, Bonds
1/35
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
36 Terms
Is a linear geometry polar or non-polar?
Usually non-polar
Is a bent geometry polar or non-polar?
Always polar
Is a trigonal planar geometry polar or non-polar?
Non-polar if all B are the same (AB3)
Is a trigonal pyramidal geometry polar or non-polar?
Always polar due to lone pair
Is a tetrahedral geometry polar or non-polar?
Nonpolar if all B are the same (AB4)
Is a trigonal bipyramidal geometry polar or non-polar?
Nonpolar if all B are the same (AB5)
Is an octahedral geometry polar or non-polar?
Nonpolar if all B are the same (AB6)
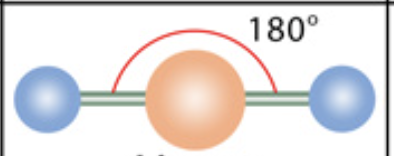
What is the molecular geometry?
Linear
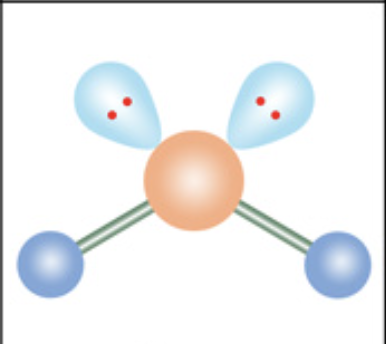
What is the molecular geometry?
Bent / angular
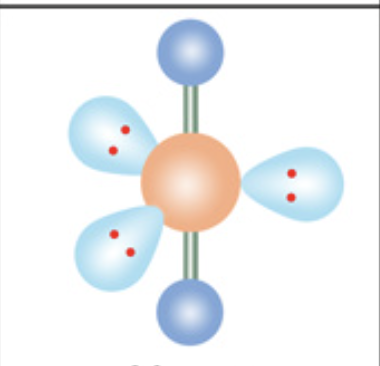
What is the molecular geometry?
Linear
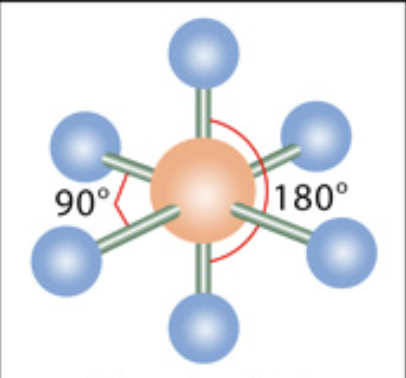
What is the molecular geometry?
Octahedral
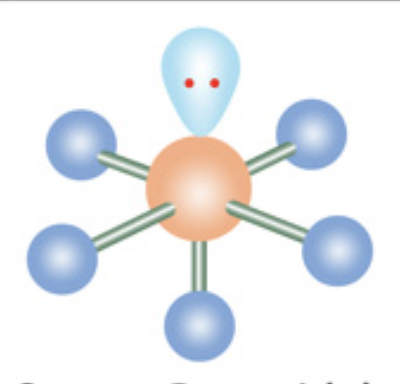
What is the molecular geometry?
Square pyramidal
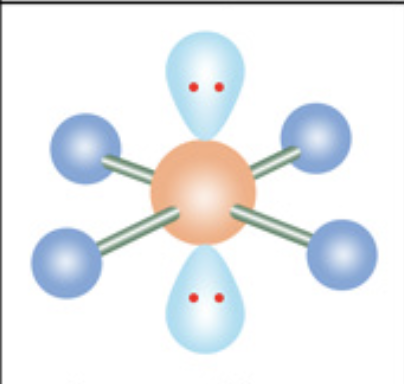
What is the molecular geometry?
Square planar
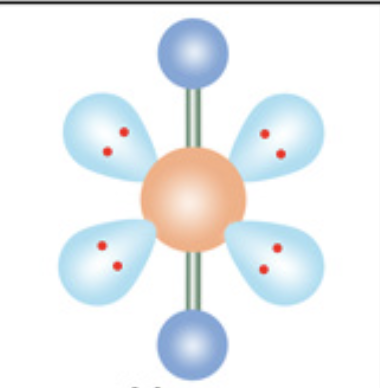
What is the molecular geometry?
Linear
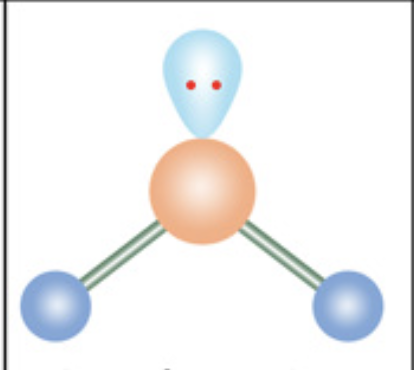
Bent / angular
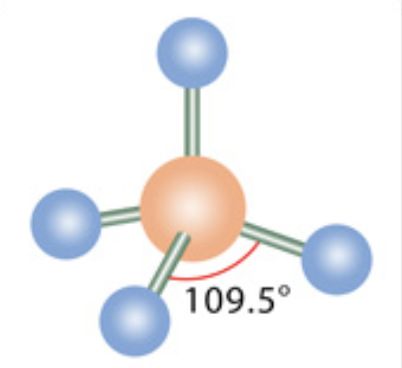
What is the molecular geometry?
Tetrahedral
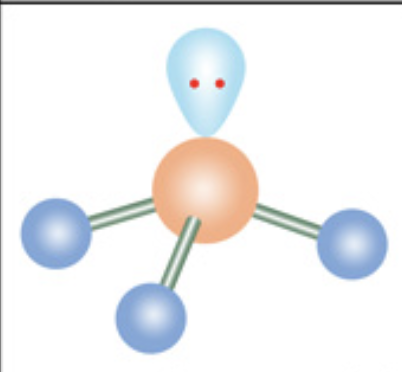
What is the molecular geometry?
Trigonal pyramidal
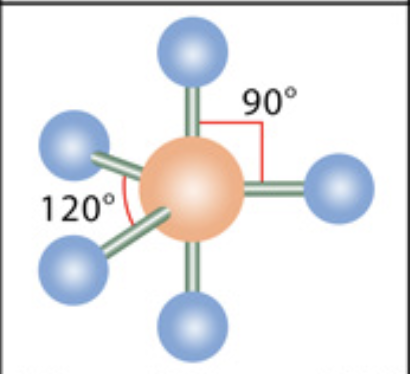
What is the molecular geometry?
Trigonal bipyramidal
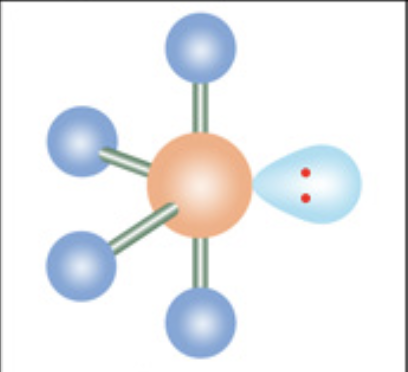
What is the molecular geometry?
Seesaw
What molecular shape has a bond angle of: 180?
Linear
What molecular shape has a bond angle of: 120?
Trigonal planar
What molecular shape has a bond angle of: 109.5?
Tetrahedral
What molecular shape has a bond angle of: 120 and 90?
Trigonol bipyramidal
What molecular shape has a bond angle of: 90 and 180?
Octahedral
What molecular shape has a bond angle of: less than 180?
Bent
Are noble gases polar or nonpolar?
nonpolar
Are diatomic molecules of the same element polar or nonpolar?
nonpolar
Are monoatomic molecules polar or nonpolar?
nonpolar
Are molecular structures with symmetry polar or nonpolar?
nonpolar
If Hydrogen is attached to N, O, and/or F, is the molecule polar or nonpolar?
polar
If a molecule is composed of only C and H, is it polar or nonpolar?
nonpolar
Is CH3OH polar or nonpolar?
polar
If a lewis structure lacks symmetry, is it polar or nonpolar?
polar
H2O, polar or nonpolar?
Polar, due to the bent shape
Electron geometry includes the lone pairs, true or false?
true
Does molecular geometry include or exclude lone pairs?
exclude