Energy, Thermodynamics, and Cell Metabolism in Biology
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
What is matter?
Material that takes up space and has mass or weight.
What is energy?
The ability to do work or cause change.
What are the forms of energy?
Potential energy, chemical energy, and kinetic energy.
What is potential energy?
Stored energy that has the potential to do work, derived from position or arrangement.
Give an example of potential energy.
Glycogen or water behind a dam.

What is chemical energy?
A form of potential energy stored in chemicals/molecules due to the position of atoms.
Why is chemical energy important for living organisms?
It is the most important type of energy for living organisms, as it is available for release in chemical reactions.
What is kinetic energy?
The energy of movement, including the random movement of atoms and molecules.
What governs energy use?
Thermodynamics, which studies energy transformations.
What is the First Law of Thermodynamics?
Energy in the universe is constant; it can be transferred and transformed but cannot be created or destroyed.
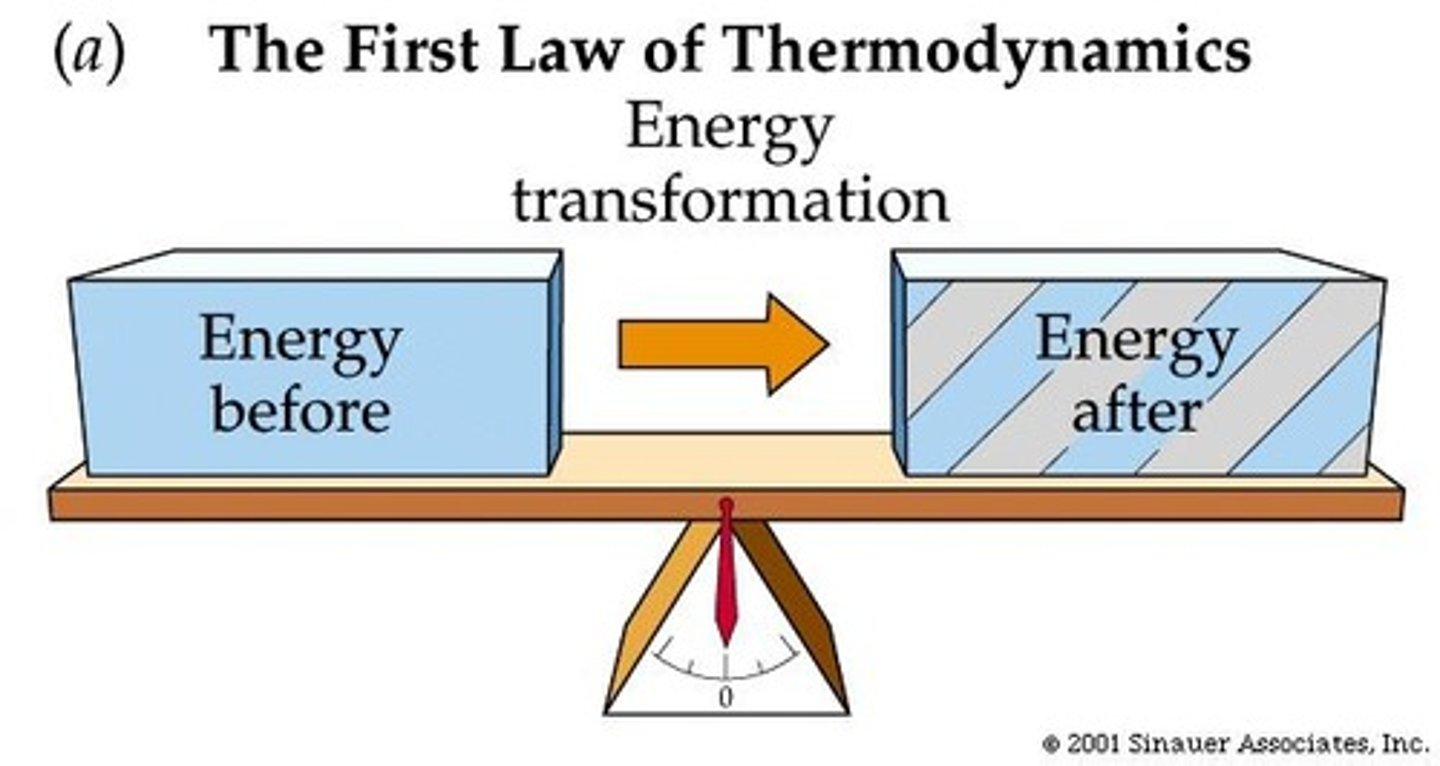
What does the First Law of Thermodynamics imply?
Energy quantity never changes; it remains constant.
What is the Second Law of Thermodynamics?
Entropy always increases; energy quality in the universe is not constant.
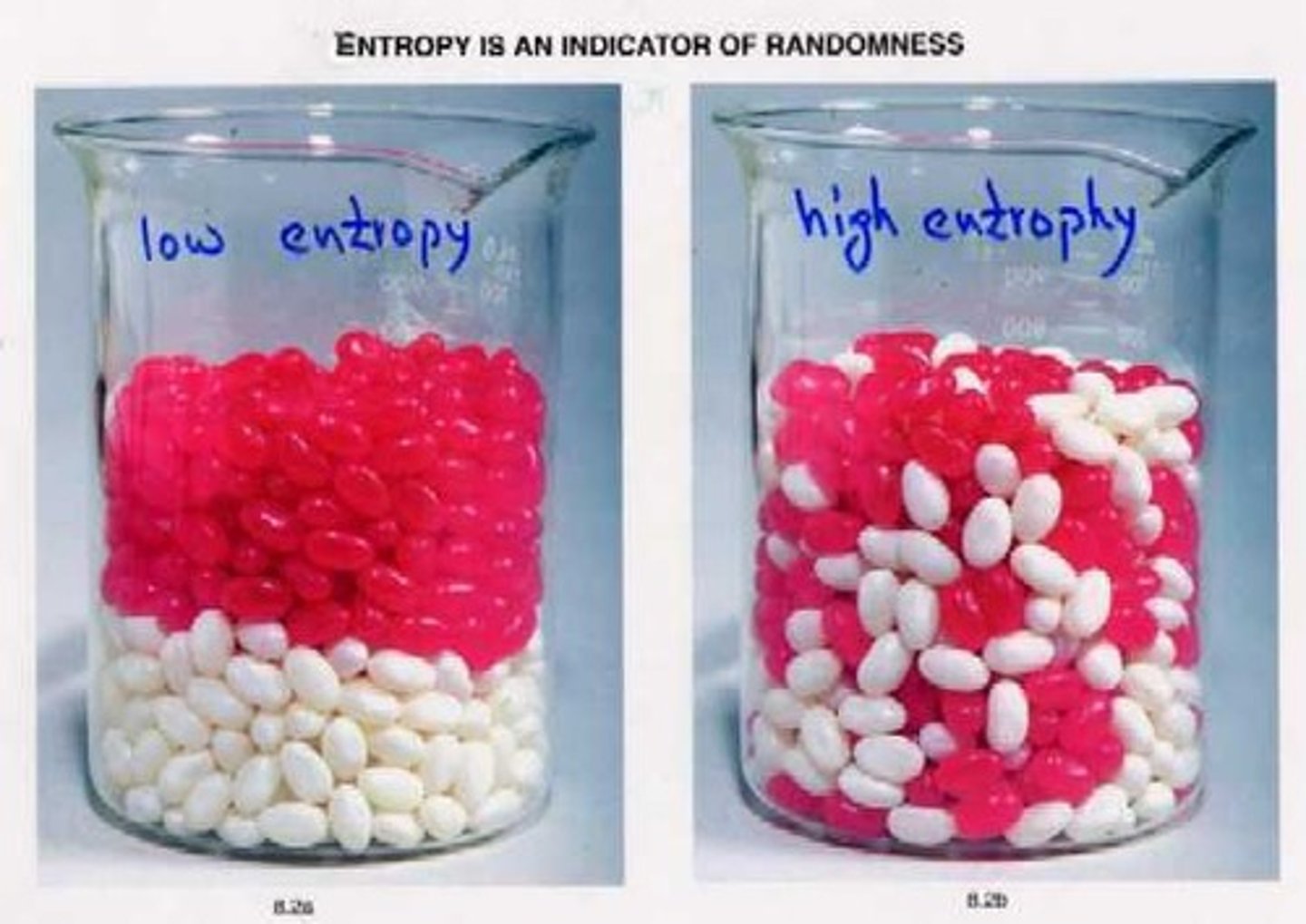
What happens to useful energy during transformations?
Some useful energy is lost as heat, leading to a decline in energy quality.
What is entropy?
A measure of disorder or randomness that increases spontaneously.
What characterizes high entropy?
More disorder and randomness.
What characterizes low entropy?
Less disorder and randomness.
How does entropy relate to the universe?
Processes that increase entropy occur spontaneously without energy required.
What is an example of high entropy?
A group of people in a chaotic environment.
What is an example of low entropy?
A neatly arranged set of objects.
What is the efficiency of energy conversion in the human body?
Energy conversion is not 100% efficient, leading to energy loss as heat.
What are the four major macromolecules?
carbs, lipids, nucleic acids, and proteins.
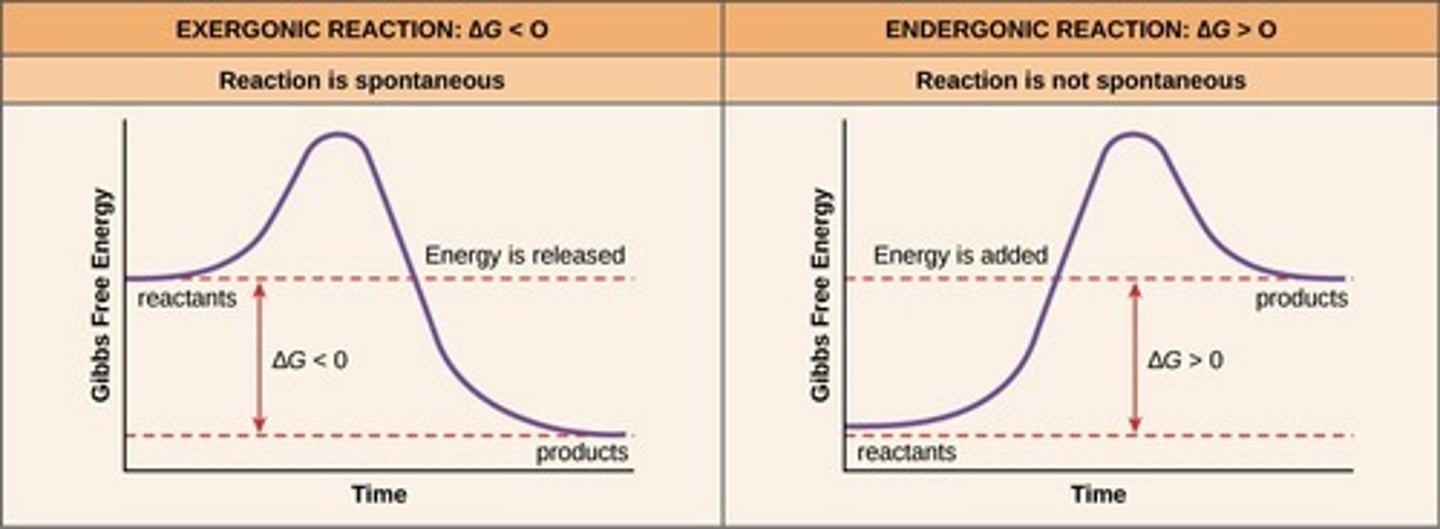
What is the primary difference between exergonic and endergonic reactions?
Exergonic reactions release energy, while endergonic reactions require energy input.
What type of reaction is cellular respiration?
Exergonic reaction (catabolic), as it releases energy by breaking down glucose.
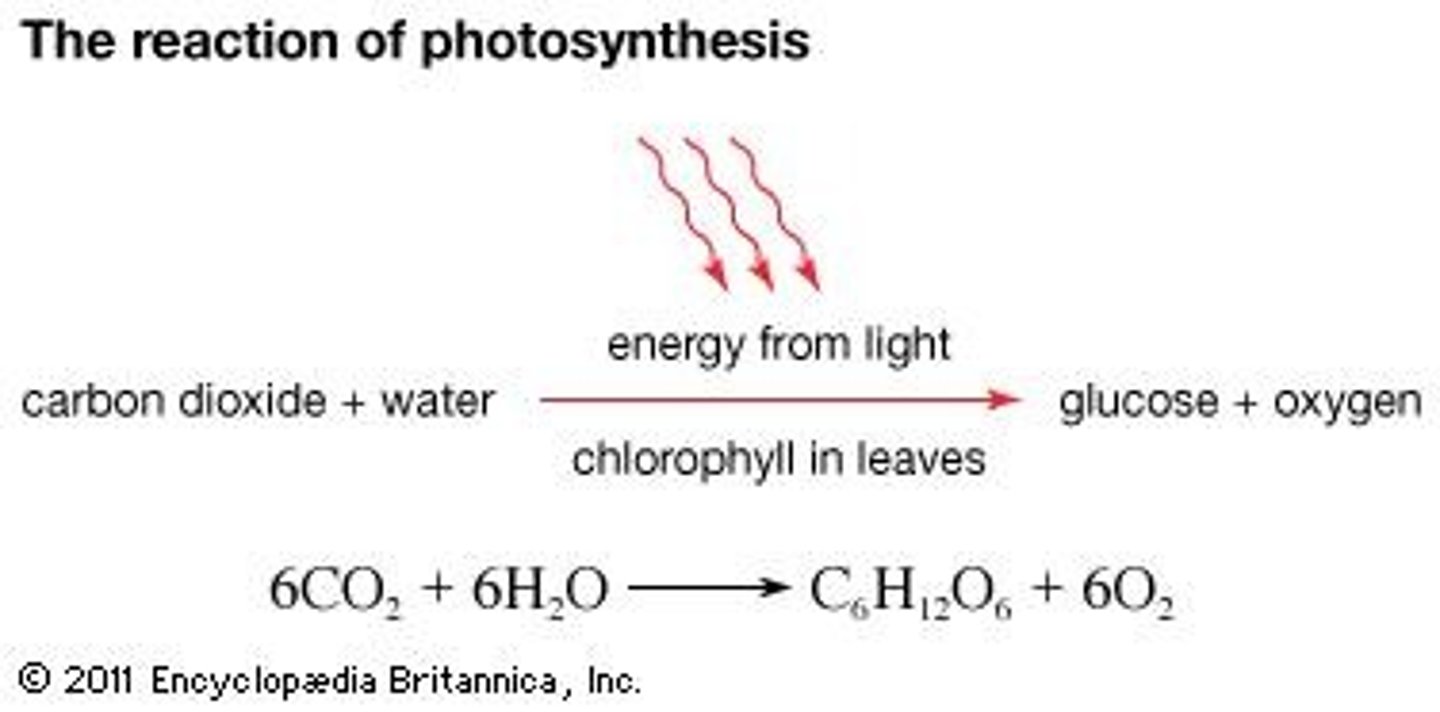
What type of reaction is photosynthesis?
Endergonic reaction (anabolic), as it requires energy to synthesize glucose from CO2 and H2O.
What does ΔG represent in chemical reactions?
ΔG represents the change in free energy, indicating the amount of energy released or consumed.
What is the role of catalysts in chemical reactions?
Catalysts increase the rate of a chemical reaction without being consumed, by lowering the activation energy.
What is the energy of activation (EA)?
The kinetic energy needed for reactants to reach the transition state and start a reaction.
What happens to the potential energy of reactants in exergonic reactions?
Reactants contain more potential energy than the products.
What happens to the potential energy of reactants in endergonic reactions?
Products contain more potential energy than the reactants.
What is metabolism?
Metabolism is all of the chemical reactions occurring in a cell or organism.
What is an example of an exergonic reaction?
Cellular respiration, where glucose is broken down to release energy.
What is an example of an endergonic reaction?
Photosynthesis, where energy is consumed to form glucose.
How do cells maintain organization?
Cells obtain and use energy through chemical reactions.
What is dehydration synthesis?
An endergonic reaction that builds larger molecules by removing water.
What is the significance of Hill Diagrams?
Hill Diagrams help visualize the energy changes and kinetics in chemical reactions.
What occurs during the transition state in a chemical reaction?
Bonds in reactants break and new bonds in products start to form, representing a high energy point.
What is the relationship between temperature and reaction rate in cells?
Increasing temperature generally increases particle movement, leading to a higher reaction rate.
How does increasing surface area affect reaction rates?
More surface area allows more particles to collide, increasing the likelihood of reactions.
What happens when the concentration of reactants is increased?
Higher concentration increases the likelihood of collisions between reactants, speeding up the reaction.
What is the main function of enzymes in biological reactions?
Enzymes act as catalysts to speed up reactions without being consumed, often under mild conditions.
What is the spontaneous direction of a reaction?
The direction in which a reaction occurs without needing an energy input.
What is the difference between catabolic and anabolic reactions?
Catabolic reactions break down molecules and release energy, while anabolic reactions build molecules and require energy.
What is the relationship between order and entropy in chemical reactions?
Reactions that increase order (like photosynthesis) are associated with a decrease in entropy, requiring energy input.
What is the significance of energy release in exergonic reactions?
Energy release makes it available for cellular processes and activities.
What is the role of energy in maintaining cellular organization?
Energy is required to maintain the high level of organization within cells.
What are reactants or substrates in a chemical reaction?
Reactants or substrates are the starting materials that undergo a chemical change.
What are products in a chemical reaction?
Products are the substances formed as a result of a chemical reaction.