Chapter 2 Bonding and Structure of Organic Compounds - Until Index of Hydrogen Deficiency
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
Ionic
Covalent
Ionic Compound
Covalent Compound
__________ bond – electrons are transferred from one atom to another
__________ bond – one or more pairs of electrons are shared between atoms
__________ compounds contain charged particles held together by extremely strong electrostatic interactions.
__________ - It is a compound whose structural units are called molecules. A molecule is a neutral collection of atoms held together by covalent bonds.
Bond length
Bond strength
Bond angle
Bond polarity
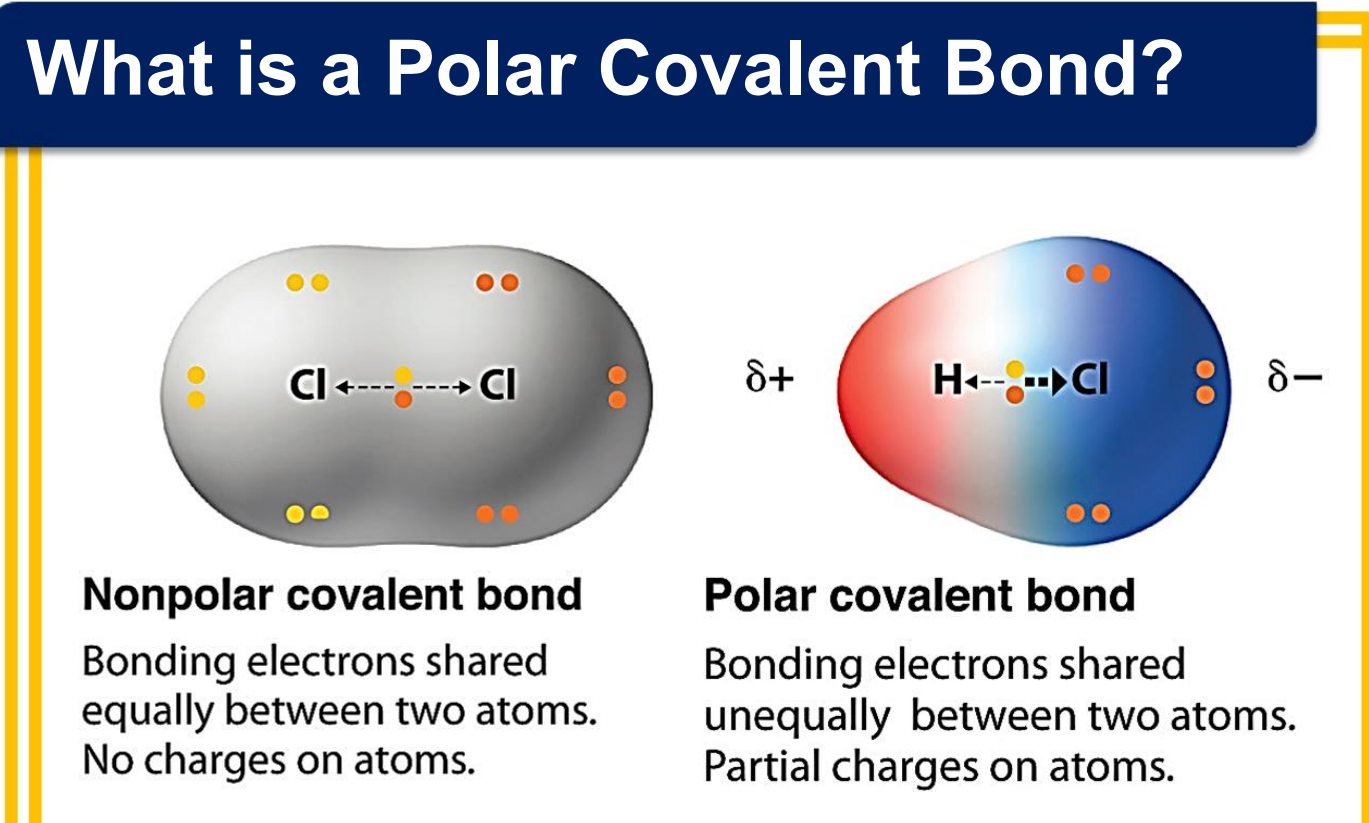
_____________ - the equilibrium distance between the nuclei of two atoms that are bonded to each other
_____________ - alternative name for bond dissociation energy - the amount of energy needed to break a bond homolytically and produce two radical fragments
_____________ - the angle formed between two adjacent bonds
_____________ - a measure of the distribution of electrons between two bonded atoms
consumed; liberated
Bond strength
the amount of energy ___________ when a bond is broken is equal to the amount of energy ____________ when a bond is formed
Electronegativity


_________________ - intrinsic ability of an atom to attract the shared electrons in a covalent bond
Dispersion or London Forces


polarizability
surface area
Polarizability

Factors that affect the magnitude of London dispersion forces:
a. relative ______________ of electrons of the atoms involved
b. relative ______________ of the molecules involved
_____________ – a measure of how the electron cloud around an atom responds to changes in its electronic environment
Dipole-dipole

_________________ forces - an interaction between molecules having permanent dipole moments
Hydrogen bonds
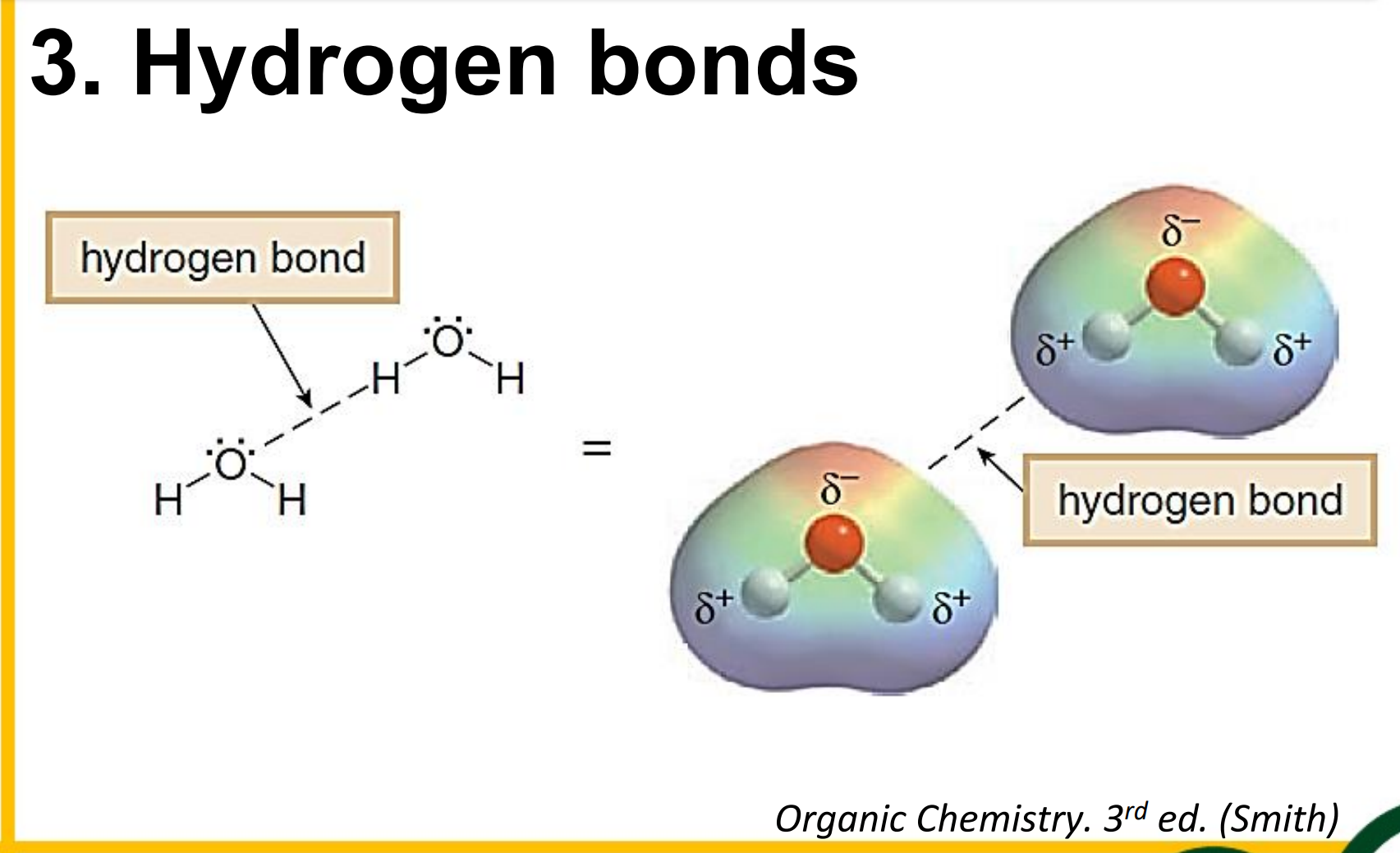
________________ - a strong dipole-dipole interaction that occurs between hydrogen atoms bonded to small strongly electronegative atoms and the nonbonding electron pairs on other such electronegative atoms
Quantum Mechanical Model of the Atom
_________________________ - the motion of an electron around a nucleus can be described mathematically by a wave equation
orbital
shell
node

___________ - a wave function which describes the volume of space around a nucleus in which an electron is most likely to be found
_____________ - a group of an atom’s electrons with the same principal quantum number
________ - region of zero electron density
Aufbau
Pauli exclusion
Hund’s rule
___________ principle - rules for determining the electron configuration of an atom
_______________ principle - no more than two electrons can occupy the same orbital, and those two electrons must have spins of opposite sign
______________ - if two or more empty orbitals of equal energy are available, one electron occupies each, with their spins parallel, until all are half-full
Valence Bond Theory (VBT)

Key ideas of VBT
a) covalent bonds are formed by overlap of two atomic orbitals, each of which contains one electron
b) each of the bonded atoms retains its own atomic orbitals, but the electron pair in the overlapping orbitals is shared by both atoms
c) the greater the amount of orbital overlap, the stronger the bond
________________ - a covalent bond results when two atoms approach each other closely so that a singly occupied orbital on one atom overlaps a singly occupied orbital on the other atom
Molecular Orbital Theory (MOT)

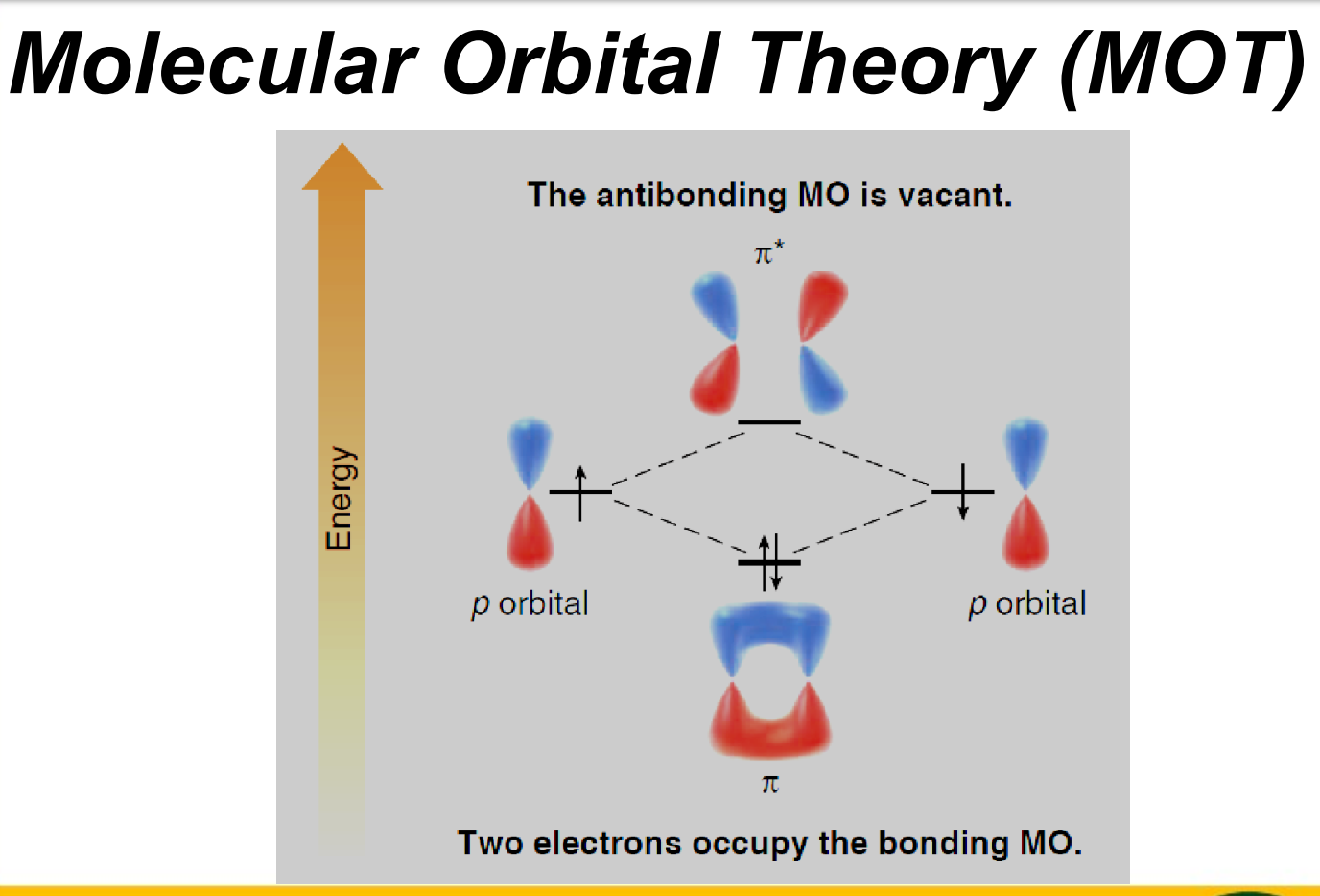
Key ideas of MOT
a) molecular orbitals are to molecules what atomic orbitals are to atoms
b) molecular orbitals are formed by combining atomic orbitals
the number of molecular orbitals formed is equal to the number of atomic orbitals combined
c) relative energies of molecular orbitals:
bonding MO (lower E than AO)
nonbonding MO (same E as AO)
antibonding MO (higher E than AO)
__________________ - describes covalent bond formation as arising from a mathematical combination of atomic orbitals to form molecular orbitals
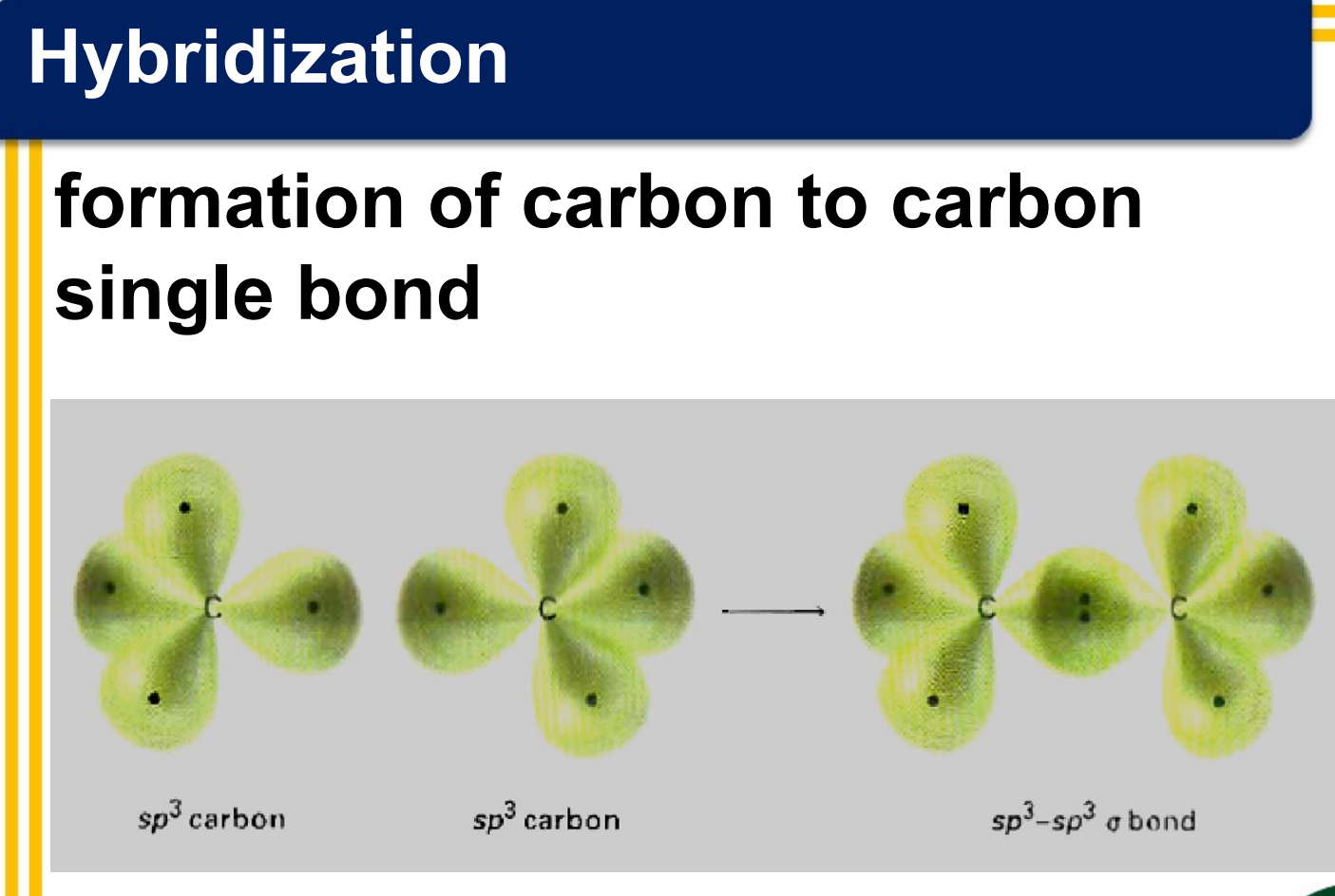
Hybridization


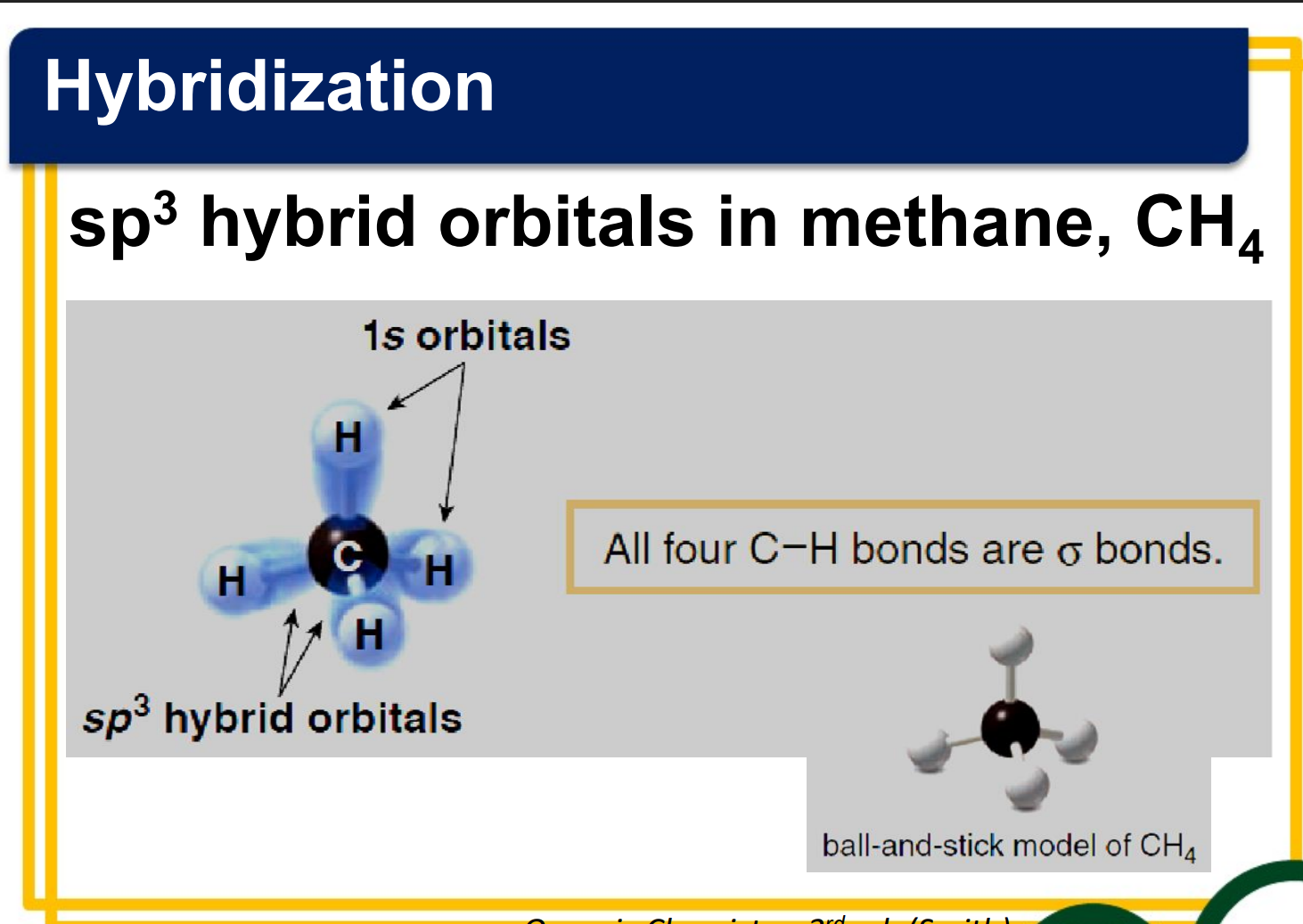




______________- combination of two or more atomic orbitals to form the same number of hybrid orbitals, each having the same shape and energy
sp2 hybridization

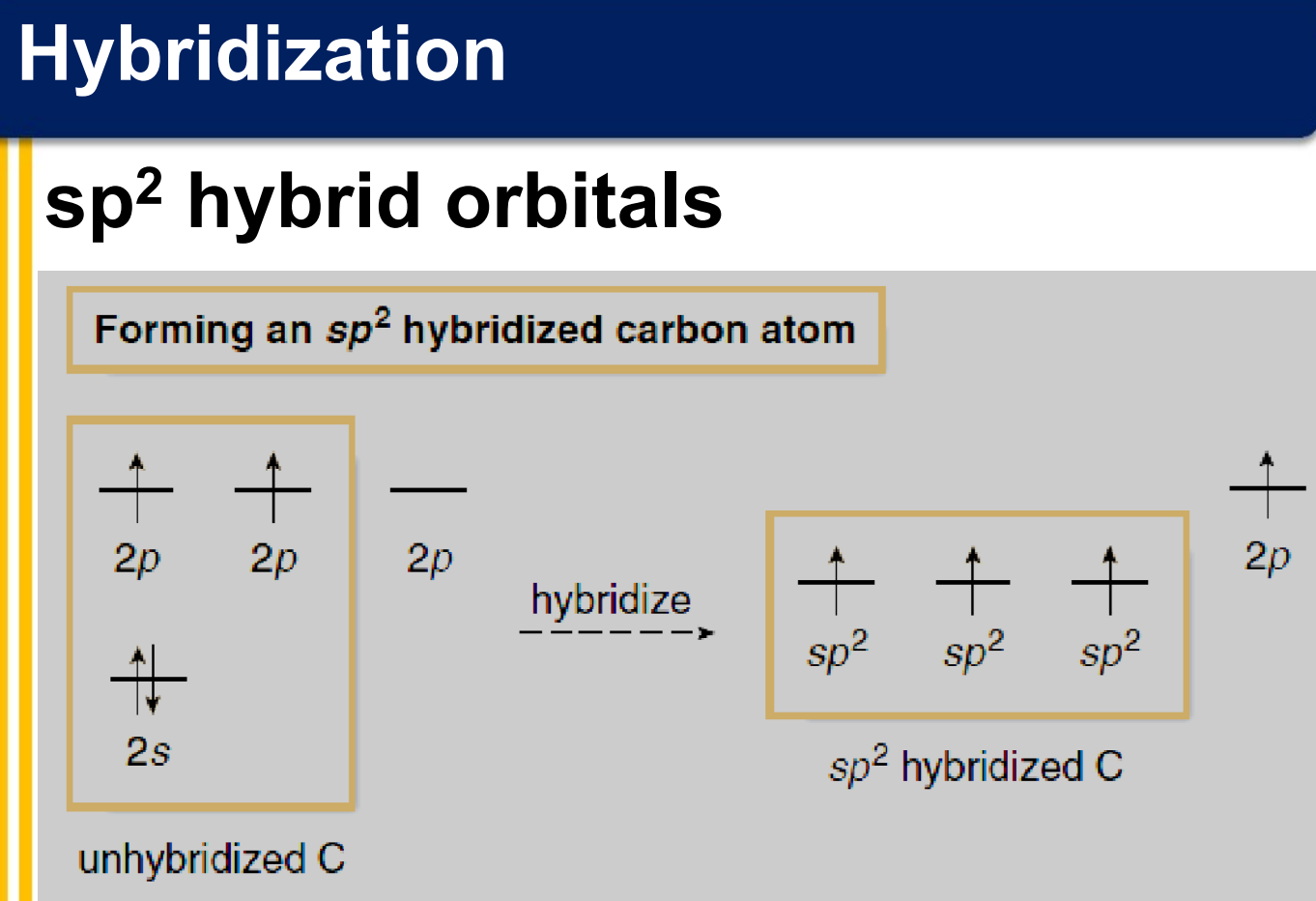




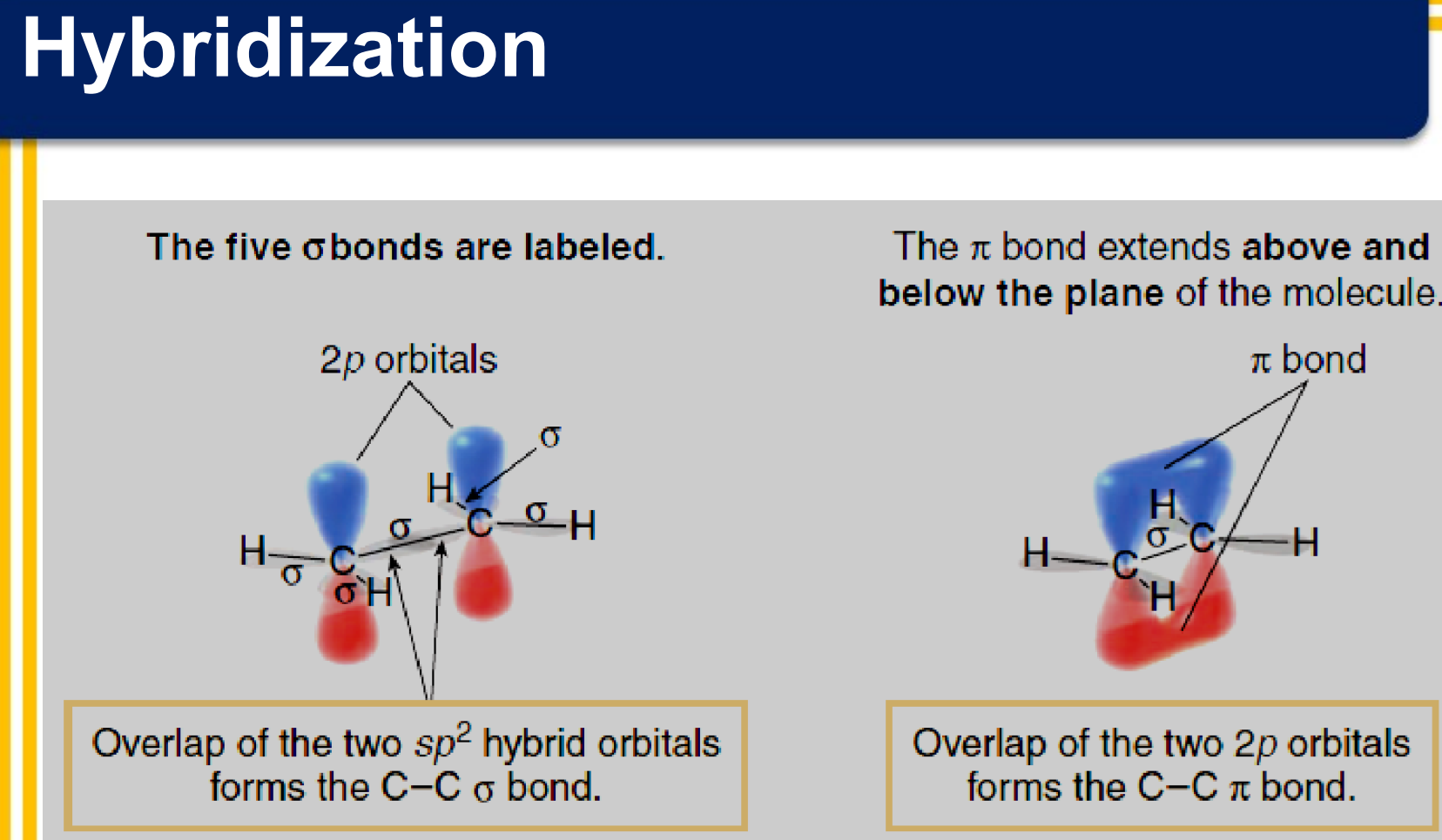
There are two covalent bonds formed between the two carbon atoms of ethene:
one σ bond from the end-on overlap of two sp2 hybrid orbitals
one π bond from the side-to-side overlap of two 2p orbitals
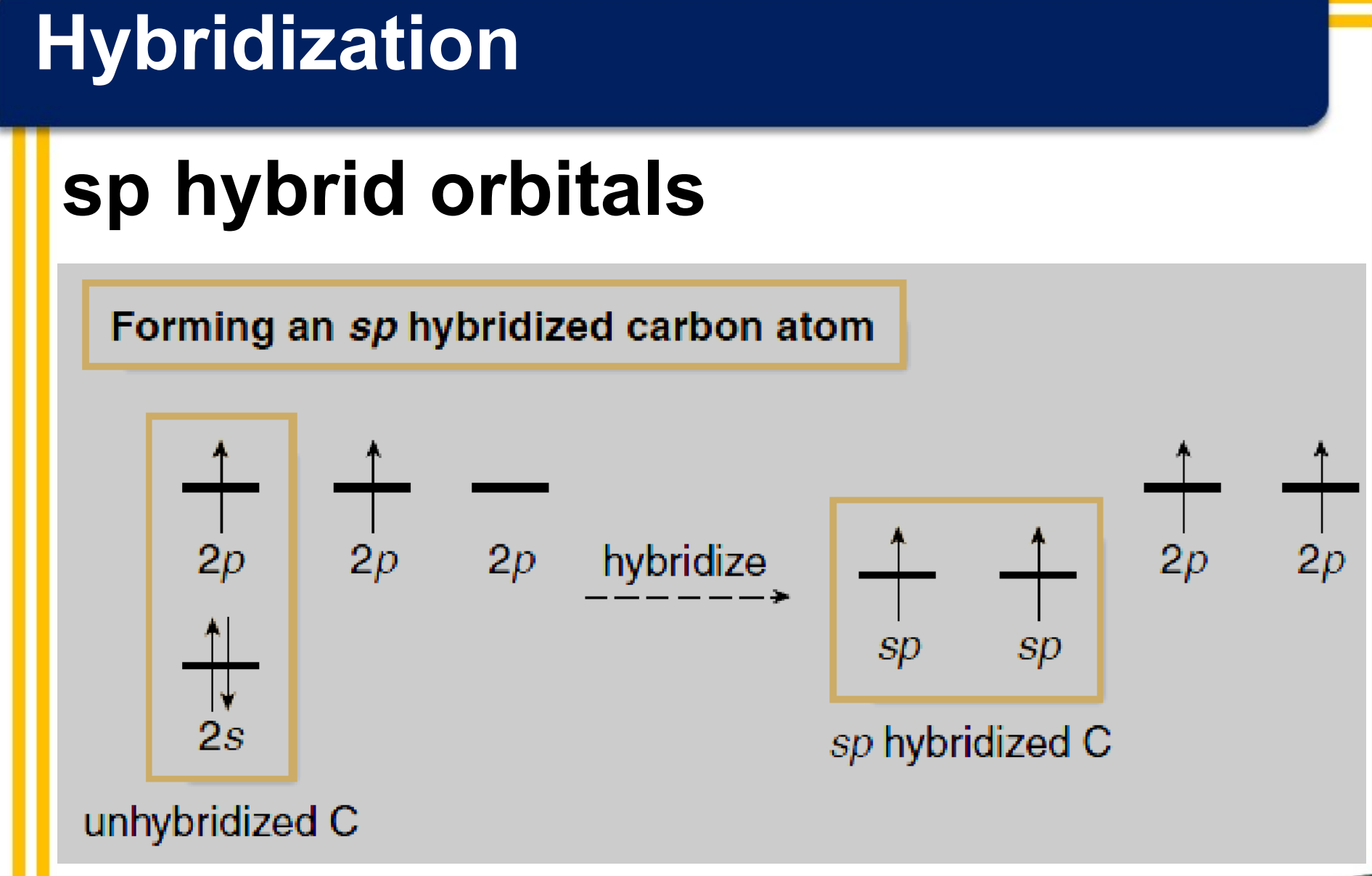

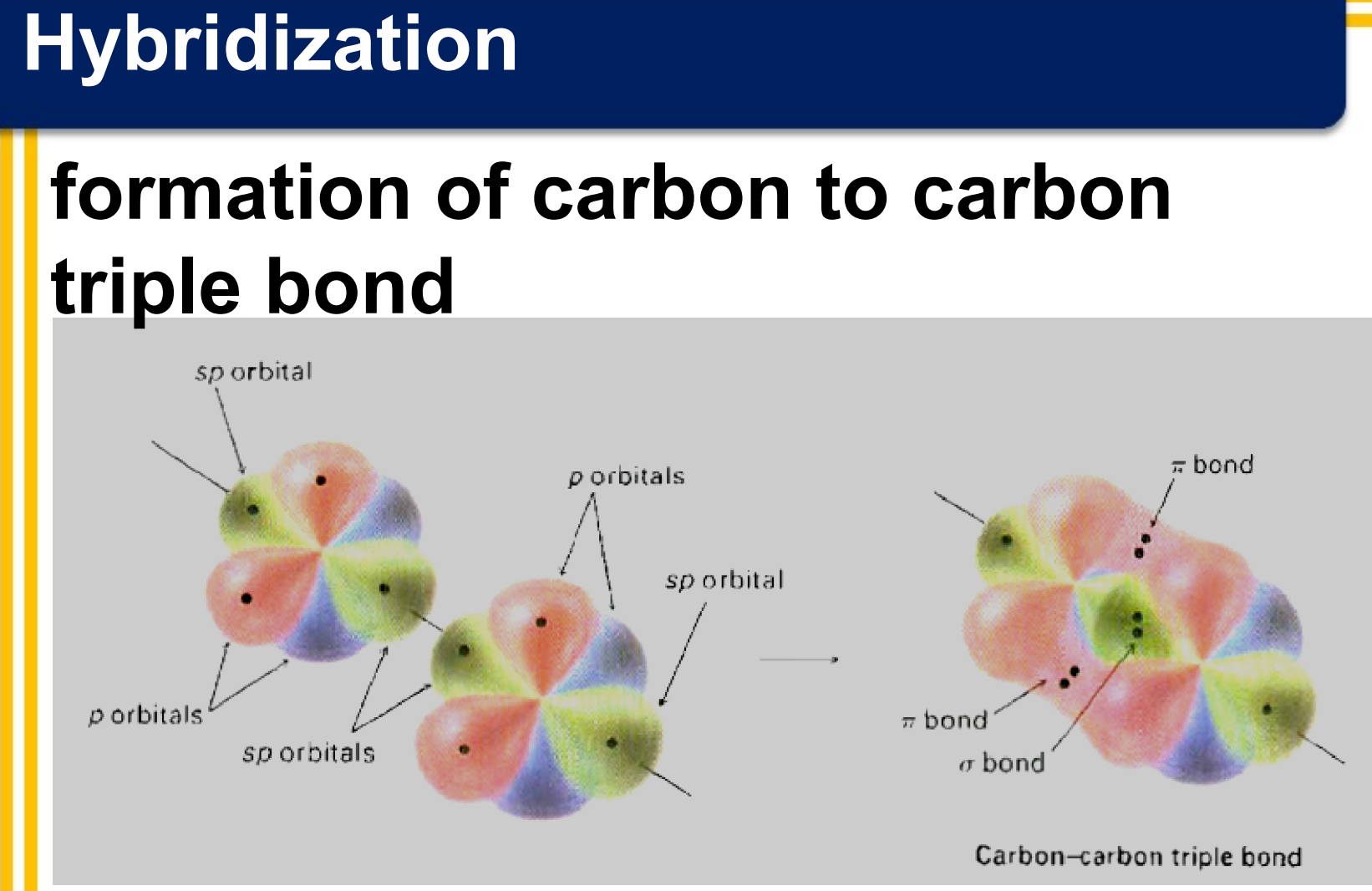
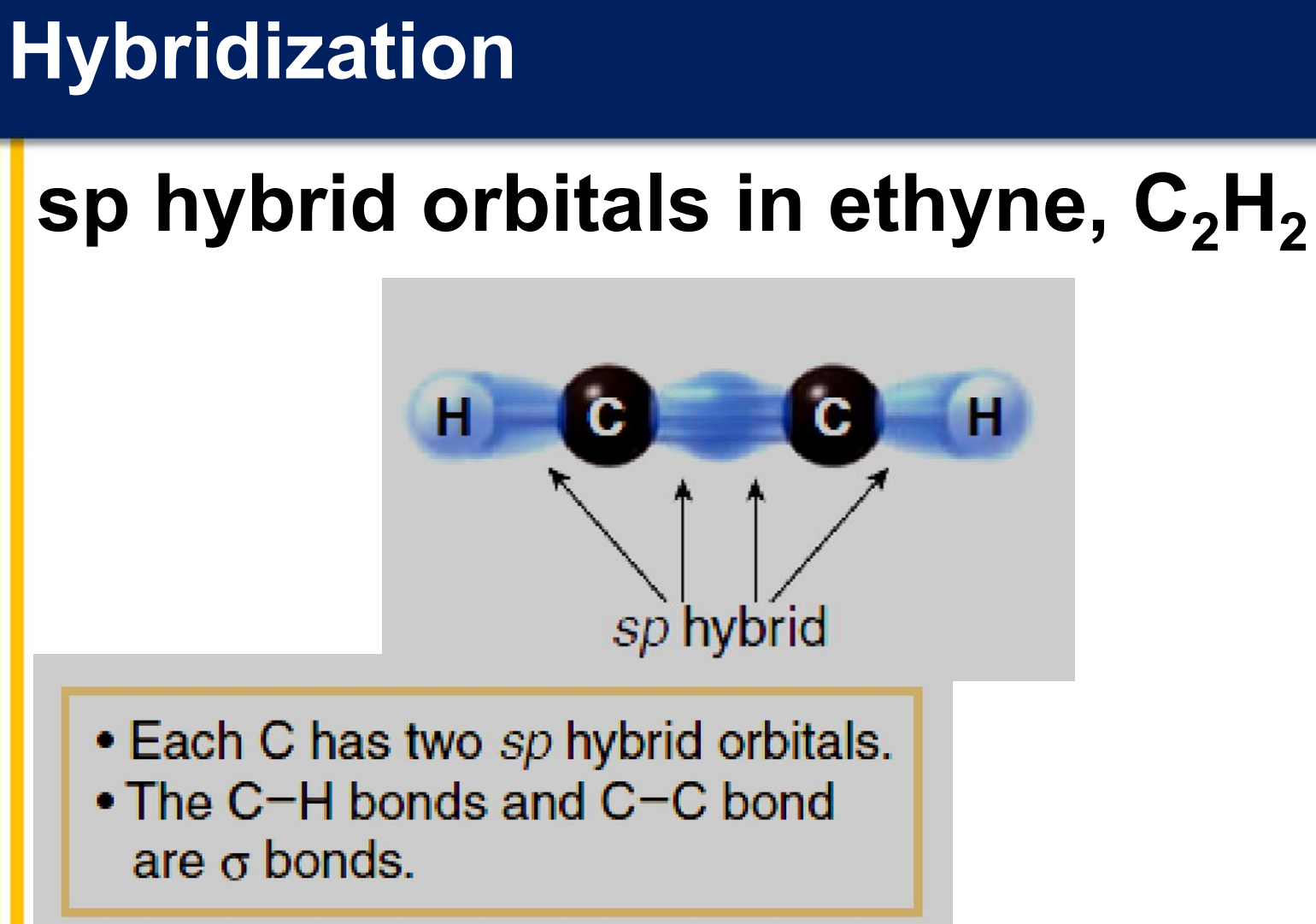

There are three covalent bonds formed between the two carbon atoms of ethyne:
one σ bond from the end-on overlap of two sp hybrid orbitals
two π bonds from the side-to-side overlap of two sets of 2p orbitals

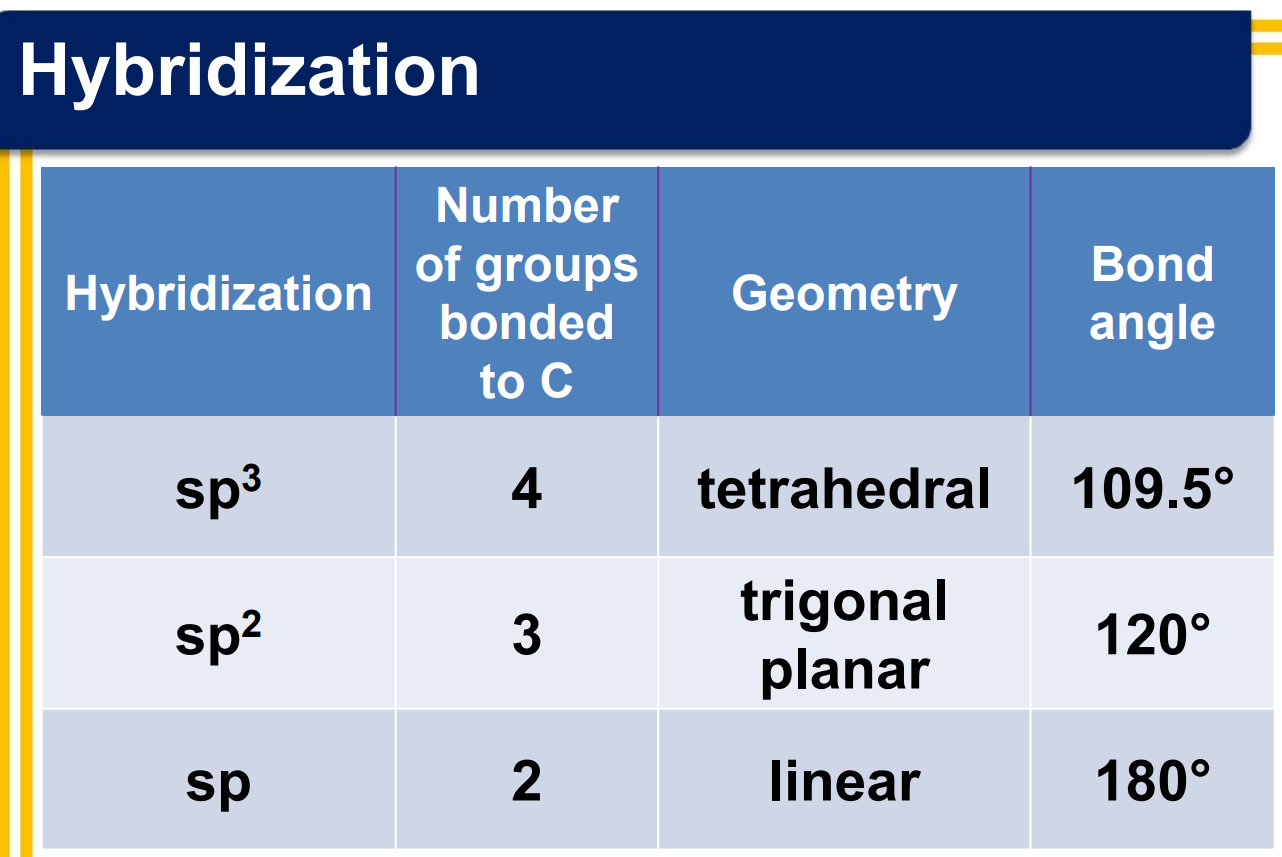
Hybridization Summary


Lewis
Kekulé
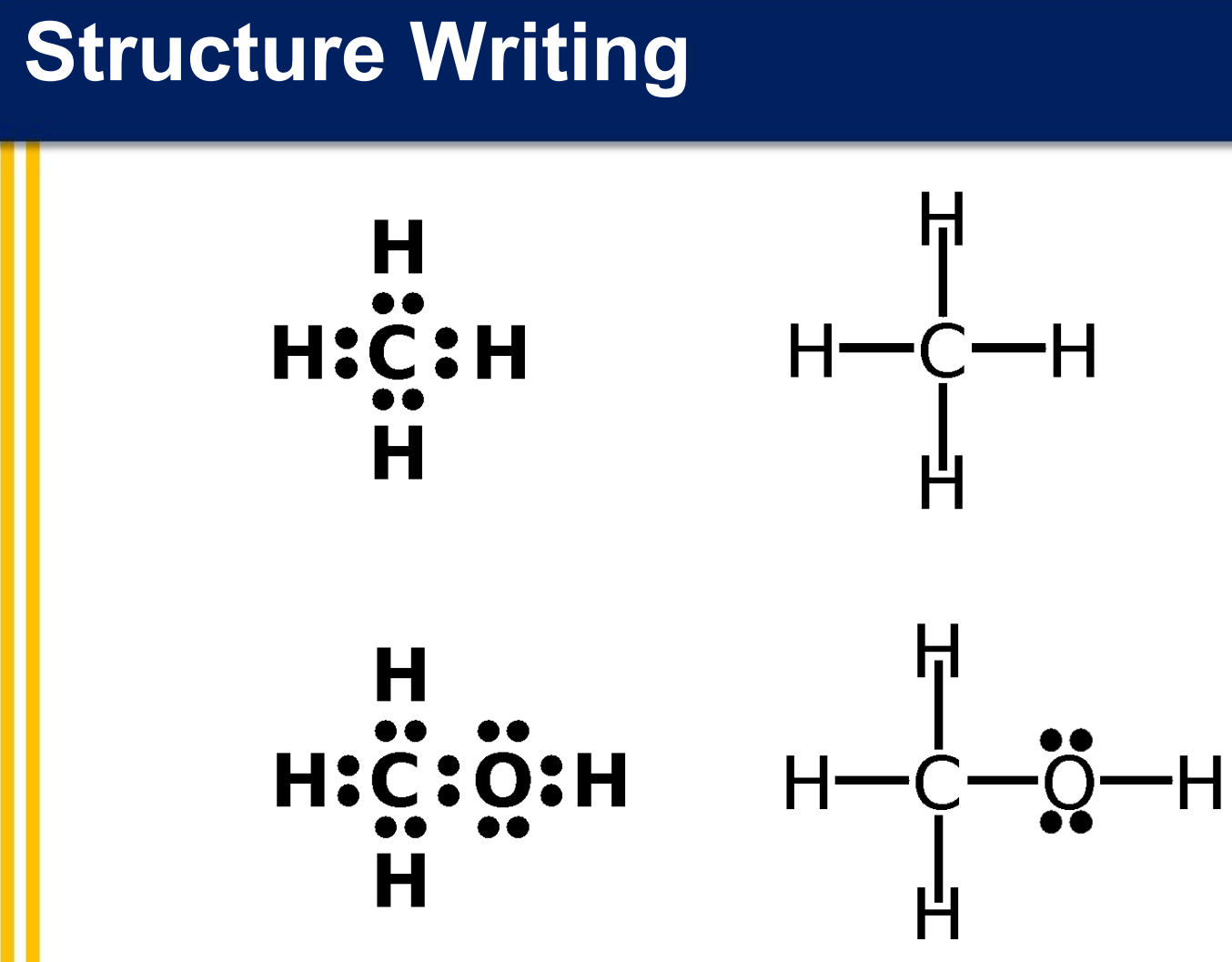
Structure Writing
__________ structures - valence electrons of an atom are represented as dots
__________ structures - a two-electron covalent bond is indicated as a line drawn between atoms
Formal Charge
________________-
the electronic charge assigned to individual atoms in a Lewis structure
the number of valence electrons in an isolated atom minus the number of electrons assigned to that atom in a Lewis structure
FC = VE – ½ BE - NBE
Resonance structures
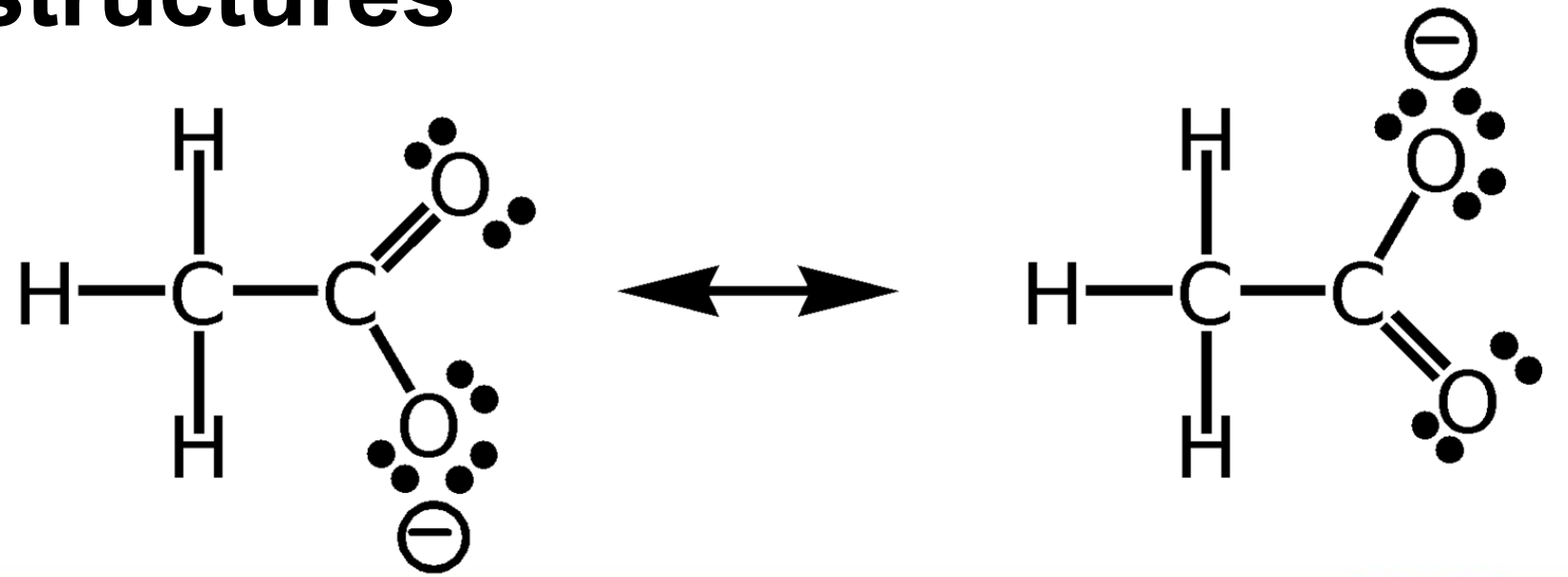
______________________
two or more structures of a molecule that differ in the placement of pi (π) bonds and nonbonded electrons
the placement of atoms and sigma (σ) bonds stay the same
the true structure is a composite or hybrid of the different contributing structures
Rules for Resonance Forms
Rules for Resonance Forms
Individual resonance forms are imaginary, not real.
Resonance forms differ only in the placement of their π or nonbonding electrons.
a curved arrow always indicates the movement of electrons, not the movement of atoms
Different resonance forms of a substance don’t have to be equivalent

Resonance forms must be valid Lewis structures and obey normal rules of valency.
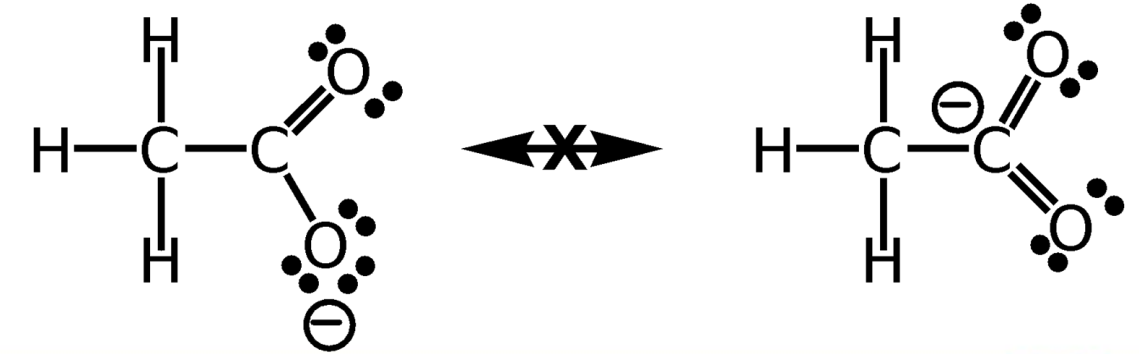
The resonance hybrid is more stable than any individual resonance form.
Molecular
Structural
Representation of Structures
_____________ formula – specifies the actual number of each kind of atom in the molecule
_____________ formula – shows the connectivity of the atoms in a molecule



Index of Hydrogen Deficiency

also called degree of unsaturation
gives a count of how many molecule of H2 needs to be added to a structure in order to obtain the corresponding saturated or acyclic form
ρ = C – ½ H + ½ N – ½ X + 1

What is the formula for the Index of Hydrogen Deficiency?
Index of Hydrogen Deficiency

takes a count of how many rings or multiple bonds are in the structure
must be an integer