biology examtwo
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
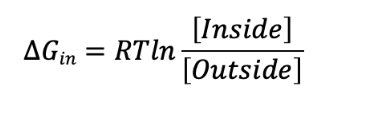
Use this formula for estimating the ΔG for moving a solute from outside the cell to inside the cell from ICA Bioenergetics and enzymes:
If an uncharged solute molecule has a higher concentration outside of the cell versus inside, the ΔG for solute movement is:
Negative, because it is energetically favorable for the solute to enter the cell.
A chemical reaction has a ΔG of +57 kJ/mol. The reaction ATP -> ADP + Pi under cellular conditions releases 57 kJ/mol. Could these reactions be successfully coupled in the cell?
No, because the overall ΔG would be zero and for the reaction to produce net products, the ΔG needs to be negative.
Some unfavorable reactions are coupled to a reaction that uses ATP. Which of the following accurately describes how ATP is used in a coupled reaction?
ATP is used to make an activated intermediate, and the activated intermediate is used to make the final product
Which of the following is often true of the active sites of enzymes?
They can bind to and position substrates in a way that encourages a chemical reaction
The uncoupled synthesis of glutamine is energetically unfavorable, but the coupled reaction ATP + glutamic acid + NH3 --> glutamine + ADP + Pi is favorable. Why does having ATP as a reactant make this reaction energetically favorable?
The reaction ATP -> ADP is very energetically favorable, so when ATP is a reactant, equilibrium favors formation of products
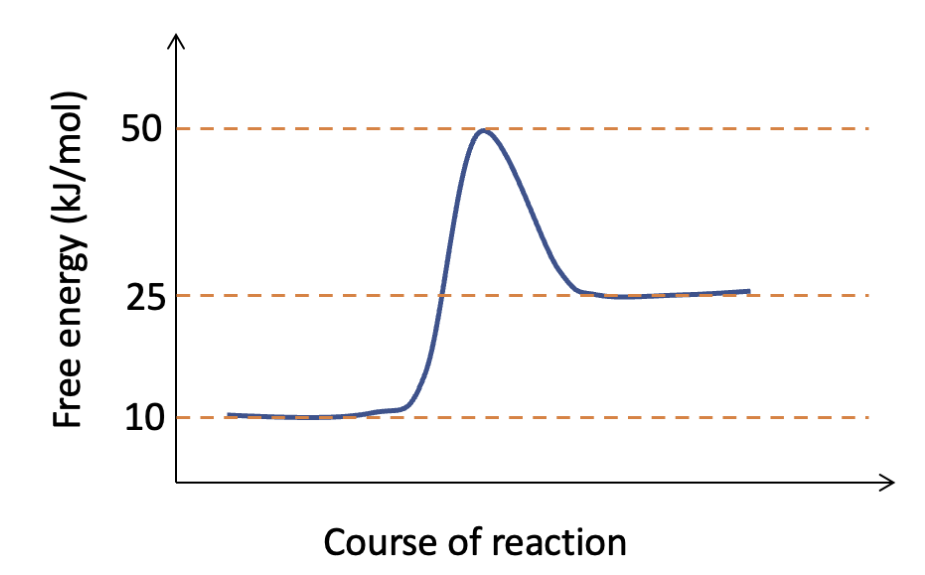
What is the activation energy for the process depicted in this graph, as it proceeds from left to right?
Approximately +40 kJ/mol.

The reaction shown has a ΔG of +14 kJ/mol under cellular conditions. Which of the following statements is true?
Glutamic acid and ammonia are produced at a higher rate than glutamine and water.
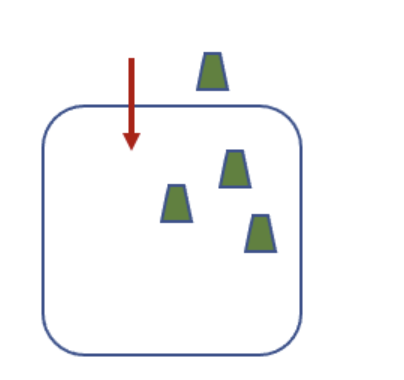
The image shows a cell that is moving a solute in the direction indicated by the arrow. This movement:
Requires coupling to a favorable process because the cell is moving the solute against the concentration gradient, which is unfavorable
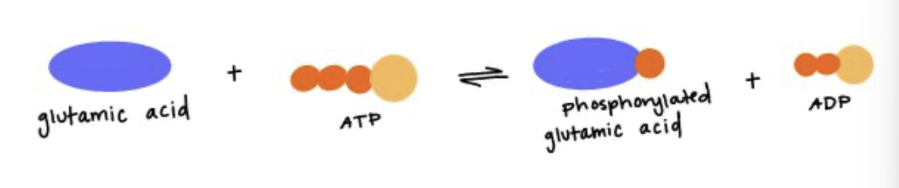
The enzyme that catalyzes the reaction shown should have what properties at its active site? Note - see the amino acid structures in the Quiz Information for the structure of glutamate/glutamic acid.
Positive charges, since both glutamic acid (glutamate) and ATP are negatively charged at the pH of the cell.
Mammalian cells typically have high intracellular concentrations of K+ as compared to the extracellular space. The ΔG of K+ movement into the cell due to the concentration gradient alone is:
Positive, because it is energetically unfavorable for K+ to enter the cell.
If a chemical reaction has a ΔG of -10 kJ/mol, which of the following is true?
This reaction releases energy to the surroundings.

The graph from ICA Bioenergetics and enzymes shows the effect of an enzyme on the activation energy for a reaction. How does this illustrate the effect on reaction rate due to the enzyme?
The enzyme decreases the energy barrier for the reaction to occur, so more molecules have sufficient energy to react and the reaction rate is increased.
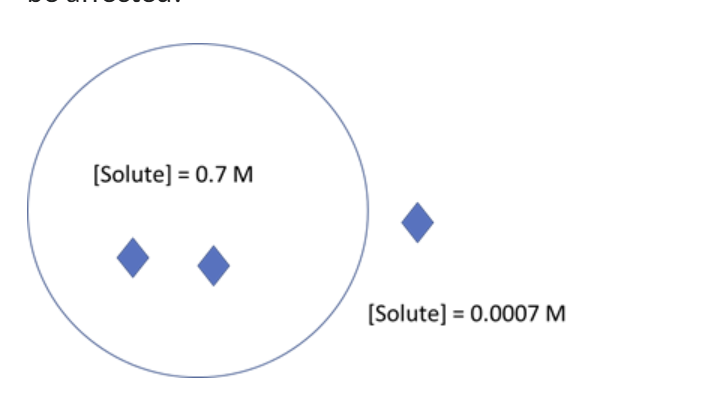
For the cell in the diagram below ΔGout = -17.1 kJ/mol. If the solute concentration inside the cell were to increase, how would the ΔGout be affected?
ΔGout would become more favorable
If the glycosylation function of the Golgi were disrupted, which of the following would be observed?
Certain proteins and lipids would not have the correct sugars attached.
Which of the following is NOT a property of membranes?
Effective barrier in the presence or absence of water
Which of the following could be considered a product of the lysosome?
Amino acids
Which of the following is a feature of eukaryotic but not prokaryotic (bacterial) cells?
Mitochondria
Which of the following proteins most likely carries out its function in the nucleus of the cell?
RNA polymerase
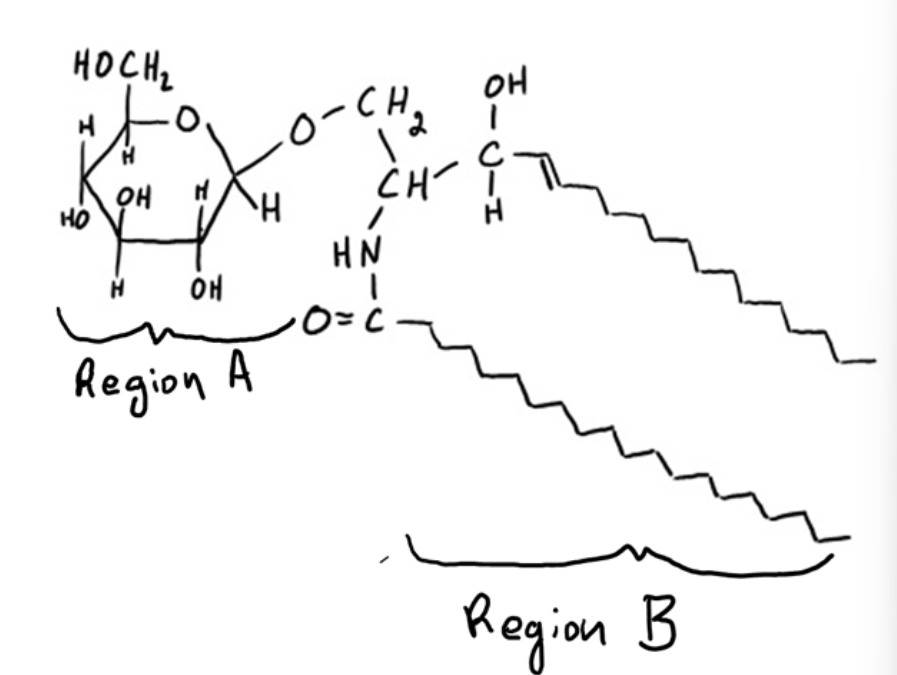
Based on its structure, is this lipid likely to be found in a cellular membrane?
Yes, because region A is primarily polar and region B is primarily nonpolar.
How and why does increasing temperature affect the fluidity of membranes?
An input of heat energy disrupts LDFs between fatty acid tails, increasing the fluidity.
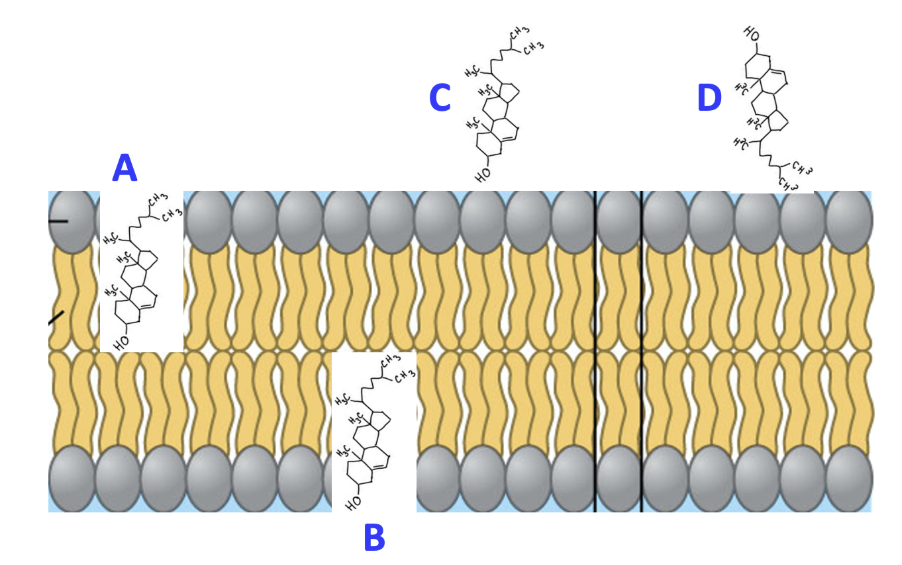
Which lettered molecule in the image shows how that molecule would be positioned in the cell membrane?
B
The lysosome has a proton pump to maintain the acidic pH of that compartment. Which of the following describes how the proton pump protein is delivered to the lysosome?
Translation completed at the rough ER -> Further modification in the Golgi -> Delivery to the Lysosome
Why is it important that acid hydrolases are separated from the cell by a membrane?
The membrane allows for differences in pH which allows the hydrolases to be active only inside the lysosome. This prevents the hydrolases from degrading the whole cell.
Some amino acid portions of proteins are embedded in a membrane, as shown by the red bracket in the image. Which of the following amino acids is most likely to be found in this sequence and interacting with the fatty acid tails of membrane lipids?
Membrane lipids are amphipathic. Why is this important for cellular function?
This allows the formation of a bilayer with hydrophilic parts on either side and hydrophobic parts clustered in the middle away from water.
If a bacterium adapted to a warm environment were moved to a much colder environment, what changes to the membrane lipids should the bacterium make?
Because decreased temperature decreases fluidity, the bacterium should decrease the saturation of fatty acid tails of membrane lipids
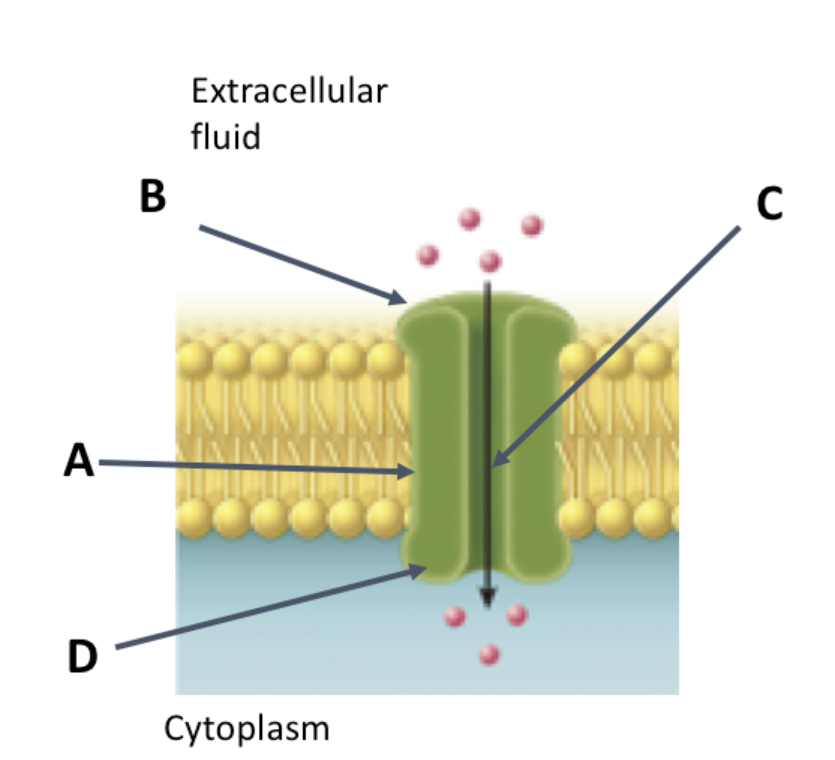
The image shows a protein embedded in a membrane. This membrane forms a pore that allows polar solutes to cross the membrane (indicated by the arrow going down the center of the protein). Which of the following statements would typically be true about the amino acids in the indicated locations and why?
Because the cytoplasm is hydrophilic, the amino acids at location D are likely hydrophilic.
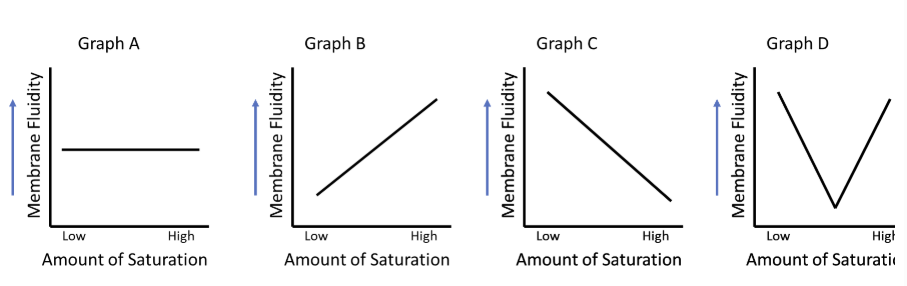
Which graph best represents the effect of fatty acid saturation on Membrane Fluidity?
graph C
What do facilitated diffusion and active transport have in common?
Both require a membrane protein to move solutes.

A cell with the indicated concentration of K+ and aquaporin proteins only (assume no other types of membrane proteins are present) is placed into fluid with the indicated concentration of K+. What will likely happen and why?
K+ cannot cross the membrane, but water enters the cell, with its concentration gradient, through the aquaporin.
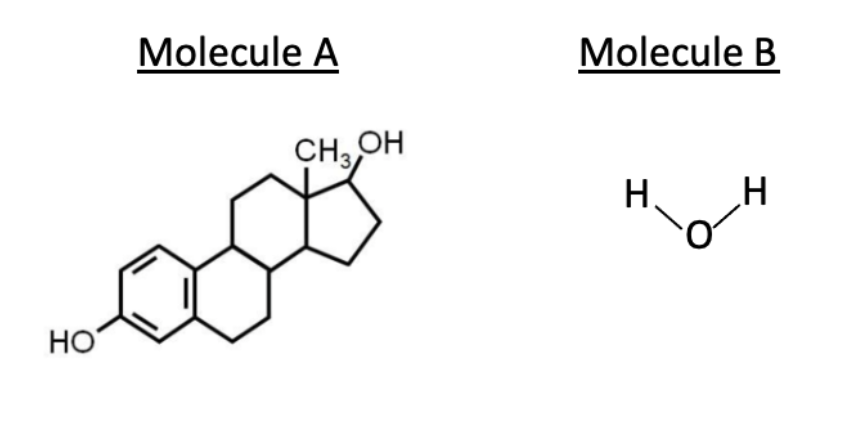
Based on their structures, which of the molecules below more likely requires a protein to efficiently cross a lipid bilayer and why?
Molecule B, since it could more favorably interact with polar molecules than membrane lipids.
Which of the following describes a transport process that would require energy input?
An amino acid being transported to an area with a higher concentration of amino acids.
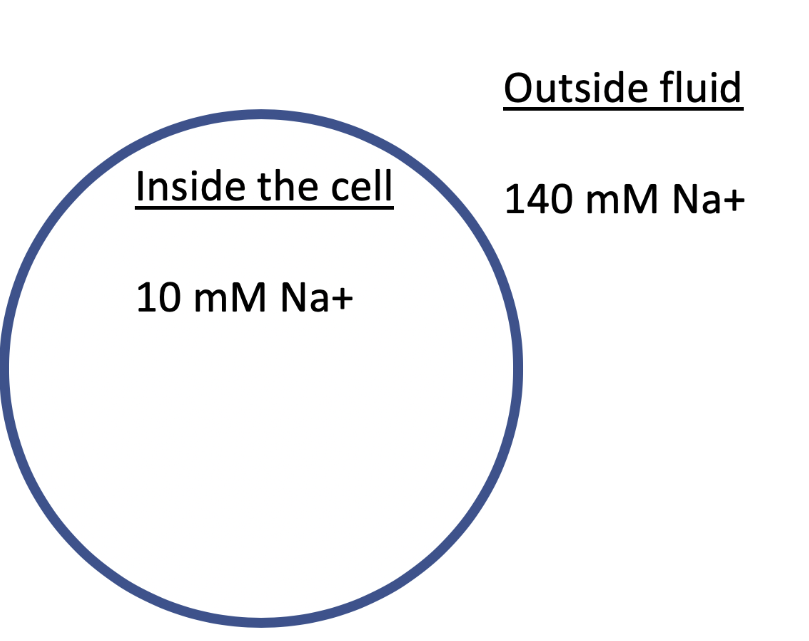
The image shows a cell with the indicated concentrations of Na+ inside and outside of the cell. What transport process likely occurs to bring Na+ into the cell? (assume the appropriate proteins are present if needed)
The reaction shown below occurs in cells and is catalyzed by Enzyme X:
A + B ⇄ C
The concentrations of A, B, and C at equilibrium are as follows: [A] = 2 mM, [B] = 4 mM, [C] = 4 mM.
If molecule C is added such that [C] = 6 mM, how will this affect the reaction?
There will be a net production of A and B, because the reaction will proceed towards achieving the equilibrium ratio of products and substrates.
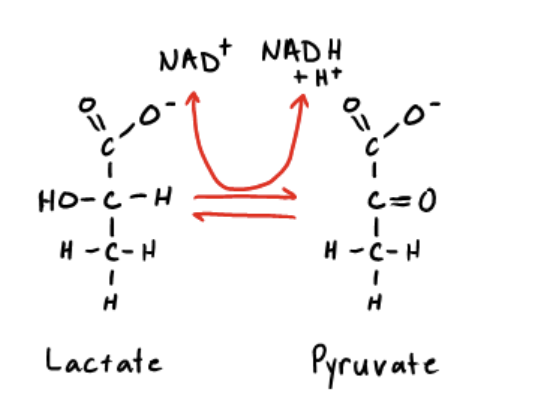
The reaction below is catalyzed by lactate dehydrogenase. When lactate is used to produce pyruvate, how is the energy released in this reaction captured?
It is directly coupled to NADH production.

Examine the following schematic of a metabolic pathway, in which A, B, C, and D represent molecules in these reactions. As the reaction B → C occurs, product C accumulates and makes the reaction B → C unfavorable. Based on the schematic, which of the following would keep B → C favorable?
The conversion of C to D, because this decreases the concentration of C
Which of the following is an example of an anabolic process?
Creating a polypeptide from amino acids

The reaction shown is part of a metabolic pathway. What happens in this reaction?
Carbon 2 is being oxidized, NAD+ is being reduced.
Consider the reaction M + NADH + H+ → O + NAD+. What type of pathway likely uses this reaction and why?
An anabolic pathway because conversion of M → O is coupled to NAD+ production from NADH.
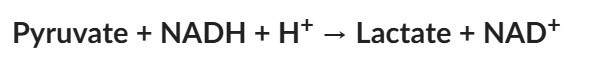
The following reaction occurs in muscle cells under specific circumstances: If the ΔG of the forward reaction (left to right) is negative under these circumstances, this means that:
Over time, net pyruvate is converted to lactate
Which statement accurately describes energetic coupling in catabolic pathways?
Energetically favorable reactions that break down molecules from food are coupled to the energetically unfavorable production of ATP from ADP + Pi.
Why is more ATP produced from the catabolism of 1 gram of fats compared to the amount of ATP produced from catabolism of 1 gram of sugars?
Because fats have more C-H bonds that allow more NADH to be produced
In pyruvate oxidation, a three-carbon pyruvate molecule is converted to a two-carbon acetyl group. What happened to the other carbon molecule?
It was released as CO2.
When no oxygen is present, which pair of processes allow conversion of NAD+ to NADH and then back to NAD+?
glycolysis and fermentation
Which accurately compares cellular respiration to glycolysis and fermentation?
Cellular respiration fully oxidizes the carbon from glucose while glycolysis and fermentation do not.
Which of the following correctly describes energetic coupling occurring during oxidative phosphorylation in cellular respiration?
The favorable movement of protons down their concentration gradient is coupled to the unfavorable phosphorylation of ADP.
The oxygen that enters cellular respiration is incorporated into:
H2O
When oxygen is present, which pair of processes allow conversion of NAD+ to NADH and then back to NAD+?
pyruvate oxidation and the electron transport chain
Which statement is correct about anabolism?
Coupling to favorable reactions is required
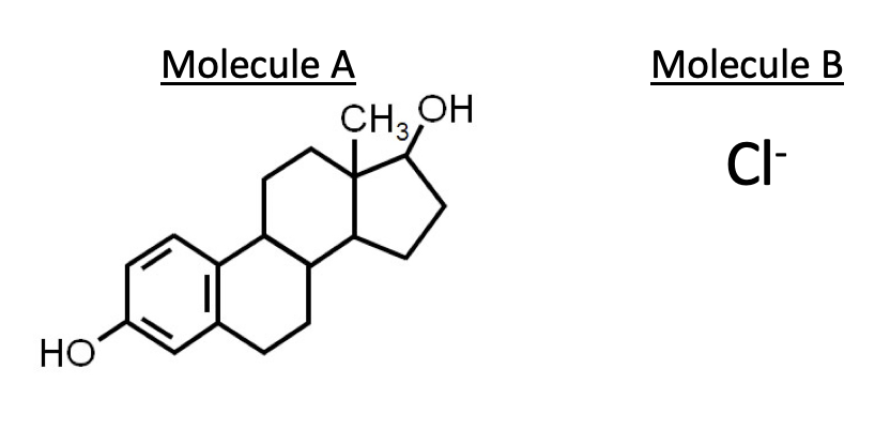
Based on their structures, which of the molecules below will more efficiently cross a lipid bilayer with no proteins and why?
Which of the following would allow an energetically unfavorable process to occur in the cell
If the unfavorable process were coupled to a process that releases sufficient energy
When the carbon in organic molecules is 'reduced' in metabolic reactions, the carbon typically gains:
bonds to hydrogen
In prokaryotic cells, transcription and translation take place:
In the cytosol
An enzyme catalyzes a reaction by:
Reducing the activation energy for the reaction
Which of the following processes is considered energetically unfavorable?
The synthesis of cellulose
Which of the following provides electrons to the electron transport chain?
NADH
The summary equation for cellular respiration is: C6H12O6 + O2 --> CO2 + H2O.
Select all stages of cellular respiration that have O2 as a direct input. (hint: see your model of matter and energy transformations from the PCA.)
electron transport
As electrons pass through Complex I - Complex IV of the mitochondrial electron transport chain,
Protons accumulate in the intermembrane space

Examine the following schematic of a metabolic pathway, in which A, B, C, and D represent molecules in these reactions. If the reaction B -> C is unfavorable under specific conditions in the cell, which of the following could make B -> C more favorable?
Increase the concentration of B.
In exergonic reactions:
The potential energy of the products is lower than the potential energy of the reactants and ΔG is negative.
Which of the following is facilitated diffusion?
A polar solute crosses a membrane through a transporter to an area of lower concentration
The organization of phospholipids in a lipid bilayer is driven by:
hydrophobic clustering
In the reaction to convert substrate S to product P, if the reaction is energetically favorable:
P has less energy than S, because some energy was released in the reaction
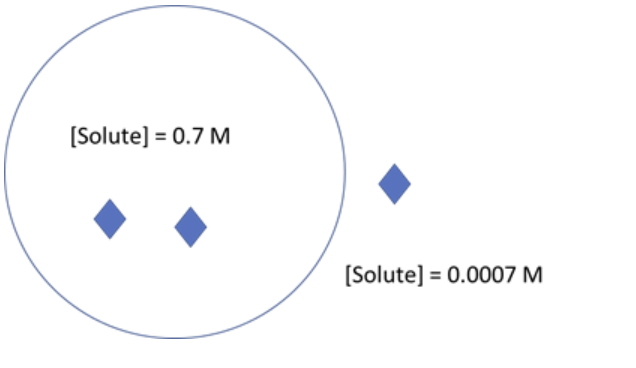
For the cell in the diagram below ΔGout = -17.1 kJ/mol. If the solute concentration inside the cell were to increase, how would the ΔGout be affected?
ΔGout would become more favorable
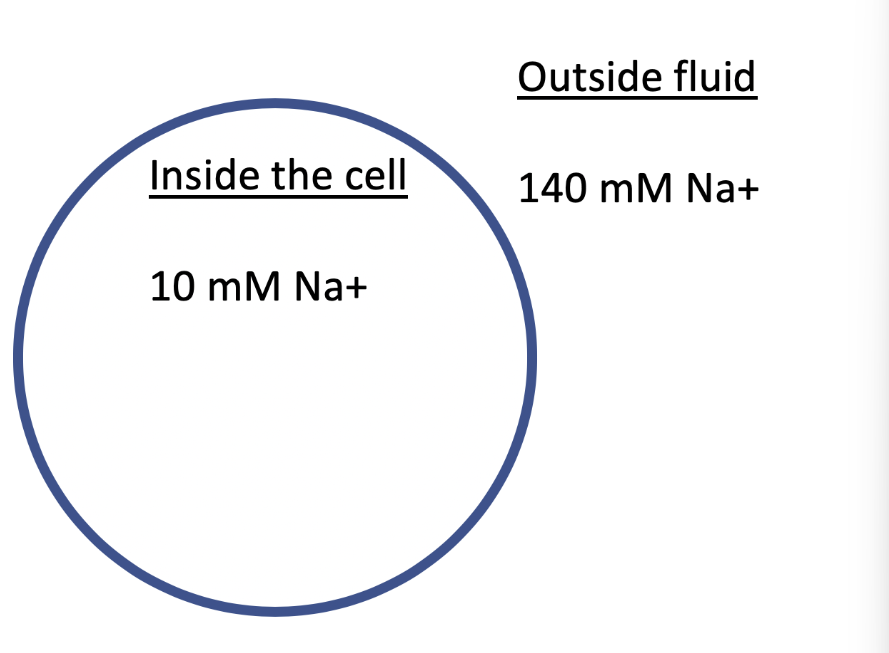
The image shows a cell with the indicated concentrations of Na+ inside and outside of the cell. What transport process likely occurs to bring Na+ into the cell? (assume the appropriate proteins are present if needed)
Facilitated diffusion, because molecules are moving with their concentration gradient and require a protein to efficiently cross the membrane.

Some amino acid portions of proteins are embedded in a membrane, as shown by the red bracket in the image. Which of the following amino acids is most likely to be found in this sequence and interacting with the fatty acid tails of membrane lipids?
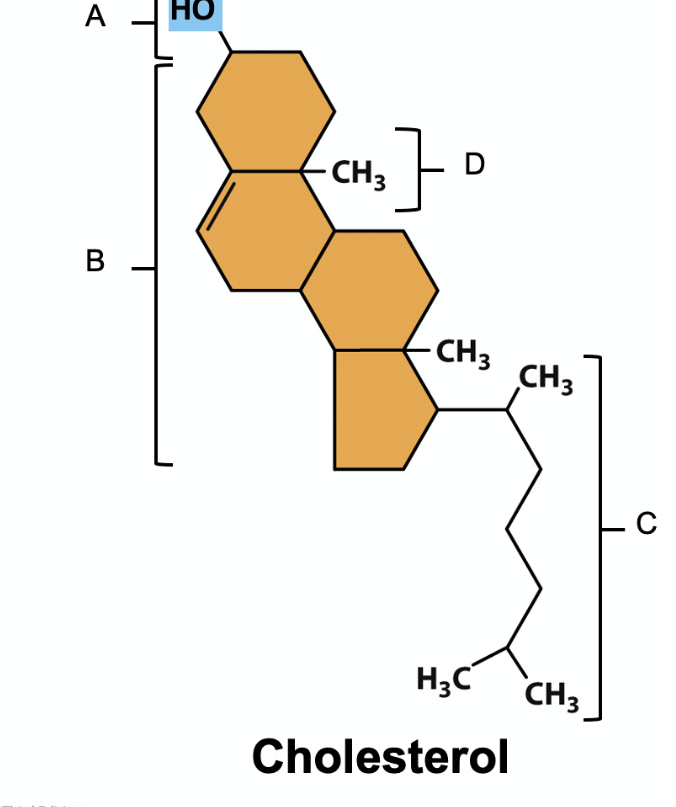
In the following figure of cholesterol, which is the hydrophilic component of the molecule?
A