Biology
1/182
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
183 Terms
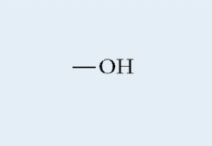
Hydroxyl
Found in all macromolecules
Hydroxide
Polar
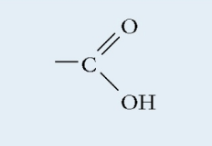
Carboxyl
Polar
Carbon double bonded to an Oxygen and single bonded to a Hydroxide
Acidic
Found in: proteins, lipids
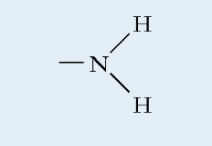
Amine (functional group)
Polar
Basic
Found in: proteins, nucleic acids
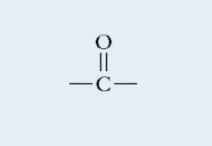
Carbonyl
Polar
Carbon double bonded to an Oxygen
Found in: carbohydrates, nucleic acid
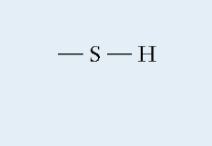
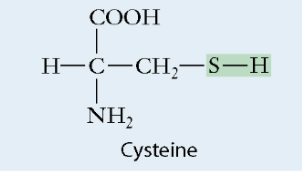
Sulfhydryl
Slightly polar
Sulfur single bonded to a Hydrogen
Found in: proteins
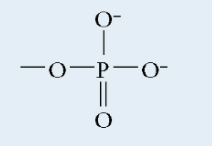
Phosphate
Polar
Negative charge
Found in: nucleic acid, phospholipid
Photosynthesis
6CO2 + 6H2O —> C6H12O6 + 6O2
Cellular respiration
C6H12O6 + 6O2 —> 6CO2 + 6H2O + 38ATP
electrochemical gradients are used to generate ATP
C6H12O6: source of energy
6 CO2: krebs cycle
6 H2O: electron transport chain
38 ATP: oxidative phosphorylation
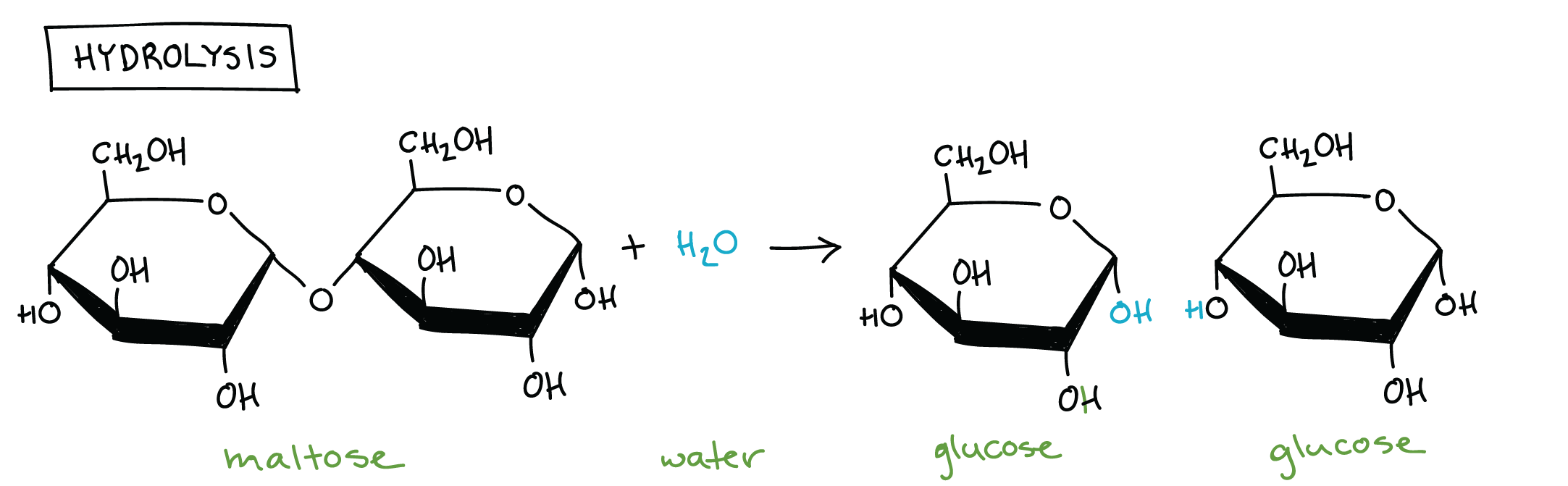
Hydrolysis (Carbohydrate)
Gaining water to form molecule
Enzymes are involved as a catalyst
Requires at least 1 of the following: hydroxyl, carboxyl
Protein
Most diverse macromolecule
Made by DNA
Very diverse
The function is exclusive to it’s shape
Examples:
Enzymes
Antigens / Antibodies
Cell repairing
Keratin (hair / nails)
Found in:
Muscles
Blood
Bones
Cells
Endocrine gland
Making a protein
Involves DNA, RNA and ribosomes
Chains of amino acid that are folded into shape
Monomers
Single pieces
Main types: sugars, amino acids, fatty acids, nucleotides
Primary protein structure
Amino acid determines what the protein is / does
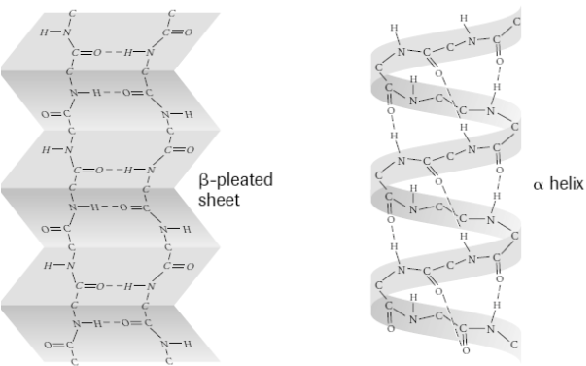
Secondary protein structure
Folding dipeptide or polypeptide into pleated sheet or twisting into helix
H bonds are responsible for the disfiguration of proteins
Tertiary protein structure
R groups interact
Di / Poly peptides become proteins
May contain Disulfide bridges if the R group has a sulfhydryl functional group
Quaternary protein structure
Clustering to form super proteins
Only occurs to bigger proteins
Polypeptide
Composed of more than 2 amino acids bonded together
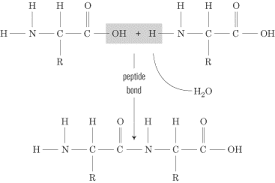
Peptide bonds
Denaturing
Reformation (permanent or temporary) of proteins
Requires heat or chemicals (acids)
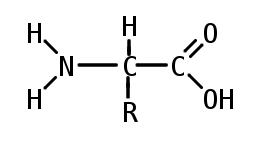
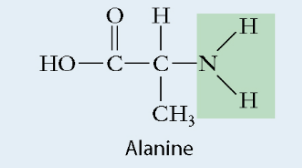
Amino acid molecule
Composed of a centre carbon, an Amine group on one side, Carboxyl group on the opposite side, single bonded hydrogen and amino acid opposite of the hydrogen
Protein monomer
Polarity depends on the R group
Carbohydrates
Sugars
CH2O (1:2:1)
Source of energy (energy fuel for the body)
Structural support (Cellulose)
Found in:
Liver
Muscles
Blood (glucose)
Brain
Monosaccharides
Monomer of sugars
Distinguished by:
Carbonyl group
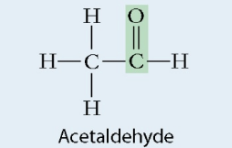
Aldehyde
Ketone
Number of carbons
Goes from chain form to ring formation
Aldehyde
Group of monosaccharide
Carbonyl group
Includes: glucose, galactose
Ketone
Group of monosaccharide
Carbonyl group
Includes: fructose
Polysaccharides
Complex carbohydrates
Composed of a large amount of monosaccharides
Storing energy
Structural support
Examples:
Cellulose
Amylopectin
Amylose (starch)
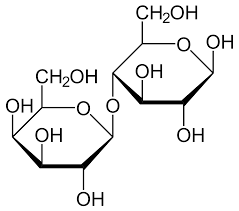
Lactose
β glucose + β galactose
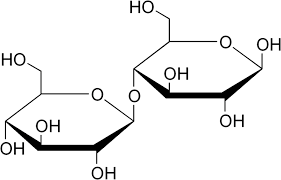
Maltose
α glucose + α glucose
Found in maltesers, wheat, grains
Used for energy
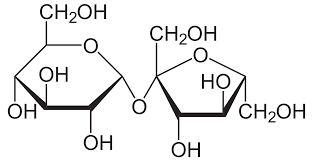
Sucrose
α glucose + fructose
Found in honey, fruits
Table sugar (cannot be tested for)
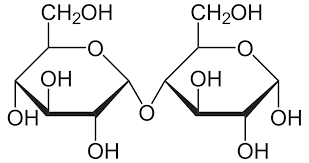
Cellobiose
β glucose + β glucose
Found in plant cell walls
Enzyme
Catalyst
Protein
Helps break down macromolecules
Active site
Lipids
Energy storing
Building membranes / organelles
Mostly non-polar
Contains Carbon, Hydrogen and Oxygen
Consists of: fats, phospholipids, sterol and waxes
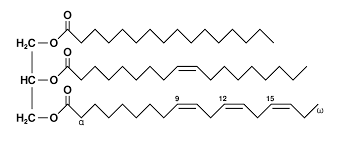
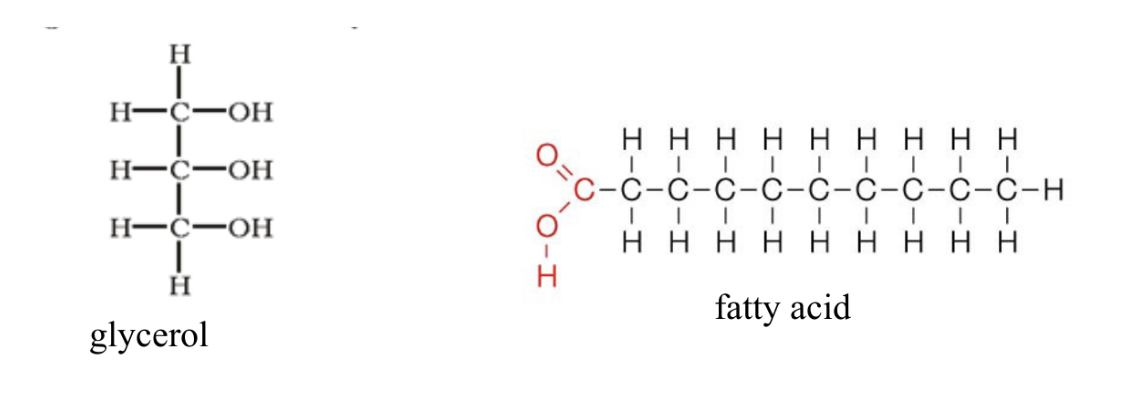
Triglyceride
Type of fat
Aka triacylglycerol
Consists of a glycerol bonded to 3 fatty acids
Fat
Most common lipid
Stores more than 2x the energy that proteins & carbohydrates can store
Consists of triglycerides
Saturated & Unsaturated
Dipeptide
Composed of 2 amino acids bonded together
Peptide bonds
Peptide bond
Bond between amino acids
Requires dehydration reaction
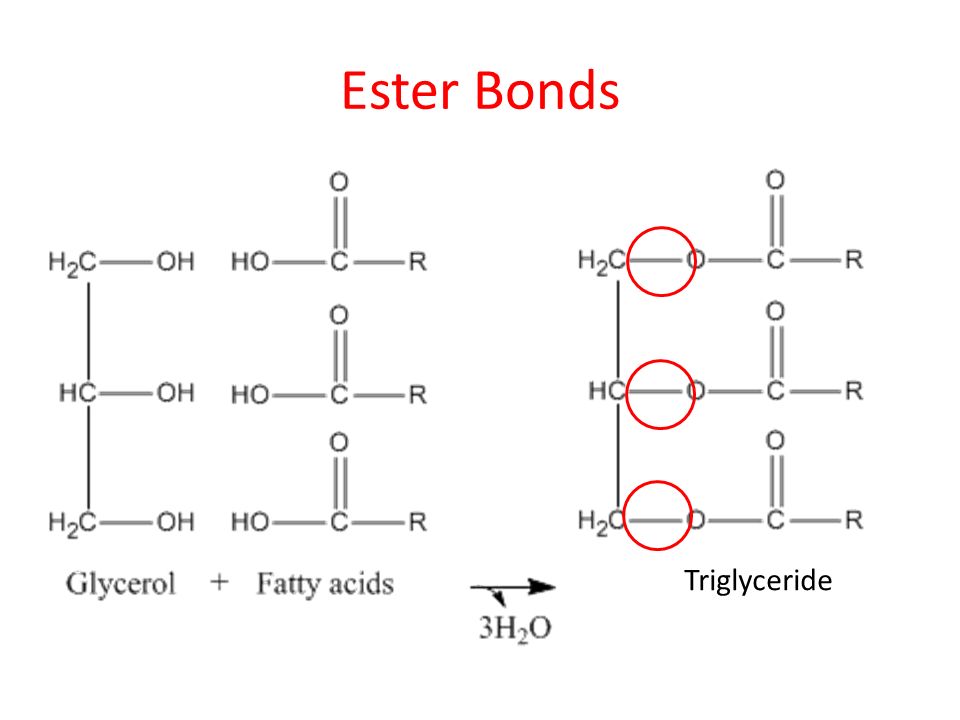
Ester bond
Found in triglycerides
Bond between glycerol and fatty acid
Requires a Dehydration reaction in order to form
Triglyceride notation
C[number of carbons] : [number of double bonds], ω-[carbon w the double bond]
![<p>C[number of carbons] : [number of double bonds], ω-[carbon w the double bond]</p>](https://knowt-user-attachments.s3.amazonaws.com/0d336baa-b474-4640-ab00-5ab0db8034b7.jpeg)
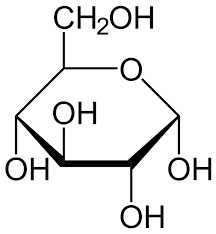
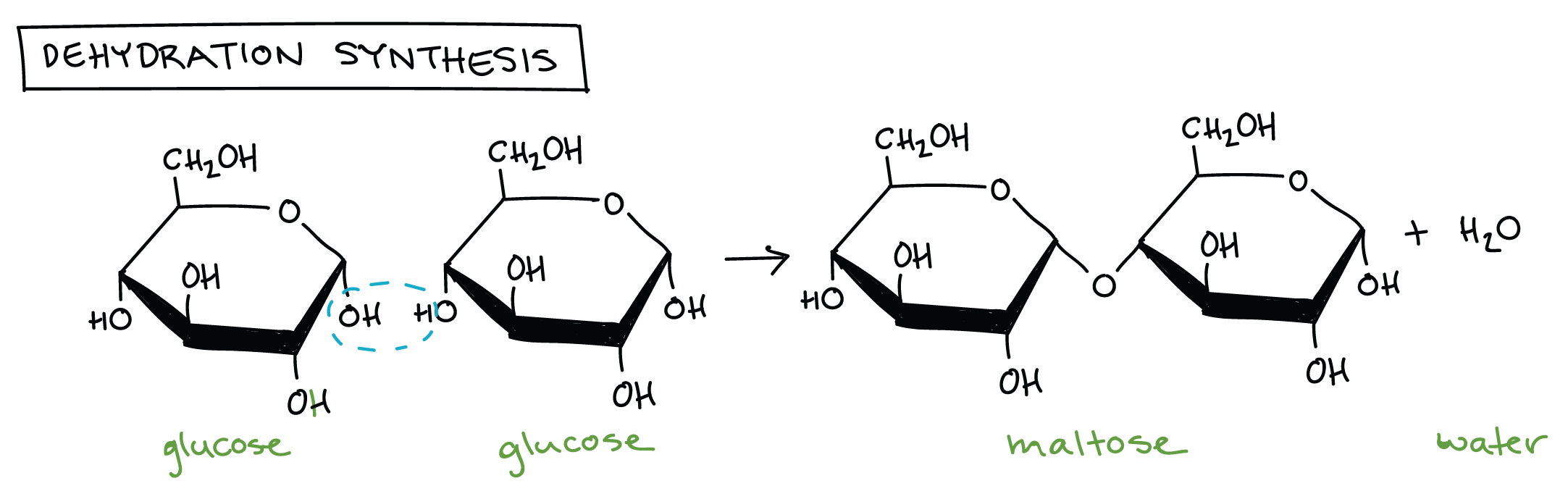
α Glucose
DDUD
Starch
Found in potatoes, wheat
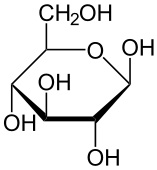
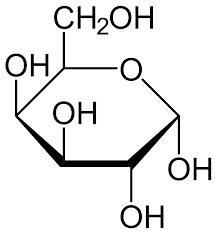
β Glucose
UDUD
Cellulose
Main component of Cellobiose
Found in fruits, plants
Dehydration reaction (Carbohydrates)
C 1-6 linkage (for maltose)
Enzymes are involved as a catalyst
Water is formed
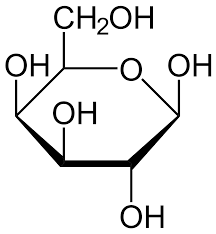
α Galactose
DDUU
β Galactose
UDUU
Component for lactose
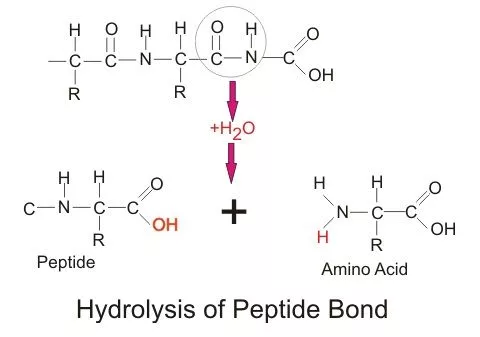
Hydrolysis (Protein)
Addition of water to amino acid
Enzymes are involved as a catalyst
The product is 2 amino acids
Dehydration (Protein)
Losing water
Enzymes are involved as a catalyst
Carbon from carboxyl bonds to Nitrogen from Amine group
Carbon gains a double bond to Oxygen (Carbonyl)
Nitrogen gains a single bond to Hydrogen
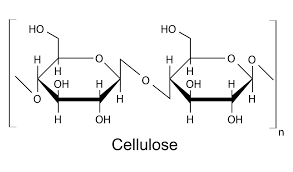
Cellulose
Large amounts of β Glucose
H bonds aid in the sturdy structure
AKA fibre
Only found in plants
Composed of Cellobioses
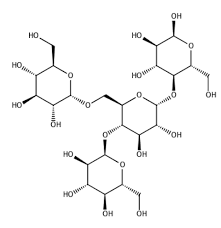
Glycogen
Hydrolization of Amylose and Amylopectin
Short term energy storage
Turned into Fat after 4 hours
Found in muscles and liver
Only found in animals
Amylose
Polysaccharide
Many α Glucoses in a chain
Starch
Amylopectin
Polysaccharide
Less soluble than Amylose
C1-6 glycosidic linkage
Benedict’s test
Test to identify monosaccharides
Solution turns red if positive
Lugol’s test
Test to identify polysaccharides
Solution turns purple-black if positive
Biuret test
Test to identify proteins
Solution turns purple if positive
Sudan red test
Test to identify lipids
Solution turns red if positive
Fatty acid
Monomer for fats
Usually bonded to an alcohol (glycerol)
Glycerol
Alcohol
Bonds with fatty acids
Composed of 3 hydroxyl groups
Oligosaccharides
2-3 monosaccharides
2 monosaccharides can be disaccharides
Attached by Glycosidic linkages
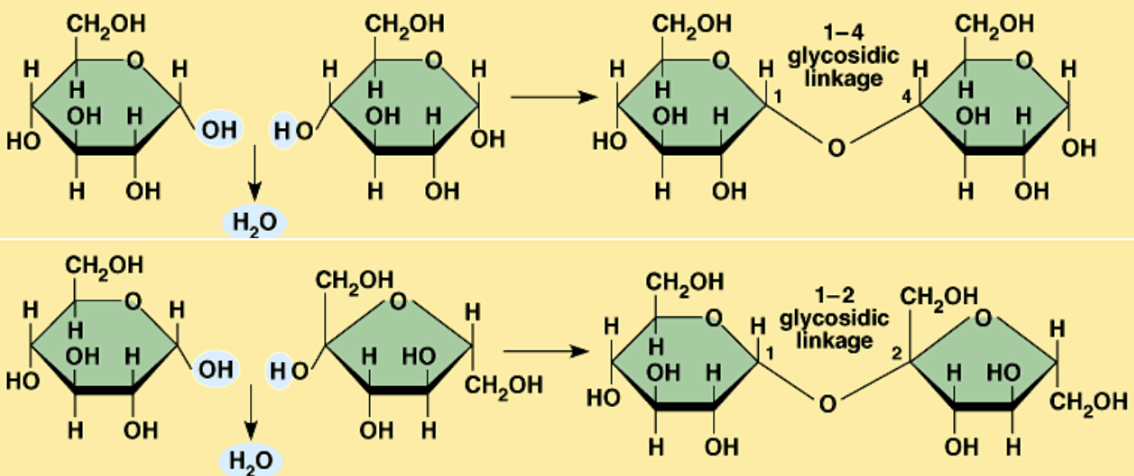
Unsaturated Fat
Presence of double bonds
Trans bonds
Cis bonds
Chitin
Similar to a polysaccharide
Is not a polysaccharide due to the presence of a Nitrogen
Violates carbohydrate convention - composed of only C,H,O
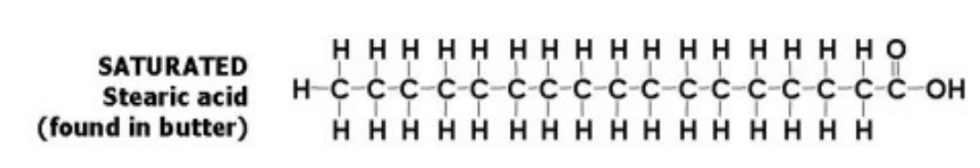
Saturated fat
AKA: Stearic acid
No double bonds
Found in: Butter
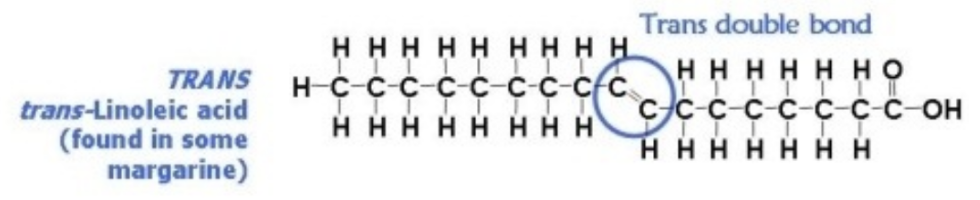
Trans bonds
Bad
AKA: Trans-linoleic acid
Hydrogens are on opposite sides
Found in: margarine
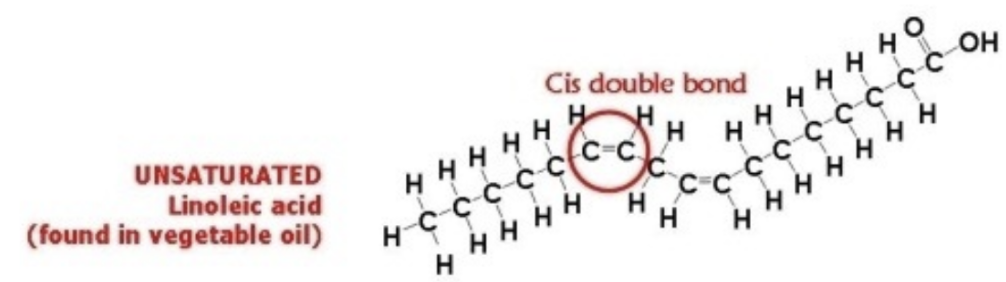
Cis bonds
Better than Trans bonds
AKA: Linoleic acid
Hydrogens are on the same side
Found in: oil
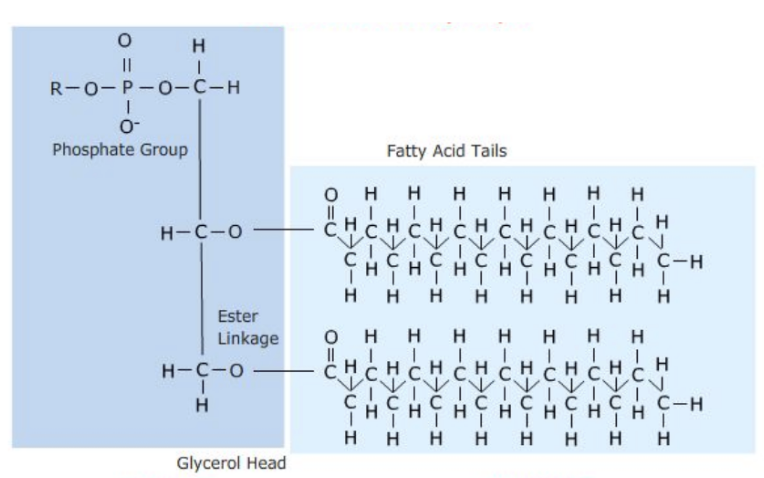
Phospholipid
Similar to fats
2 fatty acid chains
Final glycerol is bonded to a phosphate group
Hydrophilic & hydrophobic ends
Fatty acid chains are hydrophobic (Non-polar)
Phosphate group is hydrophilic (Polar / charged)
Contains bilayering
Sterols
AKA: steroids
Hydrophobic molecules
Made up of hydrocarbon rings
Used for:
Hormones
Structural purposes
Examples:
Cholesterol
Testosterone
Waxes
Long carbon chained molecules
Solid at room temperature
Used to prevent loss of water & solutes
DNA
Composed of nucleotide monomers
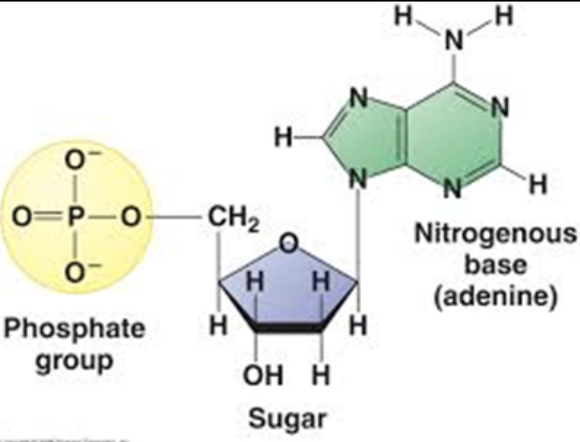
Nucleotide
DNA monomer
Made of:
Sugar
Phosphate
Nitrogenous Base
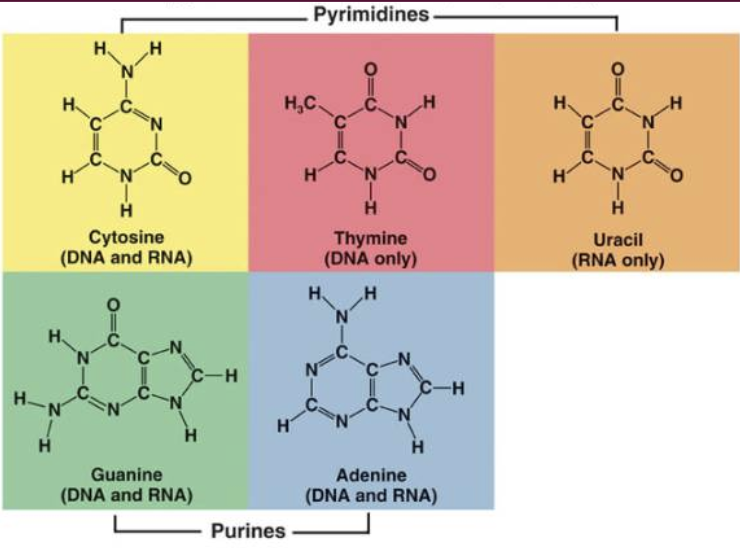
Nitrogenous Base
Pyrimidines
Cytosine
Present in DNA & RNA
Thymine
Present in DNA
Uracil
Present in RNA
Purine
Adenine
Present in DNA & RNA
Guanine
Present in DNA & RNA
Eukaryotic Cell
Contains genetic material
Has a cell membrane
Has Cytoplasm
Cytoplasm
Contains organelles / cytosol
Mostly composed of water
Filled with microtubules
Glycolysis location
Nucleus
Contains DNA
DNA combines with protein to make chromosomes
chromatin
Produces ribosomes
Chromosomes are only visible in dividing cells
Has many parts
Nucleoplasm
Nucleolus
Nuclear envelope
A double membrane
Nuclear pore complexes
Holes in the nuclear envelope

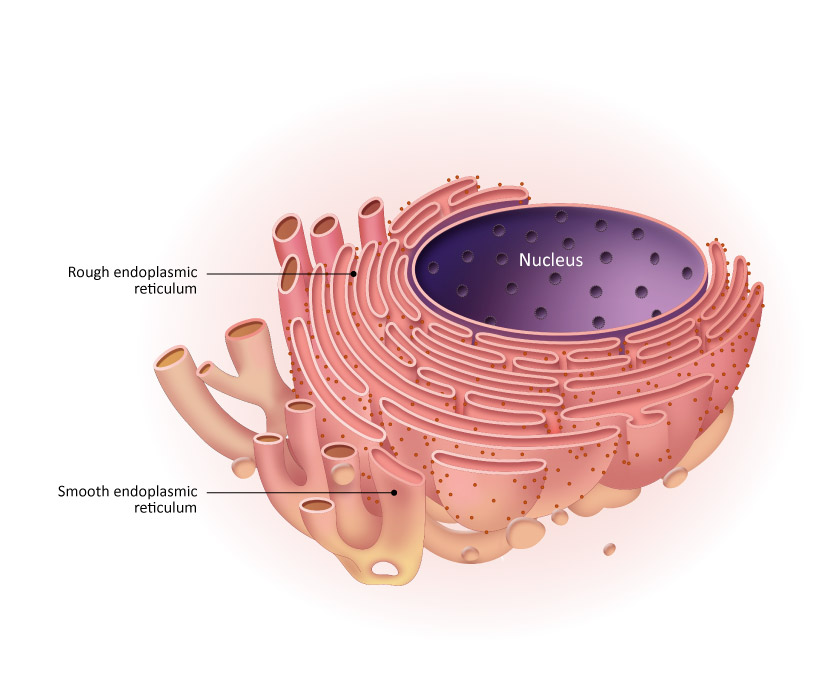
Endoplasmic reticulum
Membrane-bound tubules and sacs
Makes vesicles
Rough endoplasmic reticulum
Makes proteins
Located near nucleus
Is rough because of ribosomes attachments
ribosomes come from the nucleus
Smooth endoplasmic reticulum
makes lipids (but not triglycerides
is far from the nucleus
Cell wall
Composed of cellulose and glycoprotein
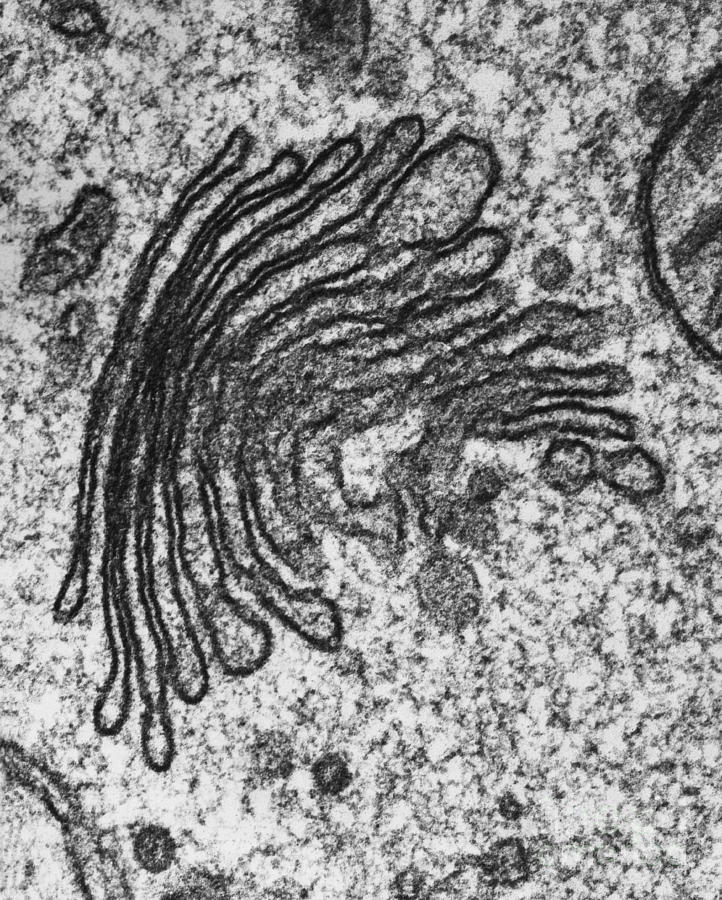
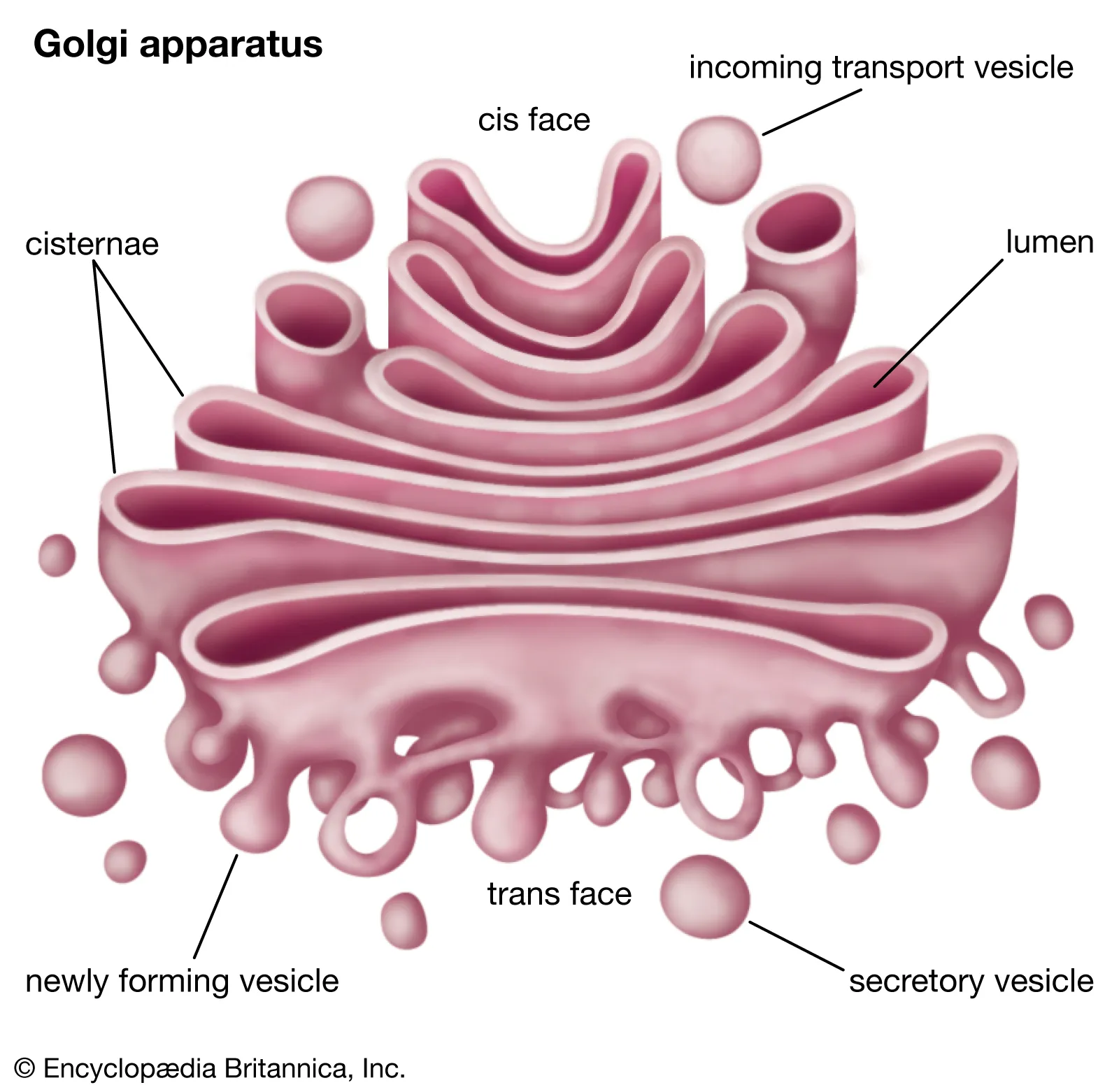
Golgi apparatus
transportation / storage of materials
Creates pectin and lysosomes
Cis face
acts as an entrance for vesicles on the inside of cell
faces the inside of the cell
endocytosis
Trans face
acts as an exit for vesicles after resources are modified
side that faces the outside of the cell (membrane)
exocytosis
Vacuole
contains enzymes for redox reactions
breaks down toxic chemicals
membrane bound sac
regulates the amount of water in the cell
stores water & nutrients
helps with endocytosis and exocytosis
PLANT:
basically all purpose function
ANIMAL:
small vacuoles (not really)
they all have a specific function
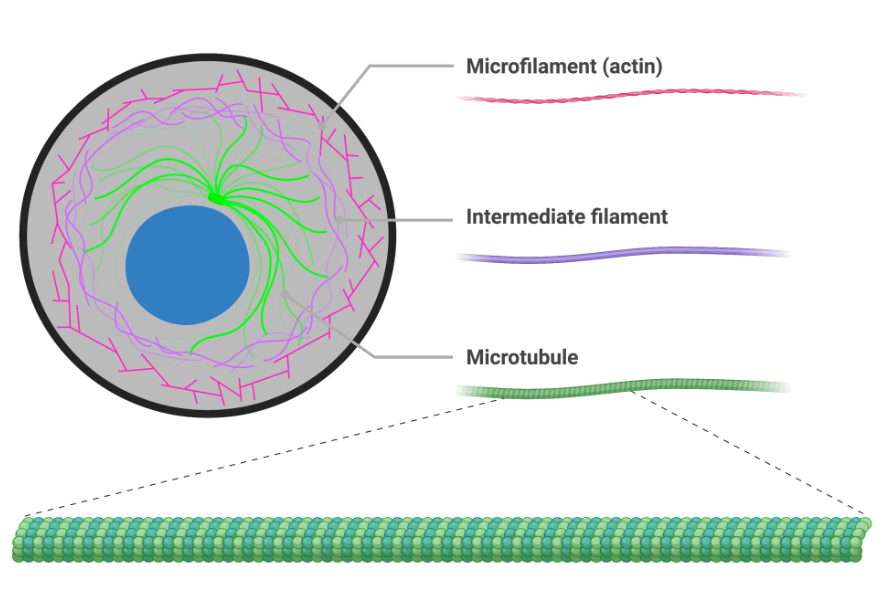
Cytoskeleton
Is a bunch of protein fibres
serves as a path for vesicles and organelles to move on
forms methods of movement
cilia
flagella
has 3 parts that can be used and rebuilt
creates appendages
micotubules
ex centrioles
acts as tracks
microfilaments
actin filaments
exists in both animal and plant cells
Endosymbiosis
Some eukaryotic organelles were evolved from prokaryotes being engulfed by another prokaryote
involves:
mitochondria
chloroplast
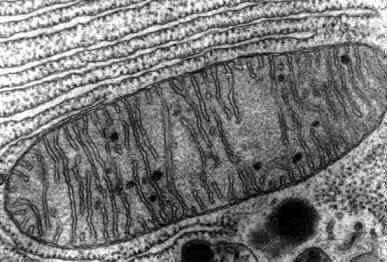
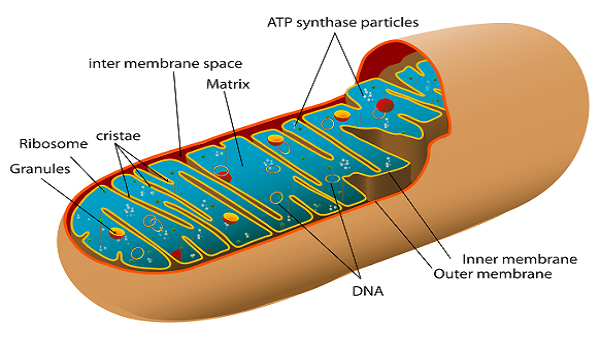
Mitochondria
2 membranes
Smooth on the outer membrane
Cristae on the inner membrane
Filled with matrix
converts nutrients into energy for the cell
aka ATP
exists in both plants and animal cells
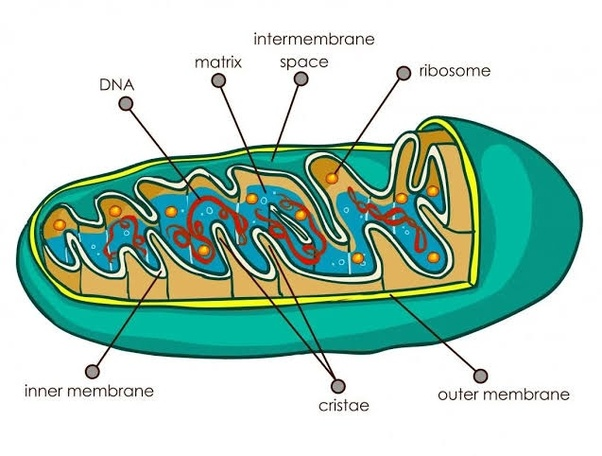
Cristae
Folds on the inner mitochondria membrane
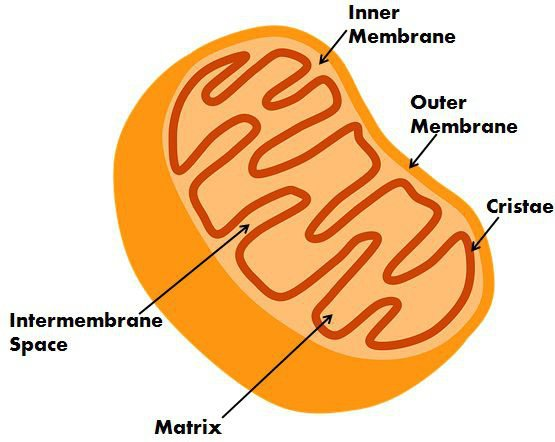
Matrix
the fluid that fills the mitochondria
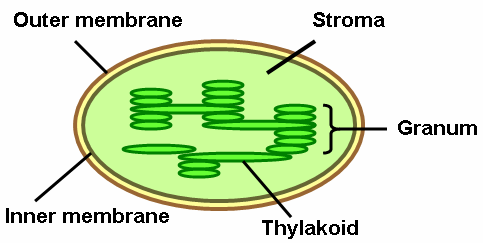
Chloroplast
has chlorophyll
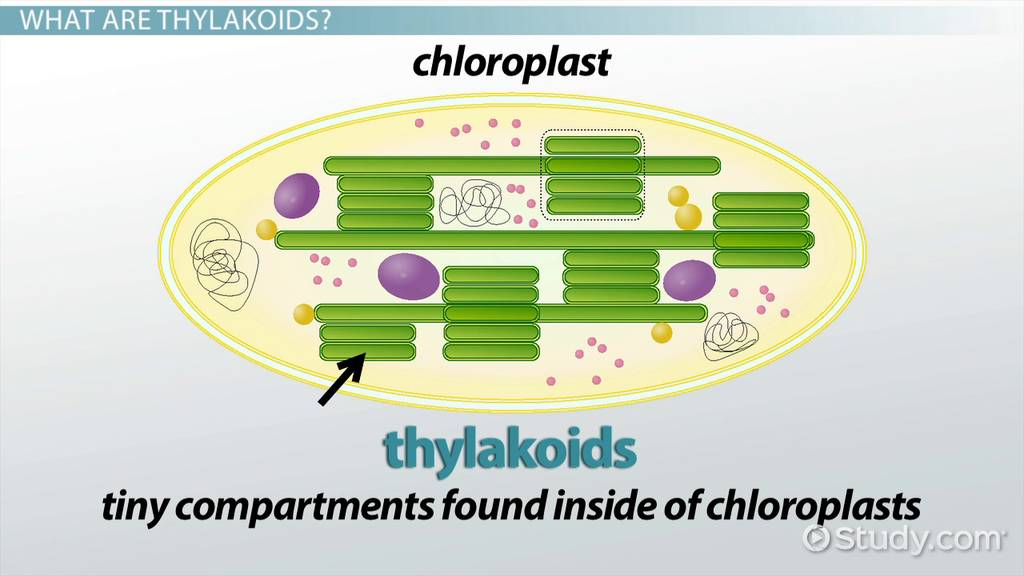
contains thylakoids
filled with stroma
has 2 membranes
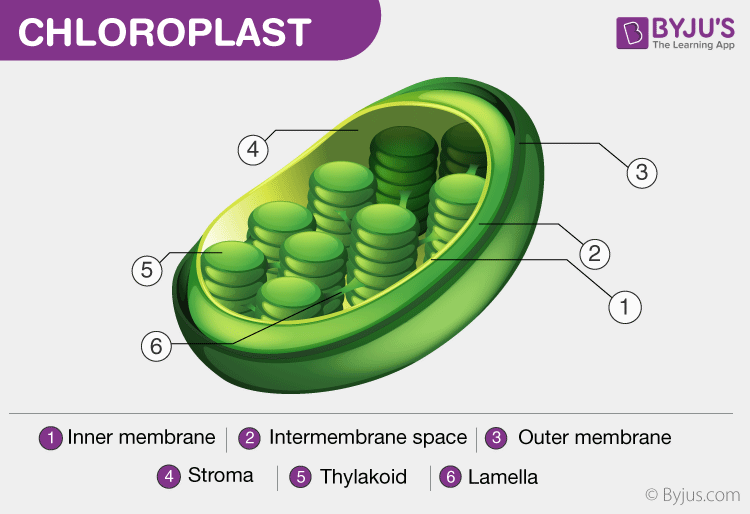
Thylakoids
smalls disks in the chloroplast
absorbs sunlight
Stroma
fluid that fills the chloroplast
carbon cycle

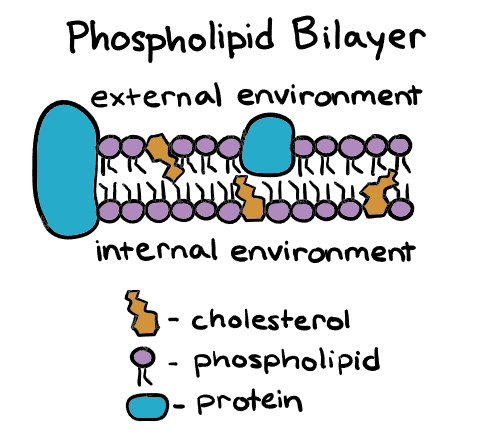
Cell membrane / Plasma membrane
seperates the inside and outside of the cell
is a phospholipid bilayer with proteins in between
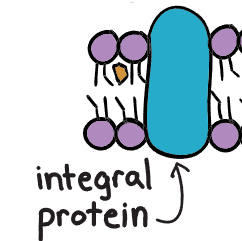
integral proteins
periphe
held together by LDF
molecules can move freely within the membrane
this allows holes to be more easily repaired
has the fluidity of vegetable oil
Exists in both animals and plant cells
Fluidity
affected by
temperature
Higher temperature = higher fluidity
phospholipid structure
more unsaturated = higher fluidity
cholesterol
acts as a buffer
@ high temp: increases melting point
@ low temp: goes in between phospholipids to prevent clustering
Cytosol
Fluid in cytoplasm
Integral protein
transports large molecules
acts as an enzyme
cell recognition
signal transduction
exists for both the inside and outside of cell
Nucleolus
inside the nucleus
contains RNA
synthesizes ribosomes
Small vacuoles
stores:
water
salts
carbohydrates
proteins
disposes waste
small
only in animal cells
Centrioles
bundles of microtubules
organizes movement of chromosomes during cell division
Ribosomes
Synthesizes proteins
Found on Rough endoplasmic reticulum
Osmosis
Diffusion of water / solvents through a semipermeable membrane
Osmotic Pressure
Minimum pressure applied to solution to change the flow of osmosis
Isotonic
Solutions that contain the same concentration of water and salt outside
same osmotic pressure
cells retain their shape
Ideal for animal cells
Not ideal for plant cells
Hypertonic
Solutions that have a high salt concentration and low water concentration inside
high osmotic pressure
Not ideal for animal cells & plant cells
Cells will shrink because it’s trying to create equilibrium (plasmolysis)
Hypotonic
Solutions that have low salt concentration and high water concentration inside
lower osmotic pressure
Not ideal for animal cells
cell will swell and bursts (lysis)
Ideal for plant cells
swells
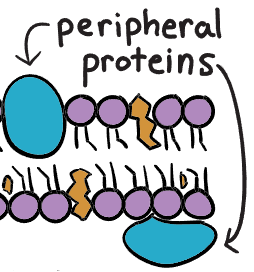
Peripheral proteins
structural support
transportation
loosely bound to plasma membrane
exists only on one side of the cell membrane
Channel proteins
proteins that allow transport of ions across the membrane
opens pores through the membrane to allow diffusion of smaller molecules
lipoproteins
Carrier proteins
proteins that bind to the specific molecule to transport it
slow rate of transportation
often used for macromolecules
glycoproteins
uses energy
uses active transport / facilitated diffusion
Active transport
movement of substances across a cell membrane
low concentration —> high concentration
uses ATP
includes: Primary active transport & Secondary active transport
Facilitated diffusion
movement of substances
may involve usage of pores in the cell membrane
high concentration —> low concentration
uses the flow of a concentration gradient
does not require energy
passive transport
Concentration gradient
the difference in concentration of a substance between two areas
allows cells to use energy to create new molecules & proteins
Electrochemical gradient
electrical gradient + concentration gradient
difference in charge & chemical concentration across a membrane
affects ions
used for nerve conductions, muscle contractions and cellular respiration