Endothermic and exothermic reactions(OG)
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Energy
The total energy of a system, is a total of the chemical energy stored in bonds and the kinetic energy of the substance.
What must happen for a chemical reaction to occur?
the bonds between the atoms in the reactant must be broken and new bonds between the atoms in the product must be formed.
What does the forming of bonds do?
Releases energy
What does the breaking of bonds do?
Absorbs energy
Endothermic Reaction
A chemical reaction that absorbs heat energy from the surroundings.
What are the features of an endothermic reaction?
1.Takes in heat energy from the surroundings
2.The temperature of the surroundings decreases
3.The energy needed to break old bonds is greater than the energy released from forming new bonds
4. the total of the products is greater than the total energy of the reactants
5.ΔH is positive
Exothermic reaction
A chemical reaction that releases heat energy to the surroundings.
What are the features of an Exothermic reaction?
1.Releases heat energy to the surroundings
2.The temperature of the surroundings increases
3.The energy released from forming new bonds is greater than the energy needed to break old bonds.
4. energy of the products is less than the total energy of the reactants
5.ΔH is negative.
ΔH
The symbol representing the change in enthalpy in a chemical reaction.
Law of Conservation of Energy
The principle that states that energy cannot be created or destroyed, only transferred.
•All energy lost or gained must go or come from somewhere.
•It is usually in the form of heat or light.
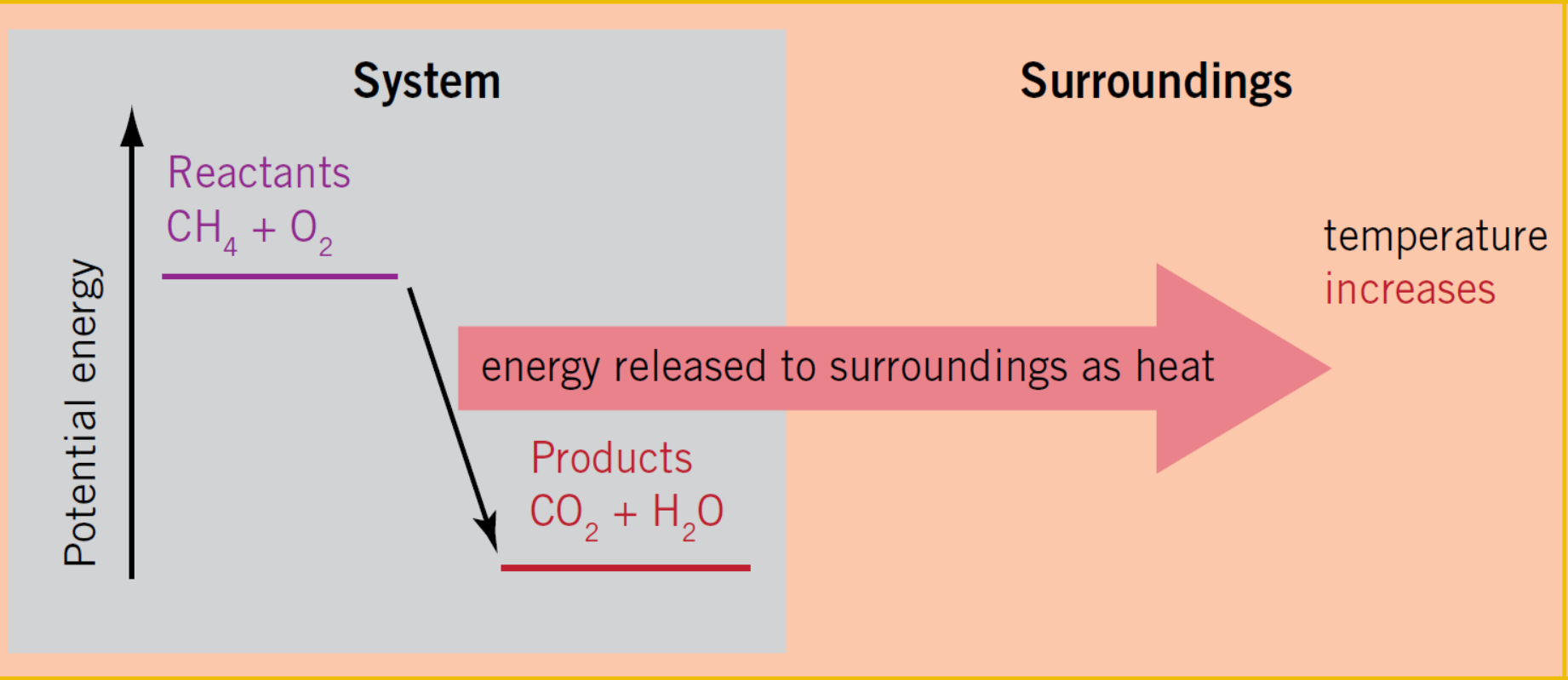
What is the reaction type for this diagram?
Exothermic
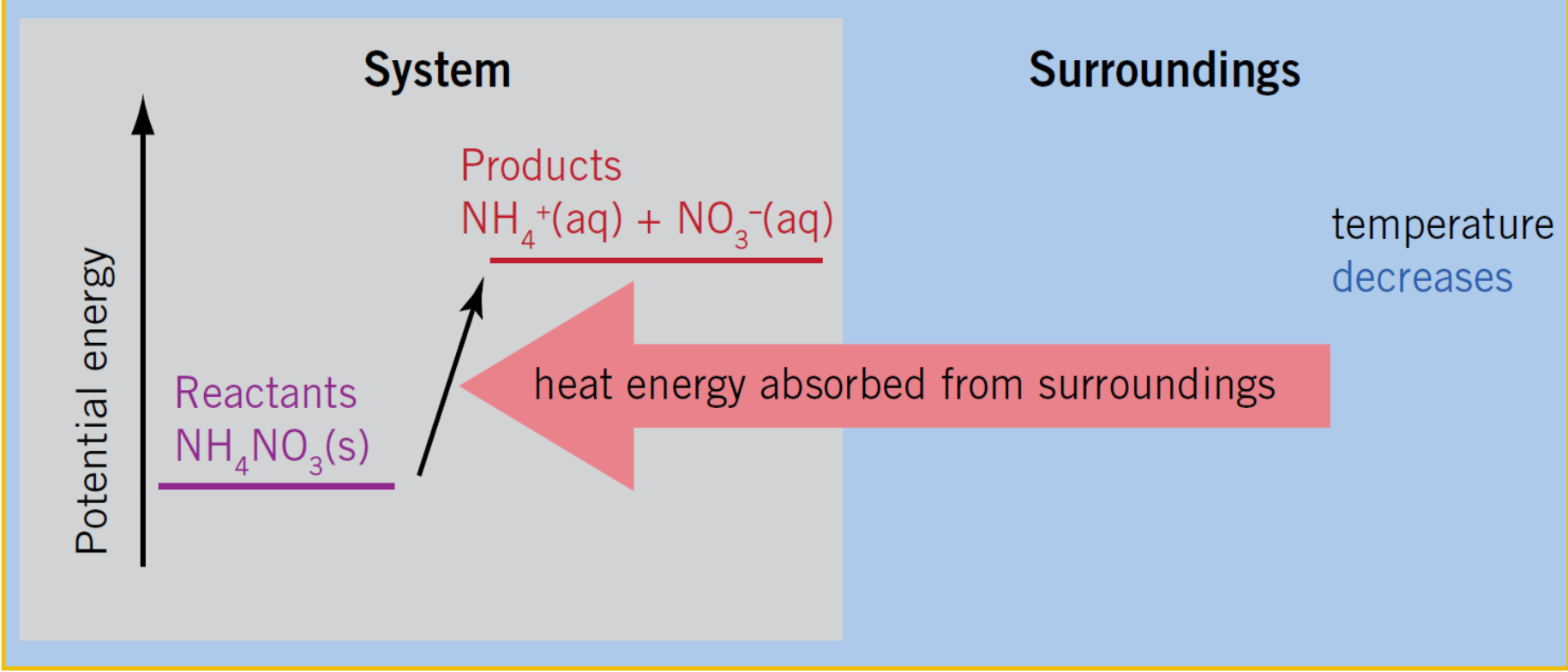
What is the reaction type for this diagram?
Endothermic