Lec 9 Biogeochemical cycles
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms

A
Most ocean doesn't have sunlight -> no photo -> biggest limiting availability
No mention of photic
Nutrient would be more limiting then
What are global biogeohemical cycles?
linked network of biological and physical processes that moves nutrients through pools within the environment
Nitrogen, phosphorus, carbon -> from food sources -> get them from plants
Cycle impacts how available they are to living organism
What are pools?
large reservoir of nutrients (e.g., oceans, atmosphere, soil)
Reservoir that is not moving
Not being used by living
What is flux?
movement of nutrients between pools
evaporation & precipitation
carbon-fixation through photosynthesis & respiration
Examples
Taken out of atmosphere -> moves into soil
Nitrogen movement
Take atmospheric carbon -> plants make starchs -> moved = flux
How can nutrients be moved?
Nutrients can be carried long distances by winds as gases and particles
by waters of streams and oceans as dissolved solutes or as particles
Phosphorus in form of Phosphate lost in ground water washed downstream
What is residence time?
Residence time: how long the molecules stays in one pool
What is the residence time in the hydro cycle?
Ice sheet: maybe 20k years frozen
Atm: evaporated -> only in atm for couple of days before being rained out
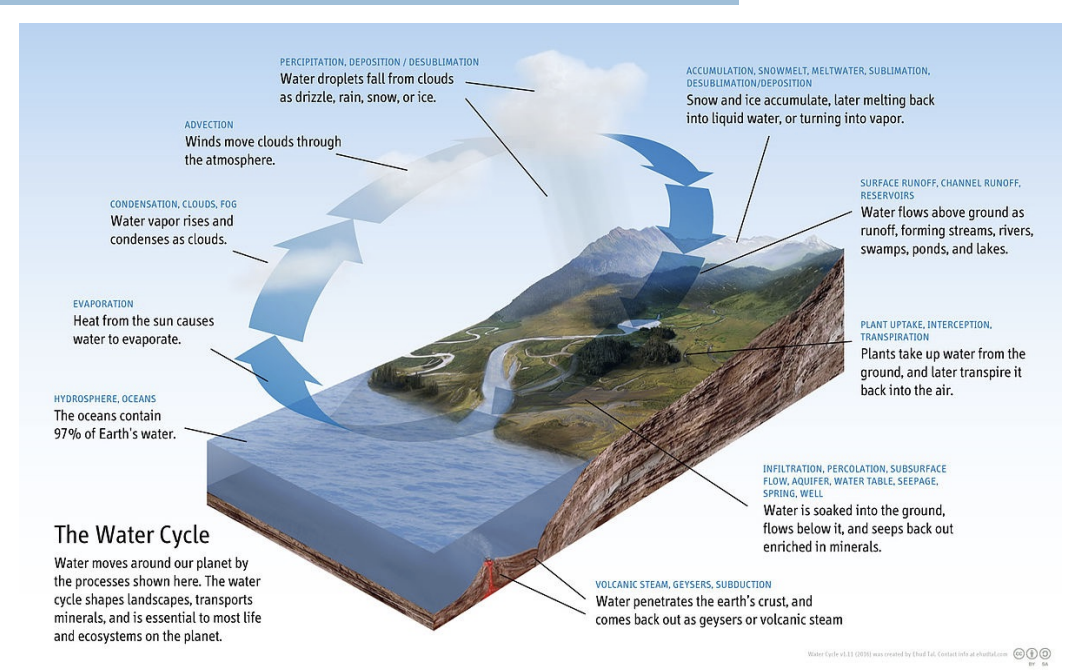
What is the hydrological cycle?
Most rain and show made from evap of oceans
Evapotranspiraiotn can also make rain
The biggest pool is the ocean (contains ~97.5% of earth’s water).
Only 0.08% of global water is in flux at any given moment.
What happens as the Earth gets hotter?
Heat Earth -> Ocean hotter -> more evap -> speeds up water cycle -> more powerful hurricanes , cyclones, more frequent -> more flooding
Speed up water cycle -> more water natural disasters
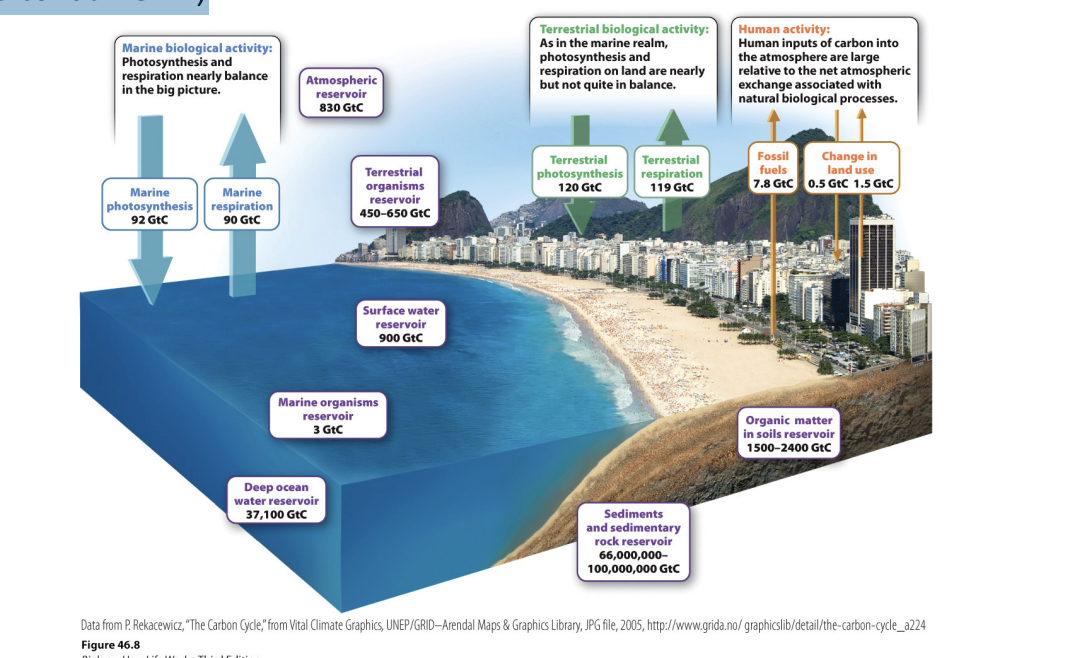
What are the major pools for the global carbon cycle?
Major pools include atmosphere, oceans, land surface (soils and vegetation), sediments and rock
Sediments and rock → 99% of global C
largest pool, acts as carbon sink
Ocean and rock big reservoirs
Most are in the atmosphere (atmospheric cycle – where most fluxes happen where it moves out and in)
Once in pools -> could be there for a long time
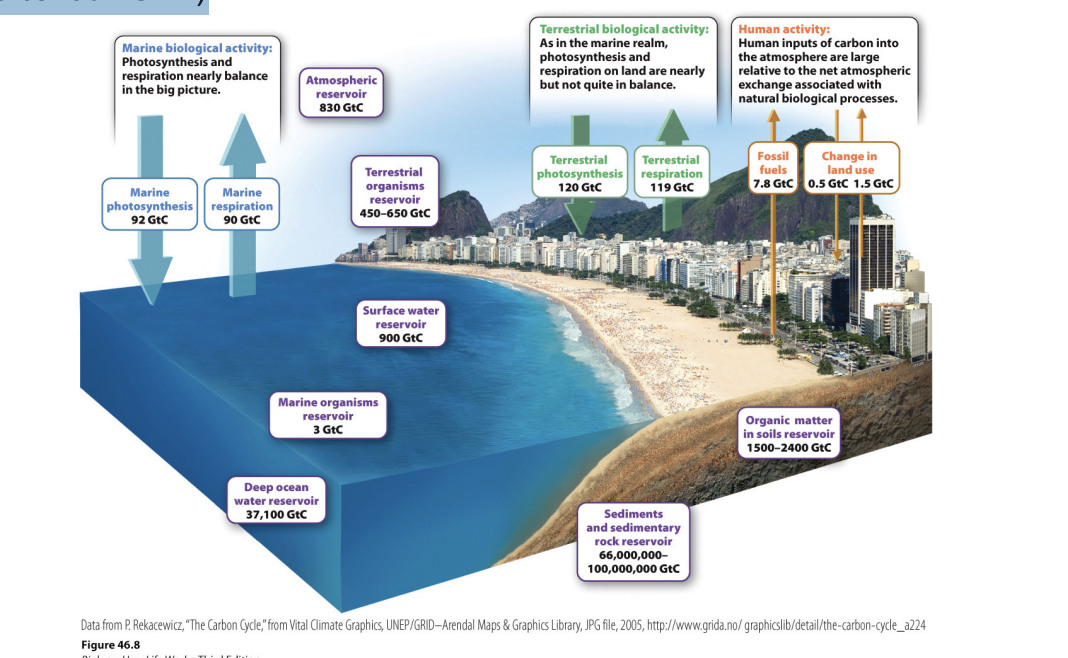
What is the difference between human fluxes and natural carbon fluxes?
Orange: human fluxes and much smaller -> don’t move as much carbon as natural fluxes
Uneven (only .5 out, more in atm) -> actions not in balance
Atmosphere thorough living organism (green arrow) = photo
Return carbon to atmosphere: cellular resp
200 Gt moving in and out of atm
Even each other out
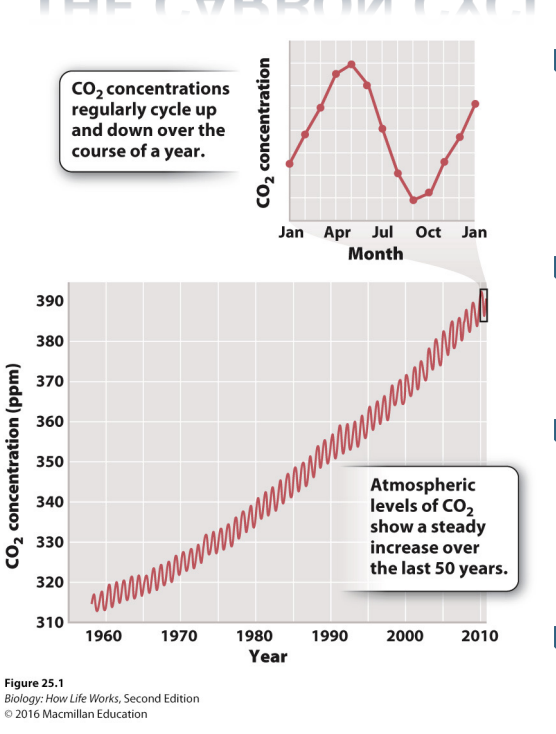
How does the carbon cycle change anually?
Record of atmospheric CO2 levels, as measured from Mauna Loa, Hawaii, from 1950s to 2010s
Above trees -> not getting atm averages from no local sources (houses etc)
Inc in carbon in atm
Annual pattern with seasonal oscillations
highest in early spring
lowest in early fall
47 billion metric tons of CO2 enters and leaves atmosphere every year due to this annual pattern
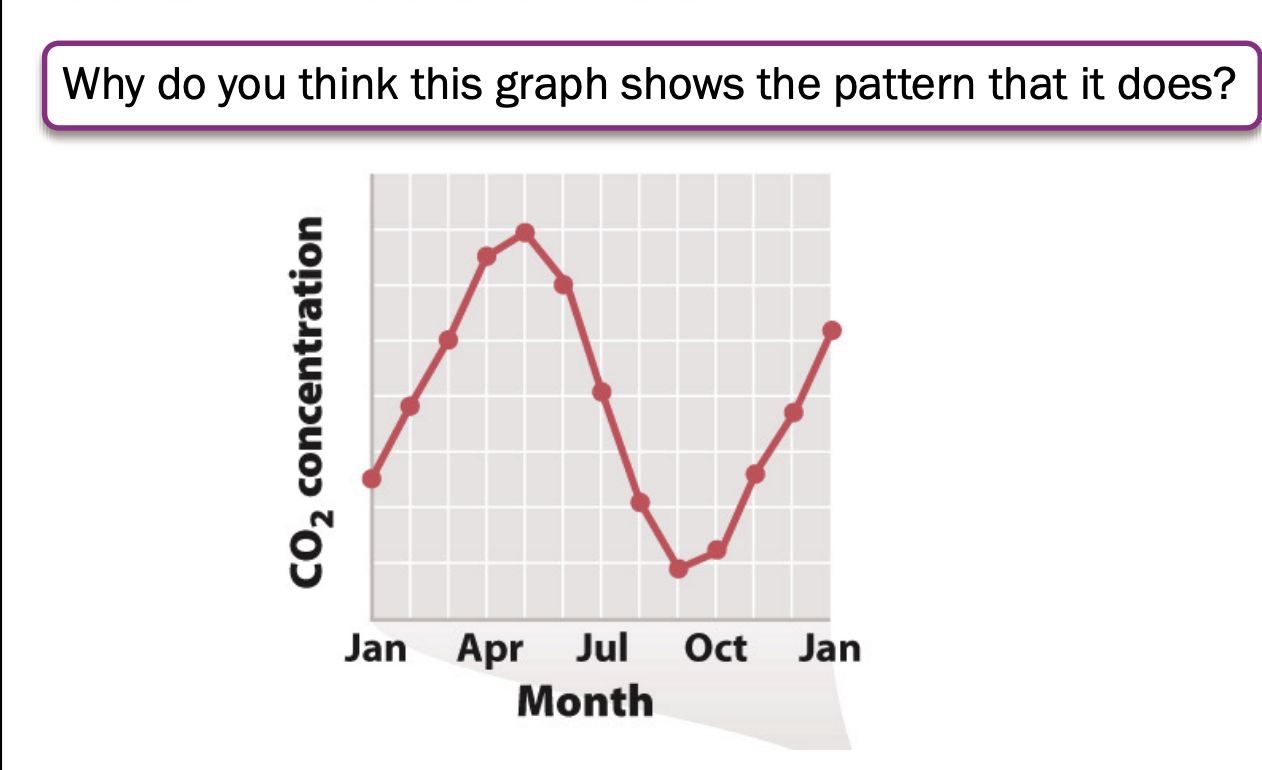
Respiration fairly constant, photo is not
Atm high in april
No leaves -> carbon increases
Lowest in sept oct
Trees active -> carbon levels decrease
Matches northern hemisphere (where most land is found) -> cycle driven by big land plants
Partly offset by southern hemi, but it has less land
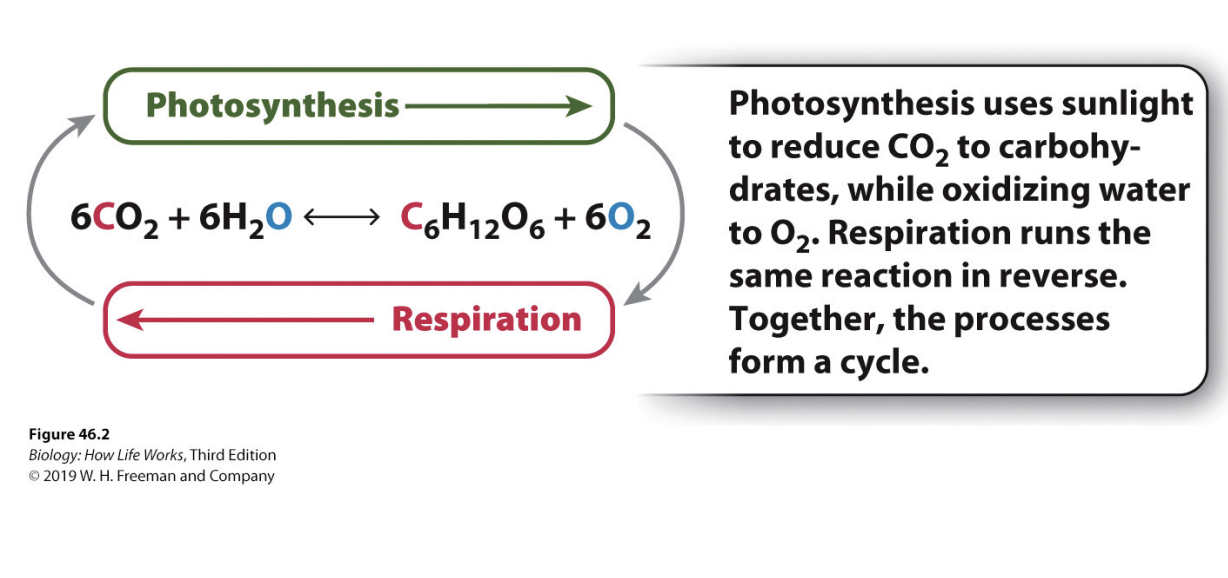
How is Photo and respiration related?
Photosynthesis and respiration typically equalize each other across year
same amount of CO2 taken out of atmosphere as enters it because of these processes
Photo: uses sunlight to reduce CO2 + oxidizing O2
Resp: same reaction in reverse
form cycle
Where is more land?
There is more land in the north
More land in North with more seasonal tropical forests ->
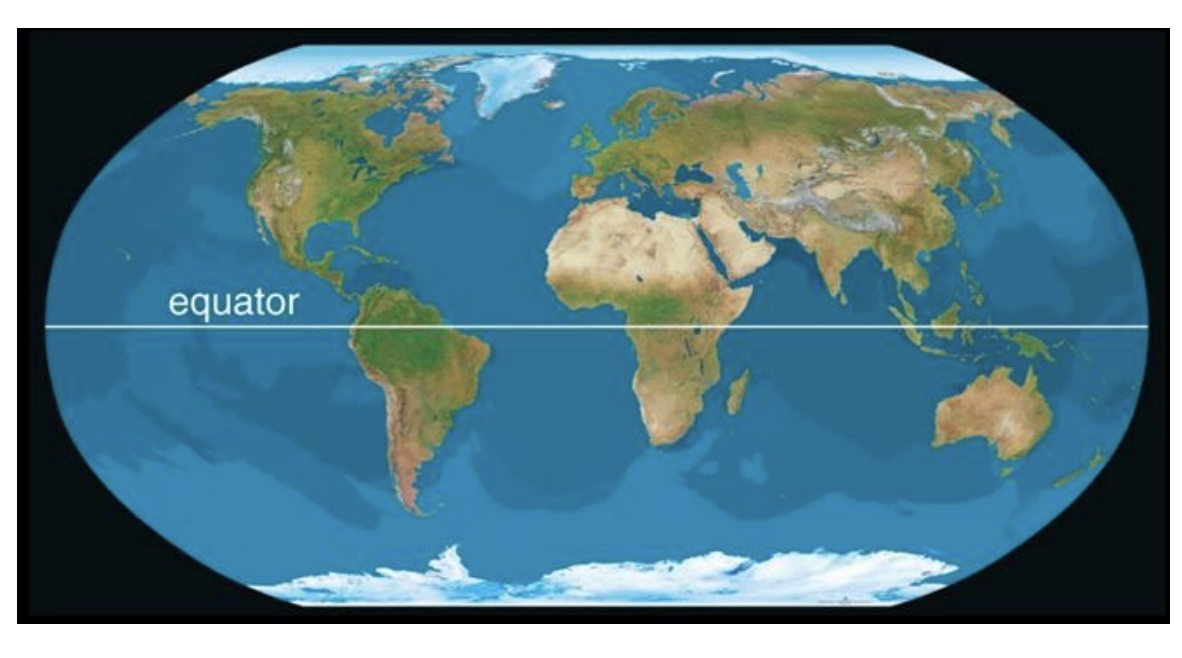
How has CO2 levels increased?
atmospheric CO2 levels increased by more than 30% in 60 yrs
If atmospheric CO2 levels have increased, then CO2 entering the atmosphere must exceed CO2 leaving it.
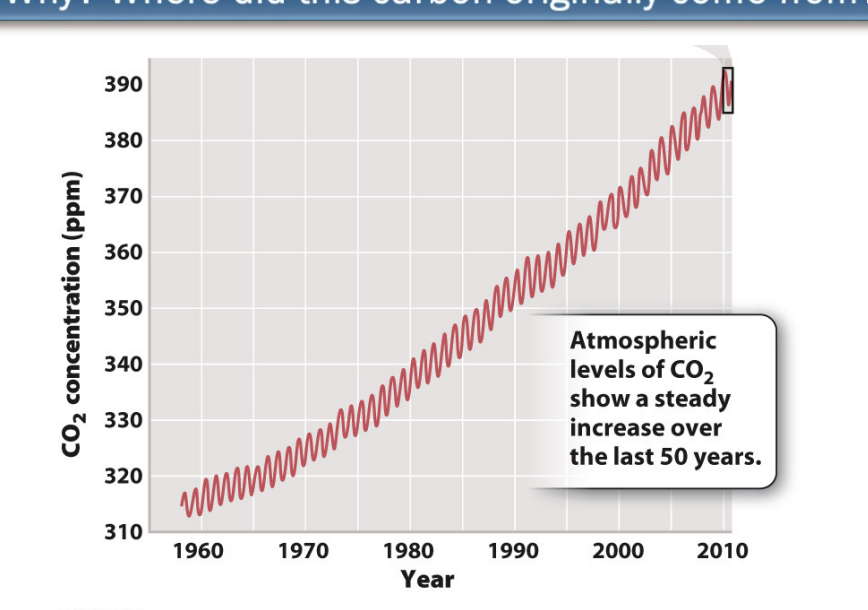
How does carbon dioxide enter the atmosphere?
Geological inputs (volcanoes, mid-ocean ridges)
Volcanic release methane, CO2
MOR: hydrothermal vents
Biological inputs
respiration
Anthropogenic inputs (burning fossil fuels, deforestation)
Burning fossil fuel and deforest
Both contain carbon
How does carbon dioxide leave the atmosphere?
Geological removal
chemical weathering → CO2 in rainwater reacts with exposed rocks
Biological removal
photosynthesis
What is BIOLOGICAL INPUT/REMOVAL or the terrestrical carbon cycle?
CO2 in atm → photo → primary produces that fix CO2 into organic carbon → primary consumers feed on primary producer → secondary consumer
aerobic resp + anarobic resp from decomposers in soil that feed on organic detritus
Co2 comes out atm and goes back to atm
Examples
Co2 -> plant -> decomp -> release to atm
Co2 -> plant -> eaten -> poop or die -> decomp
Giraffe breath it out
Anything in soil gets decomp -> released back into atm
Sometimes goes to sediments -> can stay for awhile
In food webs release to atm
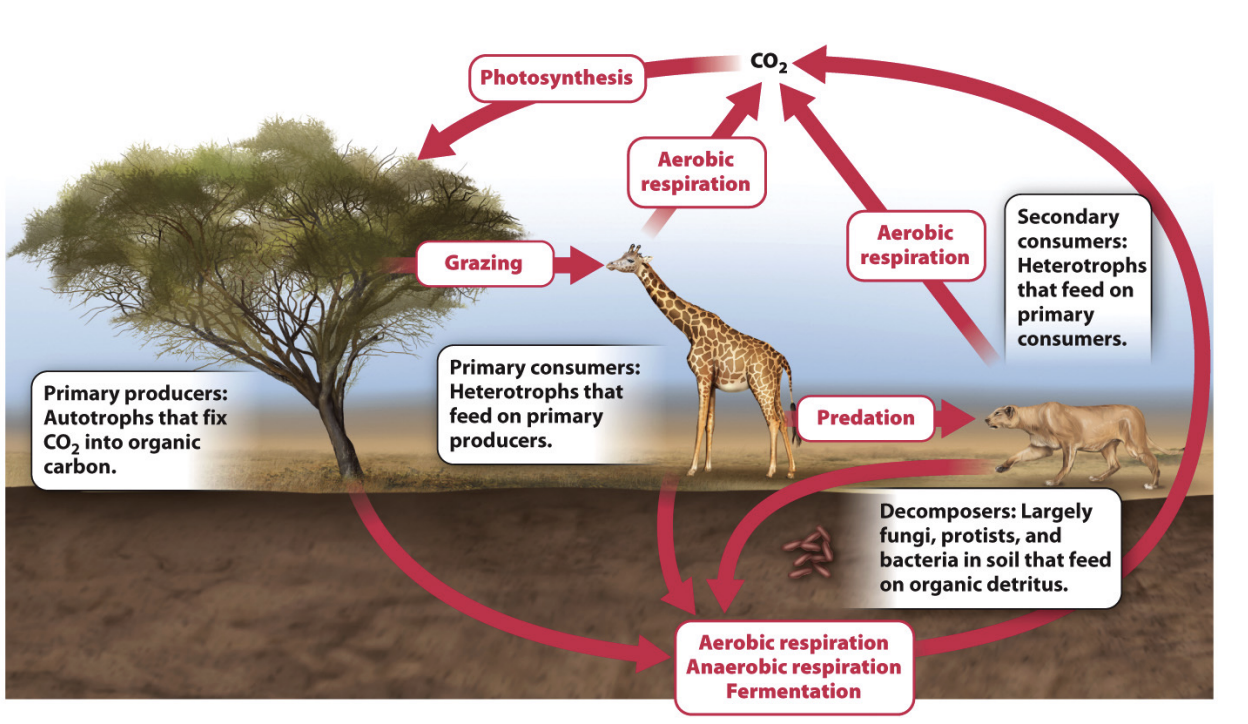
What is the biological input and removal marine carbon cycle?
Open ocean: planktonic algae and cyanobacteria are major primary producers → protists and small planktonic animals are primary consumers that feed on primary producers → fisher and other secondary consumers more carbon through food webs → bacteria decompose organic matter and →. serves as food for other organisms
Lose it to ocean sediments
Deeper oceans where its colder in form of organic matter or methane -> methane can freeze -> stays there
Get into sediments as carbon -> come back through food webs from upwelling -> move btwn water and atm
Carbon stored as acid in water
What can happen with warming temperatures in the ocean?
Lose it to ocean sediments
Deeper oceans where its colder in form of organic matter or methane -> methane can freeze -> stays there
Great dying (shut down of gulf): world gets hot -> ocean gets hot -> methane becomes gaseous methane -> greenhouse gas -> even more warming
Worst mass extinction event we know of
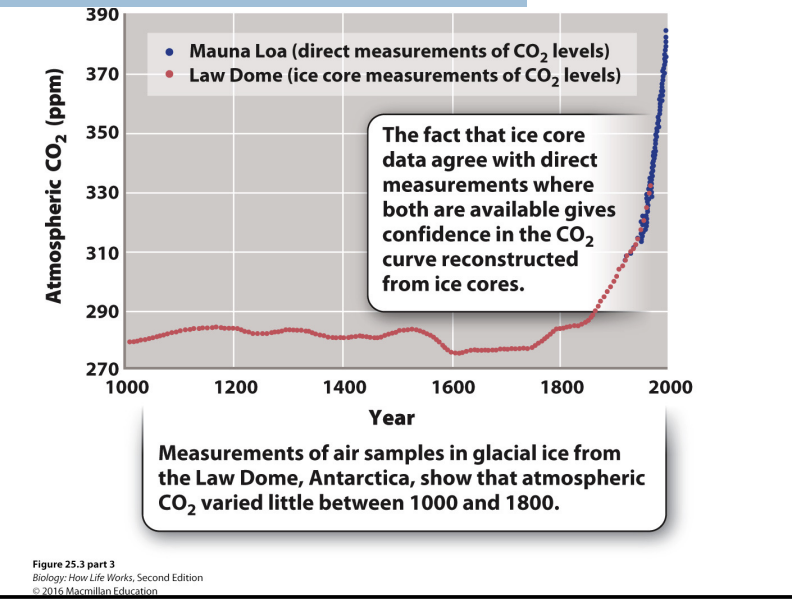
Is the increase in CO2 unusual?
Co2 levels change over time
Glacial ice as it forms → traps air bubble → ice can last for thousands of years → examine ice cores to find out what the atmosphere was like in the past.
High: greenhouse conditions
Low: ice house (where we are)
Holes that are frozen -> dig in ice caps (ice forms anually and layers) -> look for air pockets -> graph
Graph
Large increase from 1950
Something changes 1700s-1900s: humans started using fossil fuels
What are the isotopes of carbon?
12C - 99% of all carbon
13C - ~1% of all carbon atoms
14C - 1 ppt (part per trillion) of all atmospheric C
unstable → decays to 14N
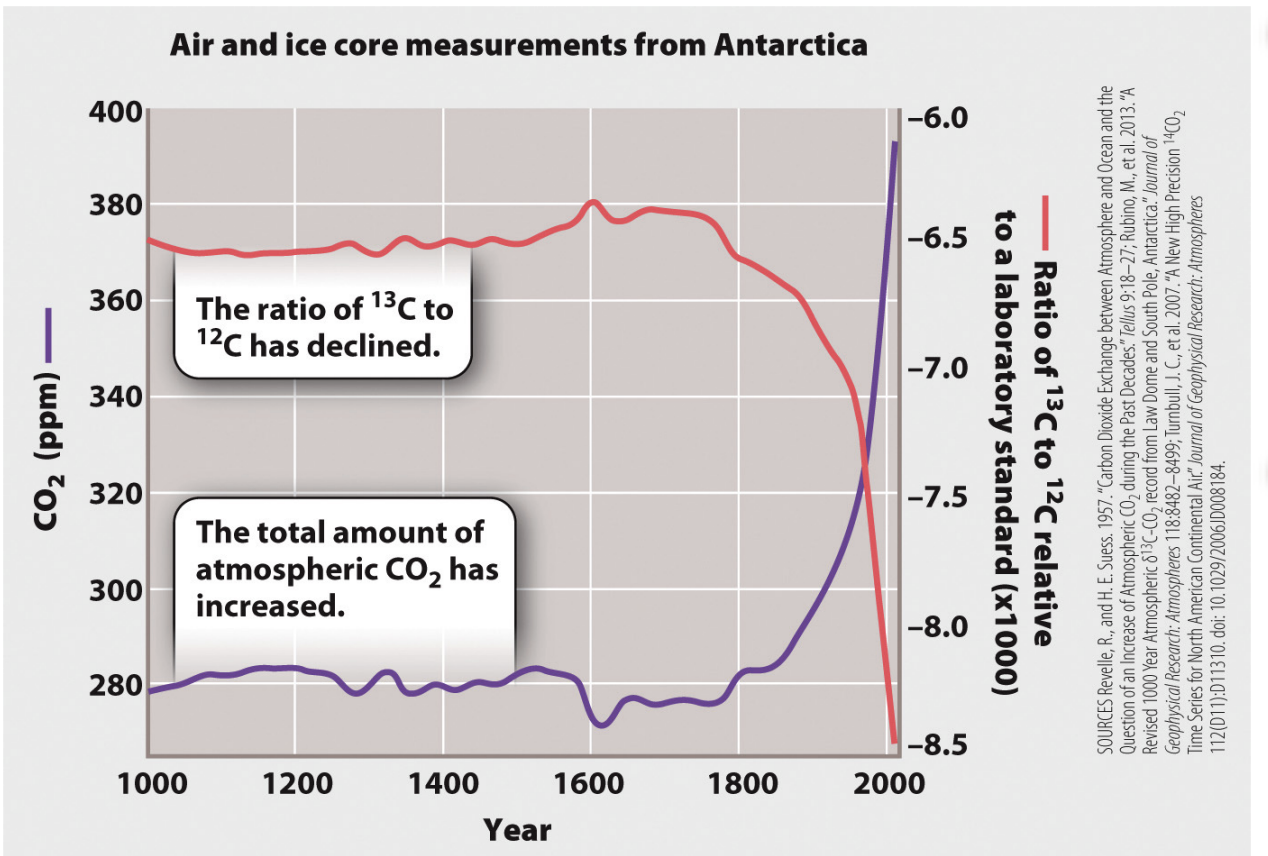
Was this increase in CO2 from human activities?
Atmospheric carbon levels have increased, but the ratio of 13C to 12C has declined.
Source of excess carbon is 13C depleted compared to carbon originally in the atmosphere
Match concentration to source
C14 is not stable -> radiodecays (use for carbon dating)
Die -> stop taking in C14 -> decays
Blue line: carbon levels have inc
Red line: source that is 13C depleted
12/13 ratio has decreased over tie -> doesn't match atm
Summary
Atmospheric CO2 increasing, but natural ratio of 13C/12C ratio is declining (can’t use 14C because it decays) is decreasing → CO2 must be coming from another source
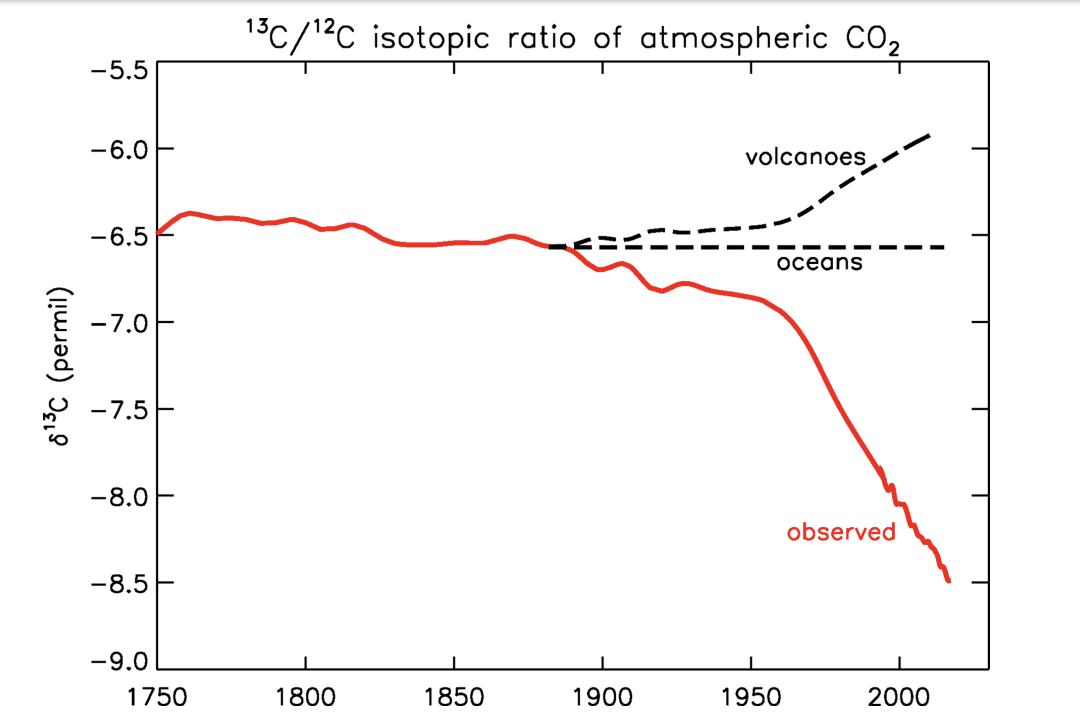
How is carbon used in plants?
plants preferentially incorporate 12C (over 13C) into biomolecules
Organic matter contains a higher proportion of 12C as a result
Plants 13/12 ratio differ from atm
Less 13 than expected from the ratio in atm
What kind of carbon is found in nonorganic sources?
carbon released from other sources (volcanic gasses or dissolved seawater) has higher proportion of 13C
sources do not match the isotopic ratio of excess carbon
too little 13 C in atm

What does the mismatch in inorganic carbon sources mean?
neither volcanic gases nor dissolved ocean waters have 13C/12C rations that mach the increase in atm carbon
If came from volcanoes or upwelling etc -> 13/12 ratio should inc
More 13 in atm
Doesn't match
Line matches plants
Plants alive today
Plants that died in carboniferous -> fossil fuels
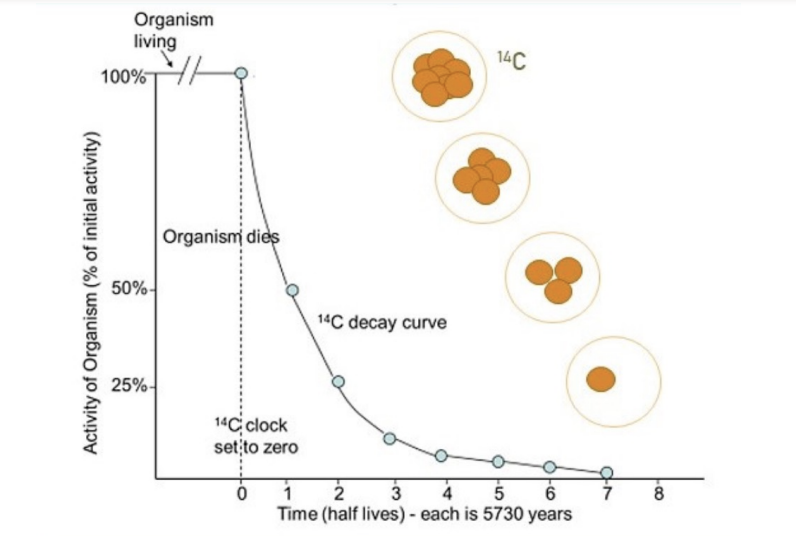
What can 14C be used for?
living organisms continually incorporate 14C into their bodies throughout life
14C decays to other forms of carbon
organism dies → amnt of 14C in body decays over time → radiocarbon date fossils
14C ratio
Comes from source that 14C depleted -> old plants
Land just started to colonize -> fell in hypoxic bogs -> turned into fossil fuels
Is this source modern or from fossil fuels?
Additional studies showed that carbon entering in the atmosphere was 14C-depleted → modern plant vegetation contains too much 14C to be the source
Ancient organic matter (coal, petroleum, natural gasses) → isotopically similar to carbon entering atmosphere
Inc in Co2 is b/c of burning of fossil fuels
Also from deforestation, but lesser
Carbon in atm is anthro
How much carbon pollution do humans create?
HUMANS PRODUCE 9 GIGATONS (1 billion metric tons0 OF CARBON POLLUTION ANNUALLY
Photosynthesis and respiration move far more carbon in/out of atmosphere annually than we do
However, our flux is largely unidirectional, and photosynthesis and respiration generally offset each other
don’t move as much carbon in and out as others -> humans largely unidirectional
Burning fossil fuels in biggest cause
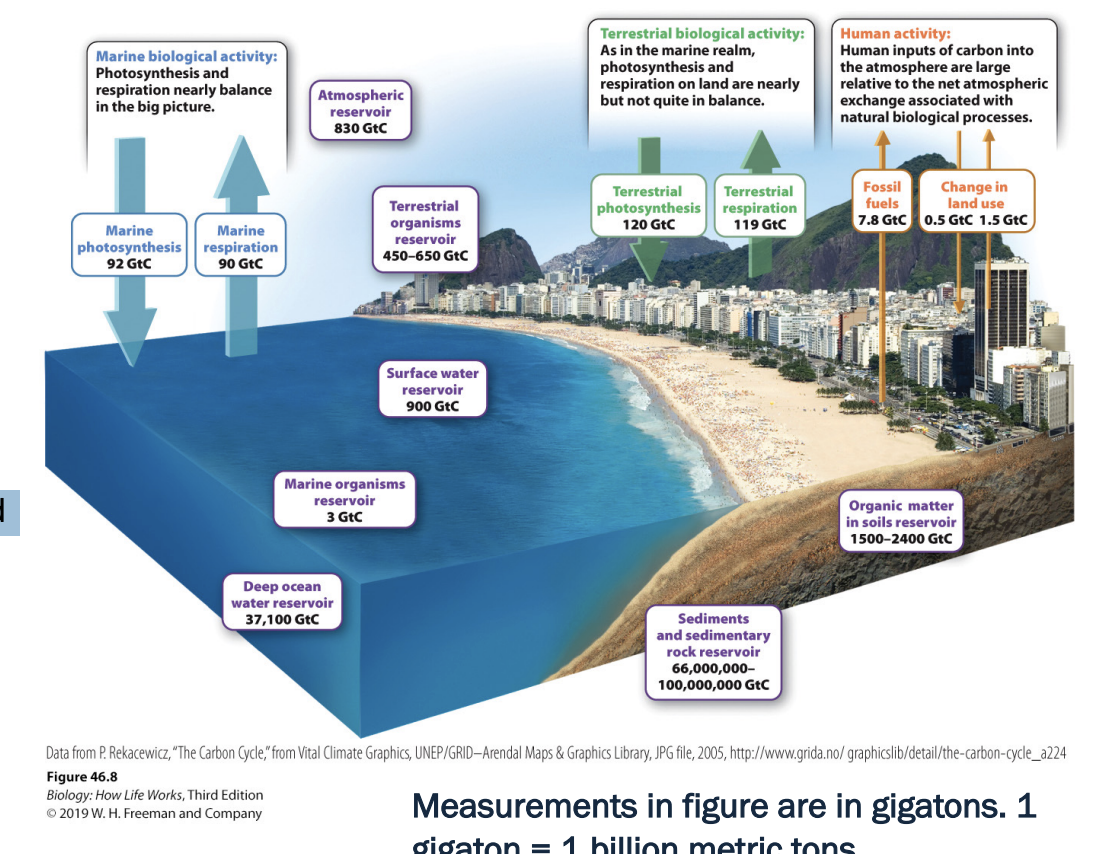
What anthro activities that add carbon?
~20% à land use changes (deforestation, agriculture)
~80% à burning fossil fuels
Not all ending in atm -> carbon stored in ocean water (deep oceans) -> eventually rise to surface and released into atm
Carbon come out of ocean eventually
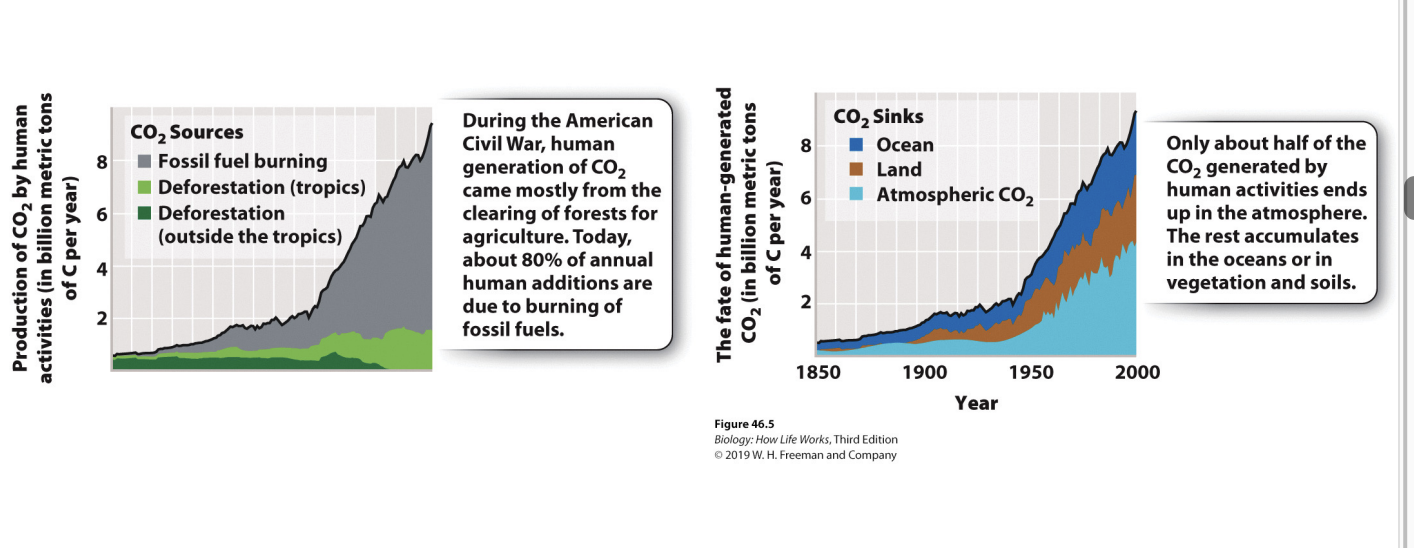
What is the nitrogen cycle related to?
This cycle is closely linked to the carbon-cycle because nitrogen is a critical component of amino acids, nucleic acids, membranes
What is the largest N2 pool?
The largest nitrogen pool is N2 gas
form not usable by most living organisms
usable by chemoautotrophs
What is nitrogen fixation?
process by which some bacteria and archaea reduce N2 gas to biologically useful NH3
this is how nitrogen enters food webs
N2 taken out of atm into ammonium
What is assimilation?
process by which primary producers obtain biologically useful nitrogen from surroundings (NO3 - or NH3)
take useful forms of nitrogen from soil or mutualistic that gets it from soil
What is denitrification?
a form of anaerobic respiration in which NO3 - is terminal electron acceptor
N in form of higher energy to lower energy
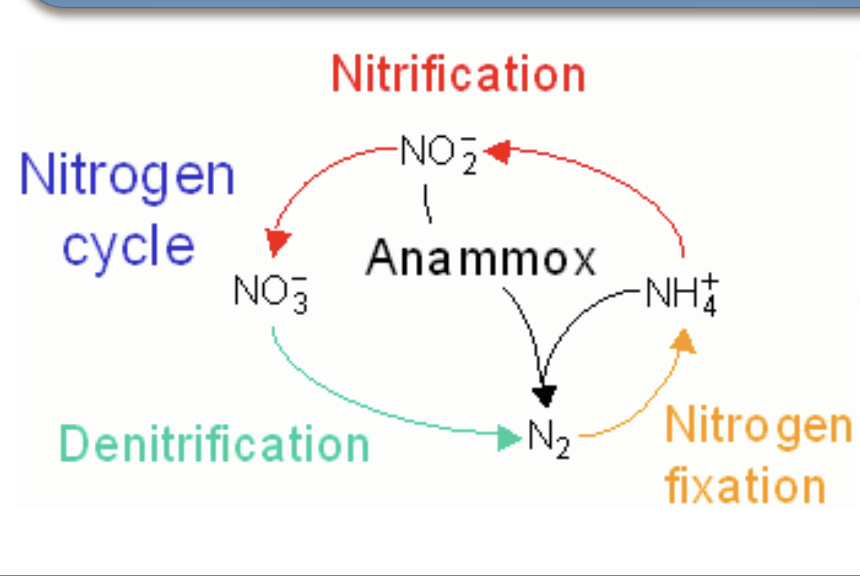
What is nitrification?
a process in which NH3 or NO2 - are oxidized to generate energy
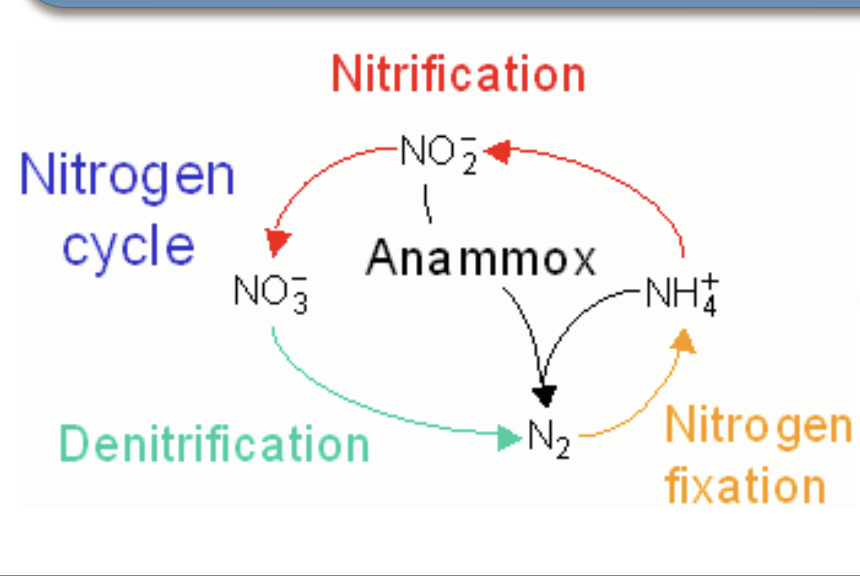
Why does nitrogen need to be fixed?
hemoautotrophic bacteria and arachaeons use NO3 - or NH3 to generate energy → releasing N2 gas as biproduct.
This causes biologically useful forms of nitrogen to decrease in the soil.
To obtain more, nitrogen fixers convert N2 gas into more NO3 - or NH3
Essentially, they do it because this is how they generate their own energy.
What is anamox?
Driven by chemoautotrophs to generate energy -> take it and go back to N2 gas
Other organisms take before they can get to it
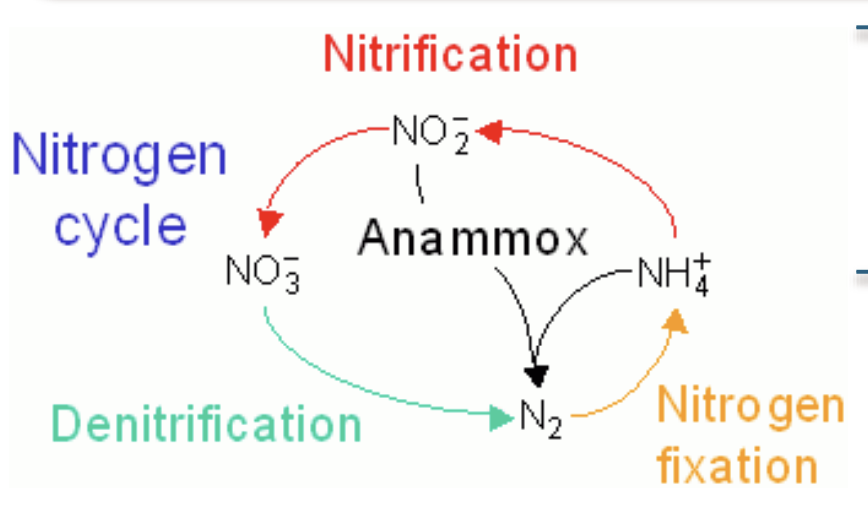
What is the nitrogen cycle?
Plants -> organisms -> back into soil (plants use ammonia) -> return to atm
Nitrogen fixation converts N2 → form that can be used by plants
primary producers assimilate biologically usable N from soil or symbiotic N- fixing bacteria
consumers obtain N from their food
decomposer return N to soil as ammonia
in Anoxic envi. some decomposing bacteria use NO3 - in resp → denitficiation (converts biological usable N back to N2)
Chemoautotrophic bacteria gain the energy needed to fix carbon by oxidizing NH3 using O2 (nitrificatoin) or NO2- (anammox)
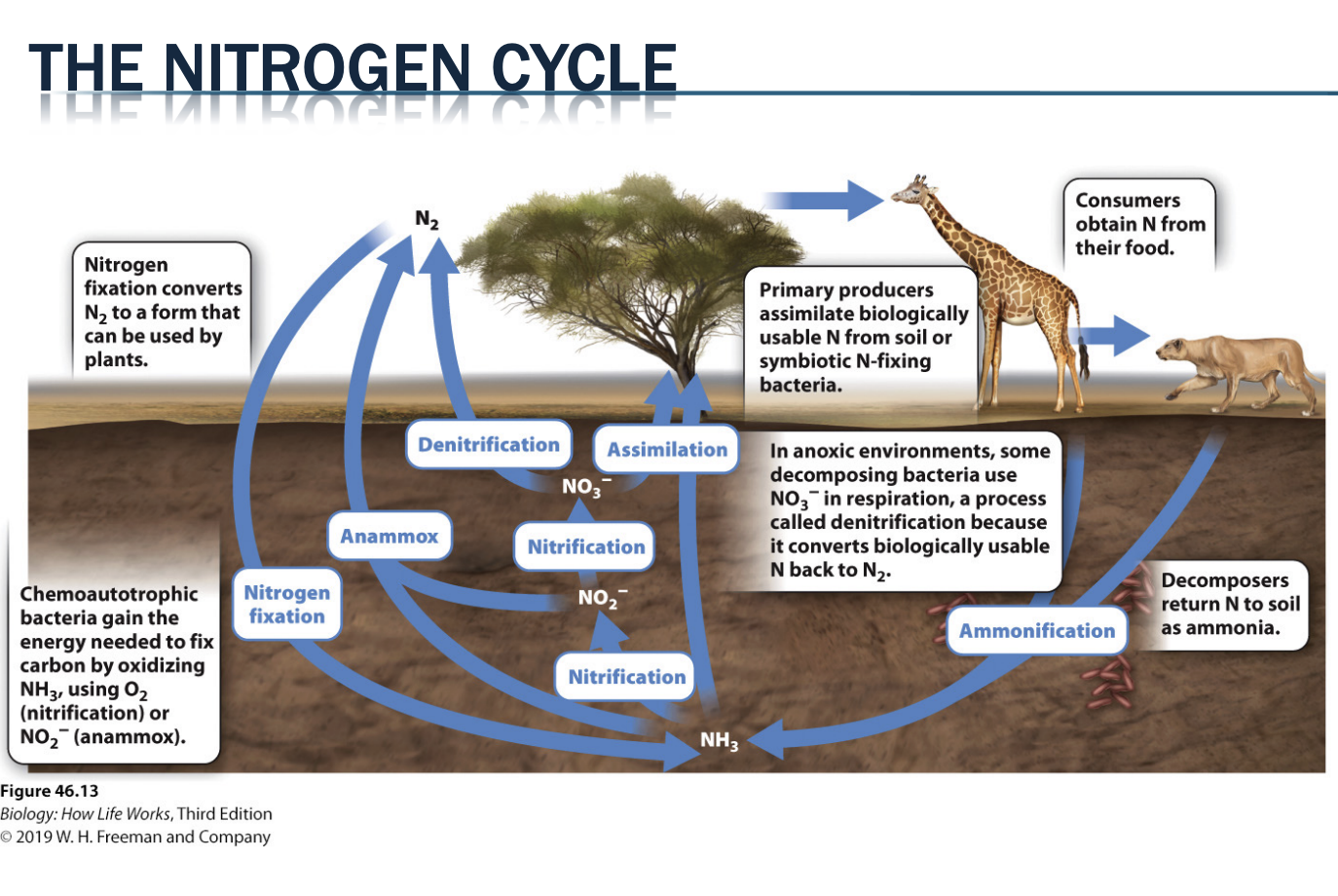
How do plants get nitrogen?
biologically useful forms of N can be in low supply, especially in young soils
plants can trade carbs with mutualists to obtain organic forms of the N they require
Lots of systems are nitrogen limited because most are in a form only chemotropic organism can use
Very young soils are nitrogen limited b/c they don't have the organisms
Form mutualists with chemoauthropic organisms
Already source of nitrogen -> convince mutualist (fungi) to get it
Soil releases carbohydrates into soil -> fungi hyphae grow towards carbs -> form mutualistic relation
Mutualists: can provide to N plants
Rhizobium bacteria
Ectomycorrhizae
What is the Rhizosphere?
soil layer surrounding actively growing roots, receives carbohydrates from plants to stimulate growth of soil microbial mutualists
What are Rhizobium bacteria?
nitrogen-fixers that exchange organically useful forms of N for carbohydrates
What is Ectomycorrhizae?
fungi that exchange water and nitrogen (obtained from soil) for carbohydrates
already have nitrogen -> fungi move it toward plant
Can protect plant form pathogens
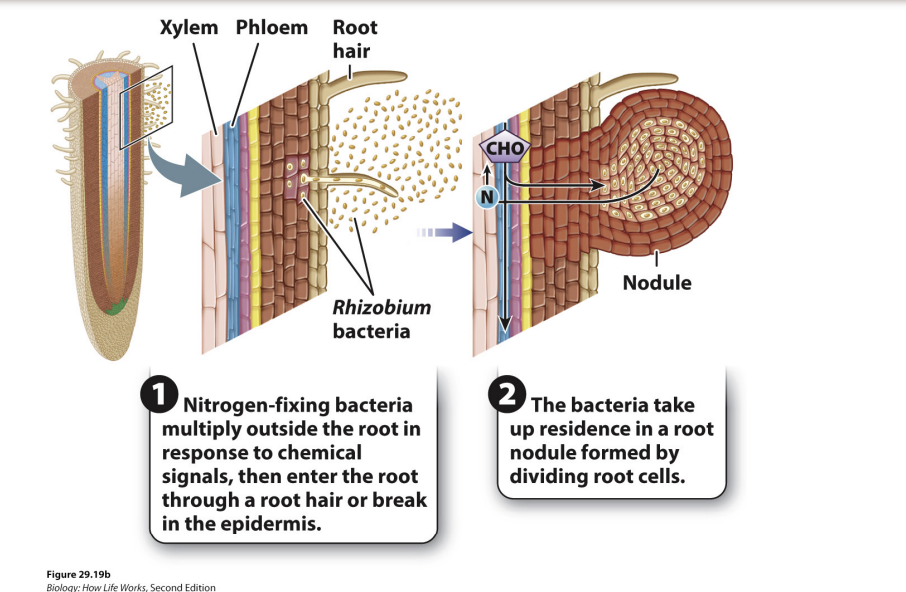
How does mutualism with rhizobial bacteria work?
Nitrogen fixing bacteria in the soil multiple in the rhizosphere in response to chemical signals from the plant roots
plants that form mutualisms with these bacteria are often referred to as nitrogen fixers
but, it is the bacteria that are doing the work
nitrogen fixing bacteria multiply outside root in response to signals → enter the root through a root hair or break in the epidermis → bacteria take up residence in a root nodule formed by dividing root cells
sugars in soil -> bacteria multiply -> plants grows around them and gives them carbs -> bacteria keep fixing nitrogen and give to plants
Rare mutualism
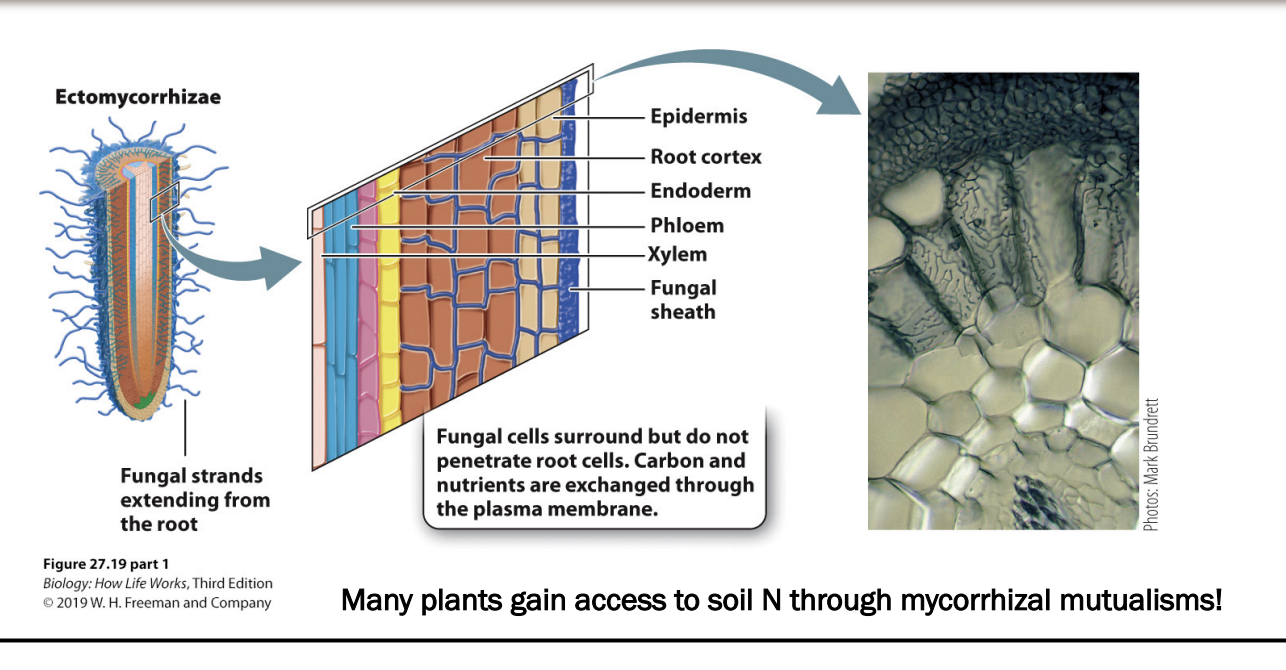
How does mutualism with ectomycorrhizal fungi work?
fungi produce a thick sheath of fungal cells that surround plant root tips and extend into root interior
fungal cells produce network of filaments that surround root cells → but do not penetrate them
Fungi grow hyphae quickly and move soil easily -> increases volume that plant can extract nutrients
fungi can provide root cells with N (obtained frmo soil) in exchange for carbs
In soli with nitrogen already (lots of decomp that releases ammonia and nitrogen fixing bacteria)
Why are mutualisms formed with mycorrhizae?
fungi can decompose organic matter to obtain nitrogen (good at obtaining other nutrients from soils)
hyphae are extensive → increases volume where nutrients are obtained
Some fungi form mutualisms with nitrogen-fixing bacteria to obtain N for themselves and their plant mutualists
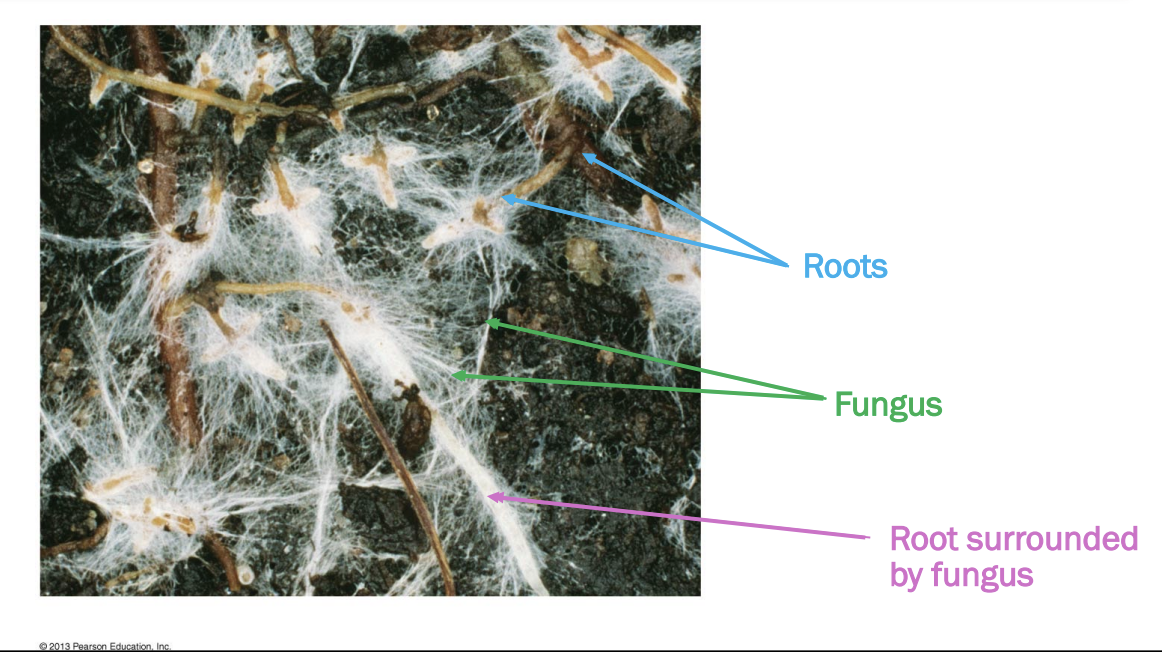
Where does mycorrhizae cover?
Each colour represents a different type of mycorrhizal association
Evolved ind multiple times -> covers most of land

D
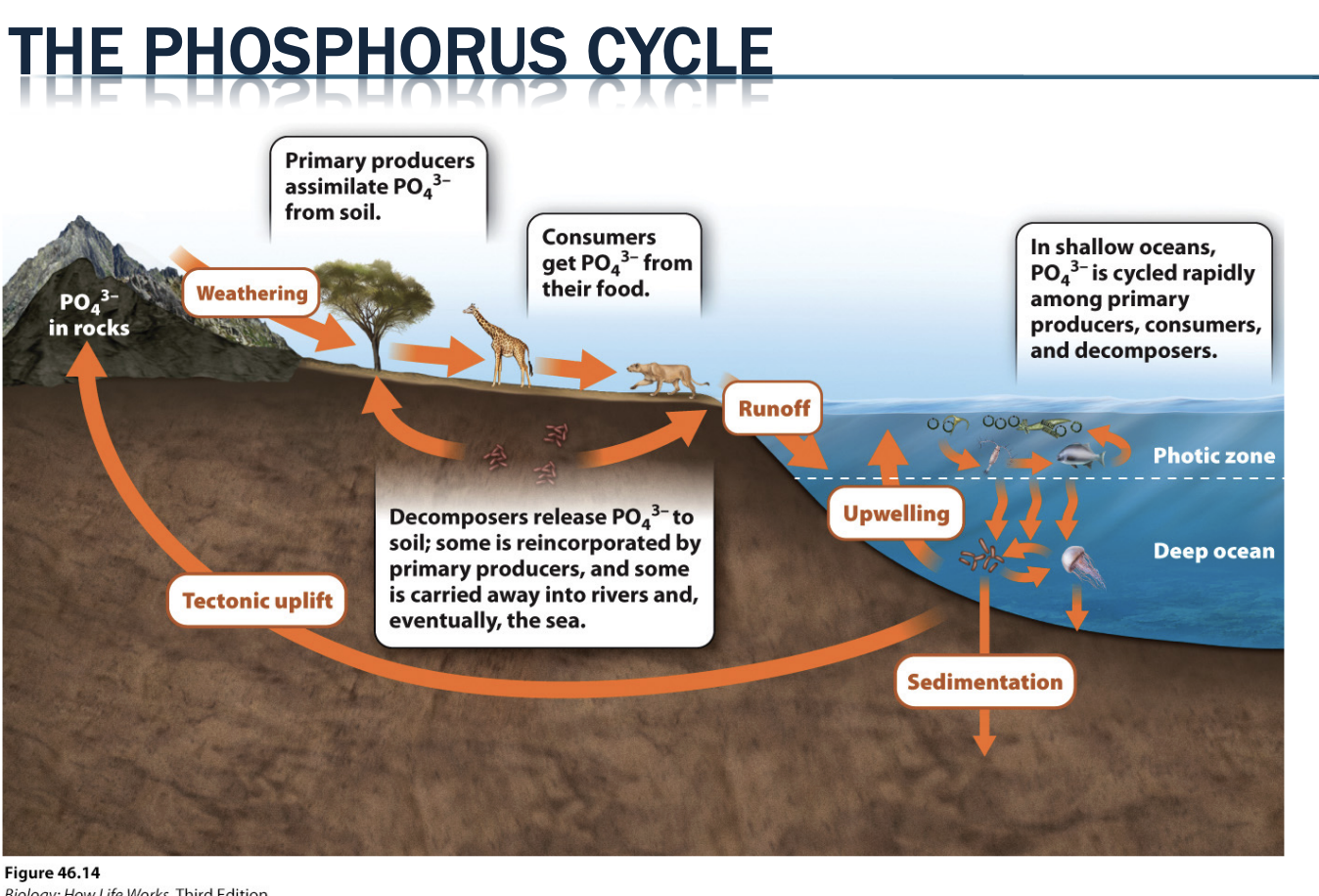
What is the phosphorus cycle related to?
Cycle is closely linked to the carbon-cycle because phosphorous is a key element for growth (nucleic acids and membranes, ATP)
What are the largest pools for phosphorus cycle?
Rocks & ocean sediments → largest pools, P atoms leave pools via uplift & chemical weathering
How does the phosphorus cycle work?
P enters food webs as PO4 3- , which is highly immobile in clay soils → hard for plants to obtain without help from mutualistic fungi
Enter as phosphate that are hard to move and hard for plants
Mutualisms with fungi
P leaves terrestrial ecosystems through soil erosion, leaching, and groundwater runoff, and marine ecosystems through sedimentation
Sedimentary cycle (not a lot in atm)
Phos moves to water -> goes into rocks
Leached from soils into ground water -> down-stream -> ends in ocean sediments -> makes continental drift to make mountain -> moves back into land
Can take over 100 millions years for a P atom to return to food webs once enters ocean sediments → coastal upwellings can considerably shorten this timing
Lost quickly
Ocean sediments can return with upwellings
Most time stays in sediments until ocean becomes land through cont drift and mechanically weathered again (and/or enter food web
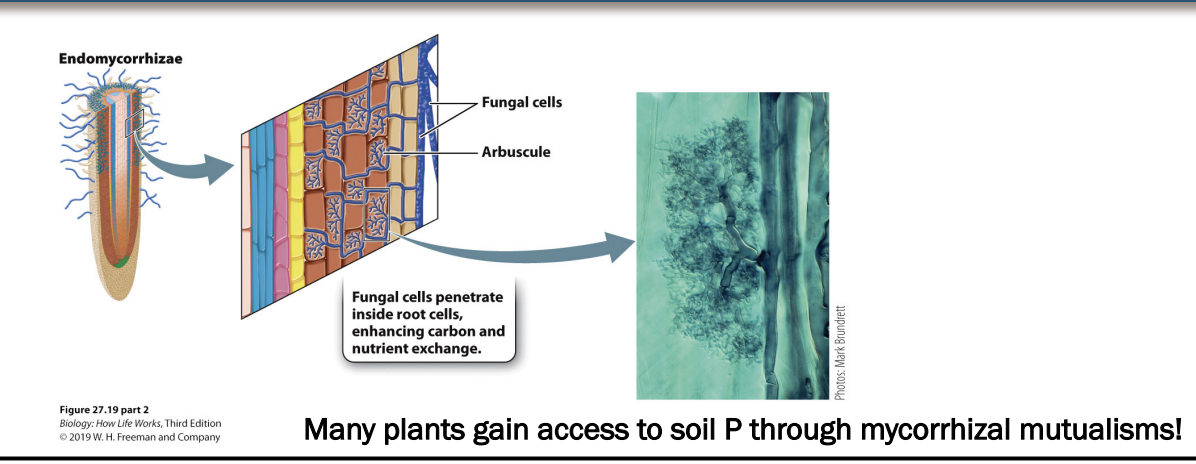
What are endomycorrhizae?
fungi that penetrate root cells, but do not form visible structures outside of root cells after this
can make them hard to study.
Instead, they form highly branched arbuscules that grow inside roots

D
Phto and respiration are the biggest carbon flux
Humans only 9