Tests for gasses
1/10
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
How to collect gas
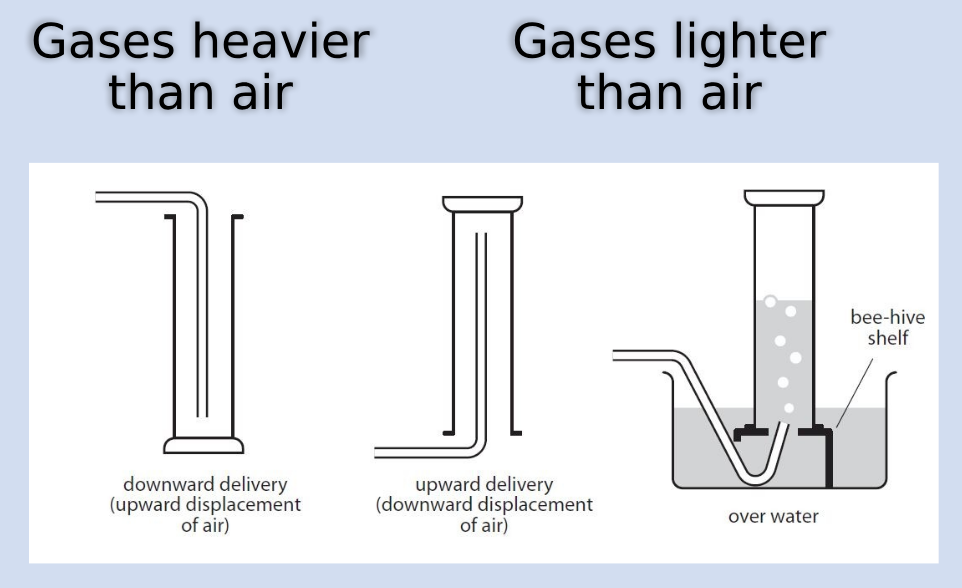
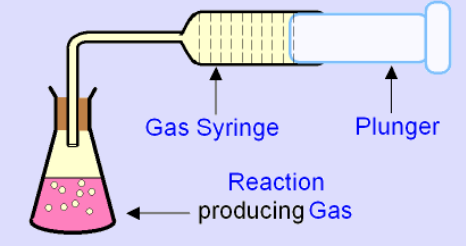
How to measure amount of gas produced
Use a gas syringe (this is the most efficient method)
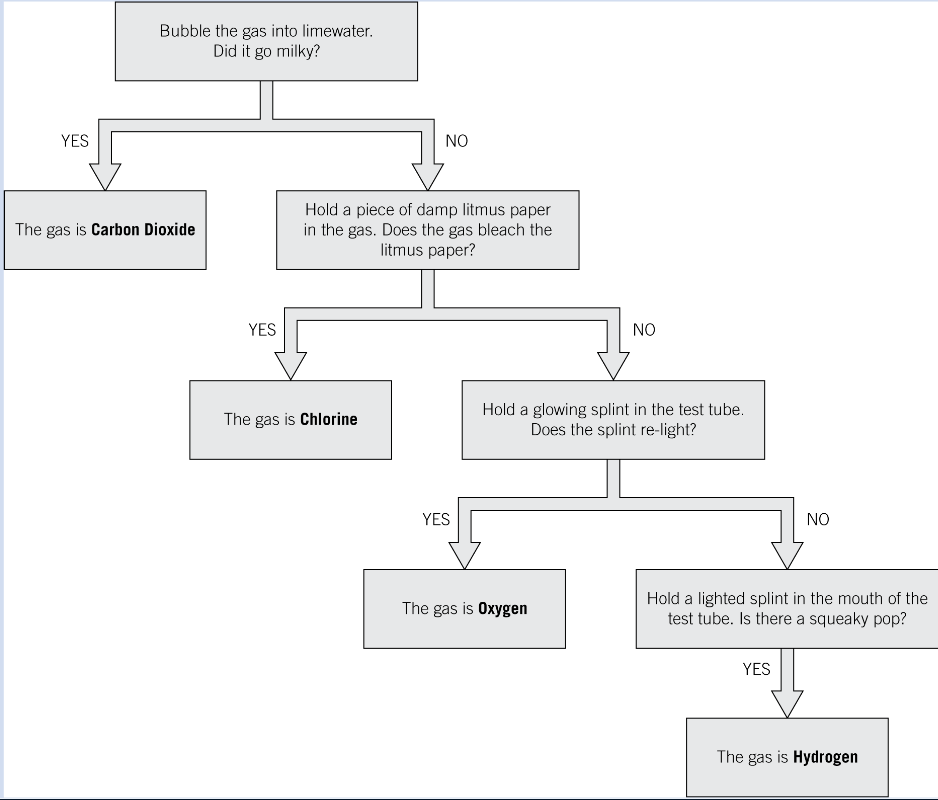
Test for Carbon Dioxide (CO₂)?
Add calcium carbonate to some hydrochloric acid and add in the substance you are testing
Attach a bung and delivery tube to the test tube and put the tube into a test tube of limewater, bubbles should start to come through the tube into the limewater
If CO₂ is present the limewater will turn milky/cloudy in colour
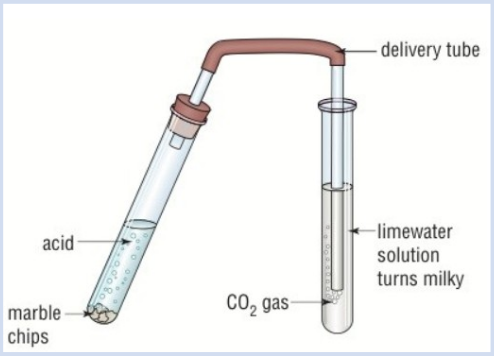
Why does limewater go cloudy in the presence of carbon dioxide?
Because a reaction is occuring

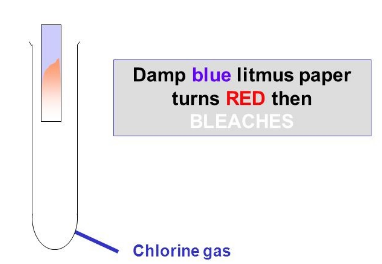
Test for chlorine
Dip damp blue litmus paper in the chlorine gas, it will turn red (because chlorine is acidic) then white (because the chlorine acts as a bleach)
The gas will also be a green-yellow colour and smell like a swimming pool, but the test above is used to confirm this
Test for oxygen
Fill a test tube with peroxide
Add yeast and wait for it to dissolve
Keep your thumb or a bung on the top to stop the gas from escaping
Add a glowing splint (not on fire but as if you blew out a candle and its the hot orang part left) into the test tube
If the splint re-lights then oxygen is present (because flames need oxygen)a
How to get hydrogen?
Mix a metal (e.g. magnesium) with an acid (e.g. hydrochloric acid) and collect the gas
because a metal + acid = Salt + water + Hydrogen Gas
Test for hydrogen
The squeaky pop test:
Fill a test tube with hydrochloric acid
Add magnesium and wait for it to dissolve
Add a light splint to the test tube
If you hear a squeaky pop, the hydrogen is present
Unknown gas, what order would you test it? (Bonus question)
Test for ammonia
Dip damp red litmus paper into the gas
If it turns blue then it is ammonia
Test for sulfer dioxide?
Add to acidified aqueous potassium manganate (VII)
It will turn from purple to colourless