C2.1 - Purity and separating mixtures
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
50 Terms
What is a pure substance? (chemistry definition)
A single element or compound with nothing else added — all particles are the same
What is a mixture?
Two or more substances physically combined but NOT chemically — can be separated by physical means
What are the melting/boiling points of a pure substance?
Sharp and specific — pure water boils at exactly 100°C and melts at exactly 0°C
What are the melting/boiling points of a mixture?
Occur over a range of temperatures — impurities lower the melting point and raise the boiling point
How can melting point data be used to assess purity?
Heat sample slowly using melting point apparatus → compare measured melting point to data table → closer to the known value = purer the sample
What does a cooling curve for a PURE substance look like?
Temperature drops, then stays FLAT (horizontal) at the melting point as it freezes — sharp melting point [DRAW: cooling curve with flat section]
![<p>Temperature drops, then stays FLAT (horizontal) at the melting point as it freezes — sharp melting point [DRAW: cooling curve with flat section]</p>](https://assets.knowt.com/user-attachments/996f62f7-60d8-4f12-be01-736bddfb49d8.png)
What does a cooling curve for an IMPURE substance look like?
Temperature drops gradually during freezing — NO flat section — melting occurs over a range (e.g. 40–50°C) [DRAW: cooling curve with gradual slope, no flat section]
![<p>Temperature drops gradually during freezing — NO flat section — melting occurs over a range (e.g. 40–50°C) [DRAW: cooling curve with gradual slope, no flat section]</p>](https://assets.knowt.com/user-attachments/988f80f9-3982-435f-b1e9-528f45e15b74.png)
What is the percentage change formula?
% change = ((final value − original value) ÷ original value) × 100
What is relative atomic mass (Ar)?
The average mass of the atoms of an element relative to 1/12 the mass of carbon-12 — it's the larger of the two numbers on the periodic table
What is relative formula mass (Mr)?
The total mass of a molecule/compound — calculated by adding up the Ar of all atoms in the formula
How do you calculate the Mr of a compound?
Identify all atoms in the formula → multiply each element's Ar by number of atoms → add them all together (e.g. H₂O: (2×1) + 16 = 18)
What does the Law of Conservation of Mass mean for Mr?
In a balanced equation, sum of Mr of reactants = sum of Mr of products
What is empirical formula?
The simplest whole number ratio of atoms of each element in a compound
What is molecular formula?
The actual number of atoms of each element in one molecule
How do you find empirical formula from molecular formula?
Divide all atom numbers by their highest common factor (e.g. C₆H₁₄ ÷ 2 = C₃H₇)
How do you find the empirical formula of an ionic compound from a lattice diagram?
Identify the ions → balance charges so overall charge = zero → ratio of ions is the empirical formula (e.g. Na⁺ and Cl⁻ → NaCl)
What is the percentage composition formula?
% mass of element = (Ar × number of those atoms ÷ Mr of compound) × 100
What is an alloy?
A mixture of metals (e.g. brass = 70% copper + 30% zinc)
Why are alloys harder than pure metals?
Atoms of different sizes distort the regular arrangement — prevents layers from sliding over each other easily
What is a formulation?
A mixture designed as a useful product, made by following an exact recipe with each component in carefully measured quantities
Give three examples of formulations
Paints, medicines, food products (also alloys)
What is filtration used to separate?
An INSOLUBLE solid from a liquid/solution (e.g. sand from water)
How does filtration work?
Pour mixture through filter paper in funnel → liquid (filtrate) passes through → solid (residue) is too large and stays on paper [DRAW: filtration setup diagram]
![<p>Pour mixture through filter paper in funnel → liquid (filtrate) passes through → solid (residue) is too large and stays on paper [DRAW: filtration setup diagram]</p>](https://assets.knowt.com/user-attachments/20e70232-22e5-4311-866b-f12fbb1351ff.png)
What is crystallisation used to separate?
A SOLUBLE solid from a solution (e.g. copper sulphate from water)
How does crystallisation work?
Heat solution to evaporate solvent → test with glass rod (crystals on rod = saturated) → leave to cool slowly → crystals form → filter → wash with cold distilled water → dry
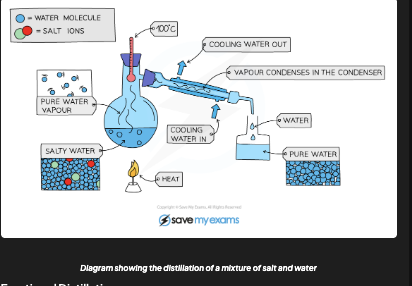
What is simple distillation used to separate?
A liquid/solvent from a solution (e.g. pure water from salt water)
How does simple distillation work?
Heat solution → solvent evaporates → vapour travels to condenser → cooled back to liquid → collected in beaker. Solute stays in flask
What is fractional distillation used to separate?
A mixture of MISCIBLE liquids with different boiling points (e.g. ethanol and water, crude oil, liquid air)
How does fractional distillation work?
Heat mixture → substance with lowest boiling point evaporates first → rises up fractionating column (glass beads increase surface area) → condenses and collected → temp increased to collect next fraction
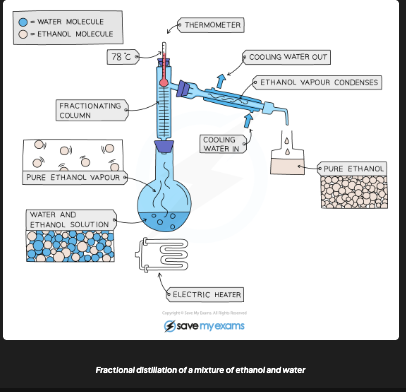
How is ethanol separated from water by fractional distillation?
Heat to 78°C → ethanol boils and distils out first → collected → stop at 78°C (water stays behind, bp = 100°C)
How do you choose the correct separation technique?
Insoluble solid + liquid → filtration. Soluble solid + liquid → crystallisation. Liquid from solution → simple distillation. Two miscible liquids → fractional distillation. Substances to identify → chromatography
What is chromatography used for?
Separating AND identifying components in a mixture of soluble substances
What are the two phases in ALL chromatography?
Mobile phase: moves and carries substances. Stationary phase: stays still, substances interact with it differently
What is the mobile and stationary phase in paper chromatography?
Mobile phase: solvent (e.g. water or ethanol). Stationary phase: the chromatography paper itself
How does paper chromatography work?
Draw pencil baseline → spot samples on line → place paper in solvent BELOW baseline → solvent travels up by capillary action → substances separate at different heights → mark solvent front → calculate Rf [DRAW: the 3-step chromatography setup diagram]
![<p>Draw pencil baseline → spot samples on line → place paper in solvent BELOW baseline → solvent travels up by capillary action → substances separate at different heights → mark solvent front → calculate Rf [DRAW: the 3-step chromatography setup diagram]</p>](https://assets.knowt.com/user-attachments/4a3d9828-d479-4a85-8d94-e38e8b75ef90.png)
Why must the baseline be drawn in pencil?
Ink would dissolve in the solvent and travel up, contaminating results
Why must the solvent be BELOW the baseline?
If solvent touches the spots they dissolve straight into it and wash away rather than separating
Why do substances travel different distances in chromatography?
Different solubilities in solvent and different affinities for stationary phase — more soluble in solvent = travels further (spends more time in mobile phase)
What is Thin Layer Chromatography (TLC)?
Works like paper chromatography but stationary phase is a thin layer of inert substance (e.g. silica) on a flat surface — mobile phase is still a solvent
What is the Rf value formula?
Rf = distance travelled by substance ÷ distance travelled by solvent front (both measured from baseline)
What is the range of Rf values and why?
Always between 0 and 1 — a substance can never travel further than the solvent front
How do you use Rf values to identify a substance?
Compare Rf to known reference values using the same solvent — matching Rf = same substance
How does a pure vs impure substance appear on a chromatogram?
Pure = one spot. Impure/mixture = multiple spots at different heights
What is gas chromatography used for?
Separating and identifying a mixture of gases or volatile substances
What are the mobile and stationary phases in gas chromatography?
Mobile phase: unreactive carrier gas (e.g. nitrogen). Stationary phase: thin layer of unreactive liquid (e.g. silica) inside a column
How does gas chromatography work?
Sample injected into column → carried by carrier gas → substances travel at different speeds based on attraction to stationary phase → each leaves column at different retention time → detector plots peaks on chromatogram [DRAW: GC apparatus diagram with column, detector, carrier gas]
![<p>Sample injected into column → carried by carrier gas → substances travel at different speeds based on attraction to stationary phase → each leaves column at different retention time → detector plots peaks on chromatogram [DRAW: GC apparatus diagram with column, detector, carrier gas]</p>](https://assets.knowt.com/user-attachments/fe32687a-9b9e-49be-a6f3-510994d2a0cb.png)
What is retention time in gas chromatography?
Time taken for a substance to travel through the column — longer retention time = stronger attraction to stationary phase
How do you interpret a gas chromatogram?
Number of peaks = number of compounds. Height of peak = amount present. Position of peak = retention time used to identify compound [DRAW: the GC chromatogram with peaks A, B, C, D]
How do you identify substances using gas chromatography?
Compare retention times to reference values for known substances run under the same conditions