Chemistry Exam 2 concepts
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
Ionic bonds
These bonds are formed between a metal and a nonmetal.
Covalent bonds
These bonds are formed between two nonmetals
Hydrogen (H2) and Oxygen (O2)
H2 is an explosive gas used as a fuel. O2 is also a gas, a natural component of the air on Earth. (O2 is ABSOLUTELY necessary for combustion/burning to occur. Both have an extremely low boiling points.
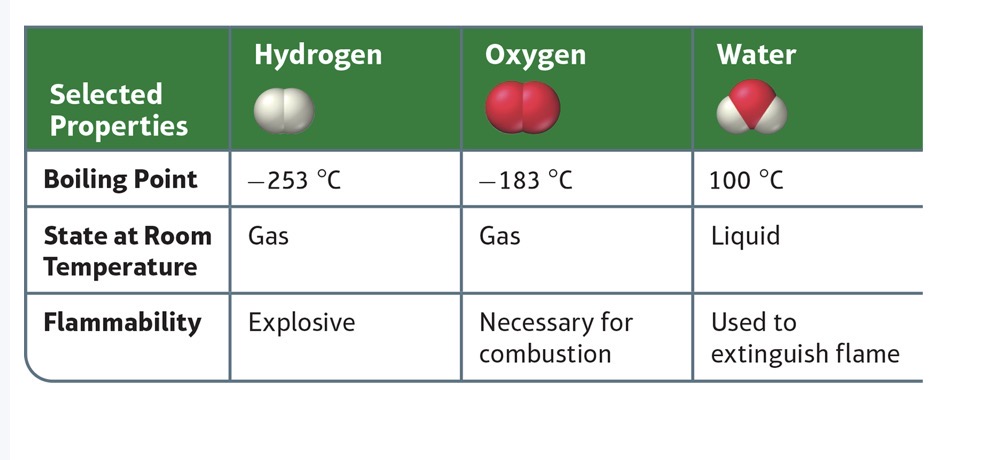
H2O (NOTHING like O2 and H2)
A liquid rather than a gas at room temperature.
It’s boiling points is degrees higher than hydrogen and oxygen.
Distinguishes flames
Key point of elements and compounds (4.1)
When two or more elements combine to form a compound, an entirely new substance results.
Compound vs. Mixtures
A mixture can mix in ANY proportions whatsoever. (Ex: a lot of hydrogen, a little bit of oxygen. Or vise versa) —→ any ratio of hydrogen to oxygen
In a compound, elements combine in fixed, definite proportions. (Ex: water is composed of water molecules that always contains TWO hydrogen atoms to every ONE oxygen atom. —→ definite proportion.)
Chemical bond
A chemical bond is the force that holds atoms together in a compound. These bonds form because they lower the potential energy of the charged particles that composed atoms.
Noble gases vs. Elements (4.2)
The rest of the elements do not possess the stability of the noble gases, the form chemical bonds to become more stable (to lower the PE of the charged particles that compose them).
More stable = Lower potential energy (PE)
Ionic Bond (More detailed)
The bond that forms between a metal and nonmetal.
metals have a tendency to lose electrons
Nonmetals have a tendency to gain them.
When a metal interacts with a nonmetal, it can TRANSFER one or more of its electrons.
Covalent bonds (More detailed)
Nonmetals tend to have high ionization energies (their electrons are relatively difficult to remove).
When a nonmetal bonds with another nonmetal, neither atom transfer electrons to the other. THEY SHARE!
Covalent Bonds and Molecular Compounds
Covalent bonded atoms form molecules, and the resulting compounds are called MOLECULAR COMPOUNDS.
molecular compound: a compound composed of two or more covalently bonded nonmetals. This is not an ionic compound!
Chemical Formulas
A chemical formula indicates the element present in the compound and the relative number of atoms or ions each.
An empirical formula indicates the relative number of atoms of each element in a compound. Ex: hydrogen peroxide is HO
A molecular formula indicates the actual number of atoms of each element in a molecule of a compound. Ex: hydrogen peroxide is H2O2.
Structural formula
A structural formula uses lines to represent covalent bonds and shows how atoms.
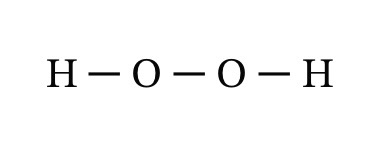
Lewis structures
In the Lewis model, we represent valence electrons as dots and use Lewis structures to depict molecules. This model helps predict whether a set of atoms will form a stable molecule and what that molecule might look like.
Octet
Eight valence electrons, signifying a filled outer electron shell for s and p elements
Lewis structure of Helium
Helium is an exception. Its electron configuration and Lewis symbol contains only two dots.
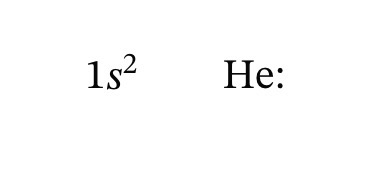
Ionic compounds
Composed of cations (metal) and anions (nonmetals) bound together by ionic bonds. The basic unit of an ionic compound is the formula unit.
Formula Unit (NOT A MOLECULE)
The smallest, electrically neutral collection of ions in an ionic compound.
Rules for ionic bonding (Lewis Model)
To represent ionic bonding, we move electron dots from the Lewis symbol of the metal to the Lewis symbol of the nonmetal, so the metal becomes a cation and the nonmetal becomes an anion.
Crystalline lattice
a result of the attractions between the cations and anions.
Lattice Energy
The energy change (released) upon formation of one mole of a crystalline compound (ionic). OR textbook definition: the energy associated with the formation of a crystalline lattice of alternating cations and anions from gaseous ions.
ionic compounds usually give off quite a bit of energy as heat.
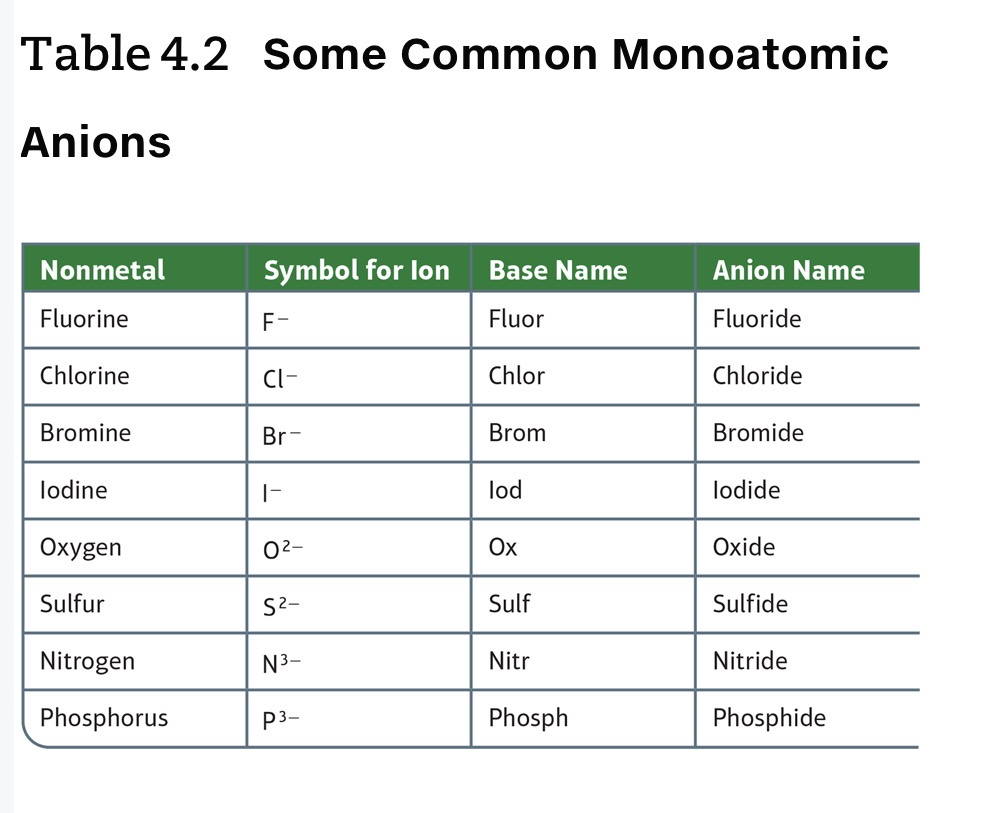
Naming Ionic Compounds
Some ionic compounds have common names and systematic names for different types of compounds, including ionic ones. Even if you are not familiar with a compound, you can determine its systematic name from its chemical formula.
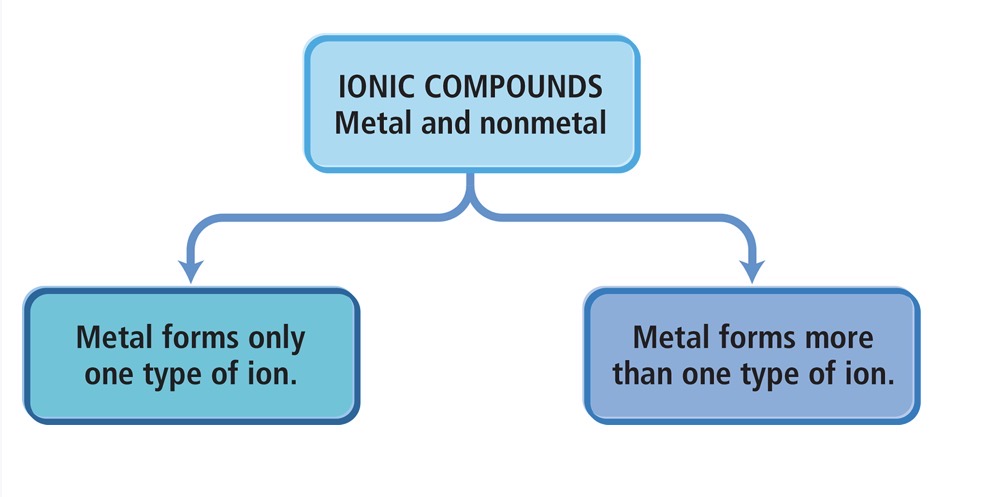
Two types of ionic compounds
Type 1: Metal forms only one type of ion. The metal forms an ion that always has the same charge. (we don’t specify the charge in the name of the compound)
Ex: Sodium → always Na⁺
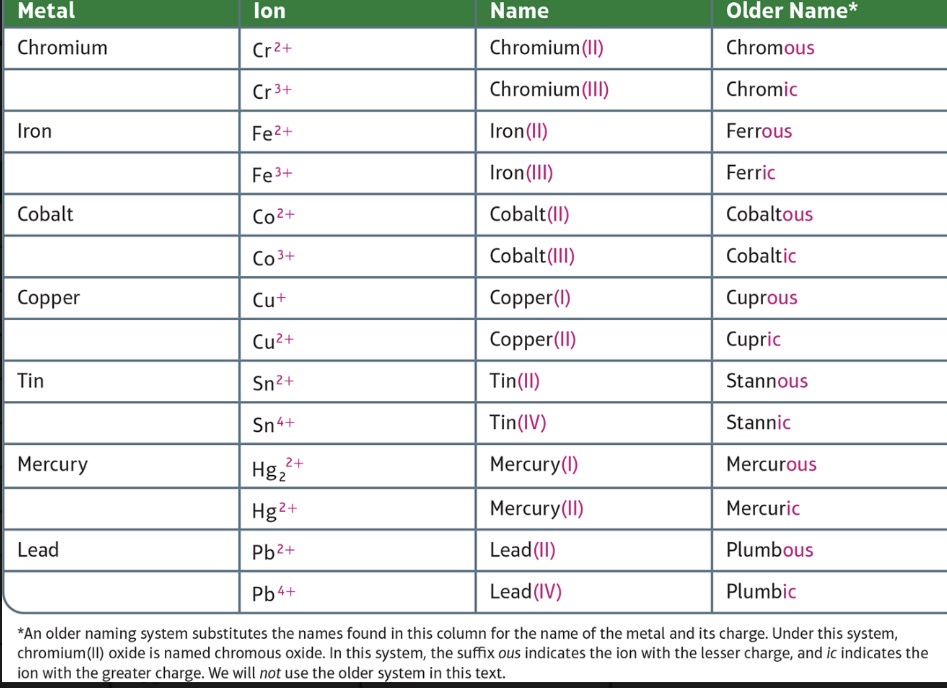
Metal forms more than one type of ion
First type: Metals Whose Charge Is Invariant from One Compound to Another

Second Type: Metal ion with a charge that can differ in different compounds
Usually transition metals. Some are main-group metals such as Pb and Sn.
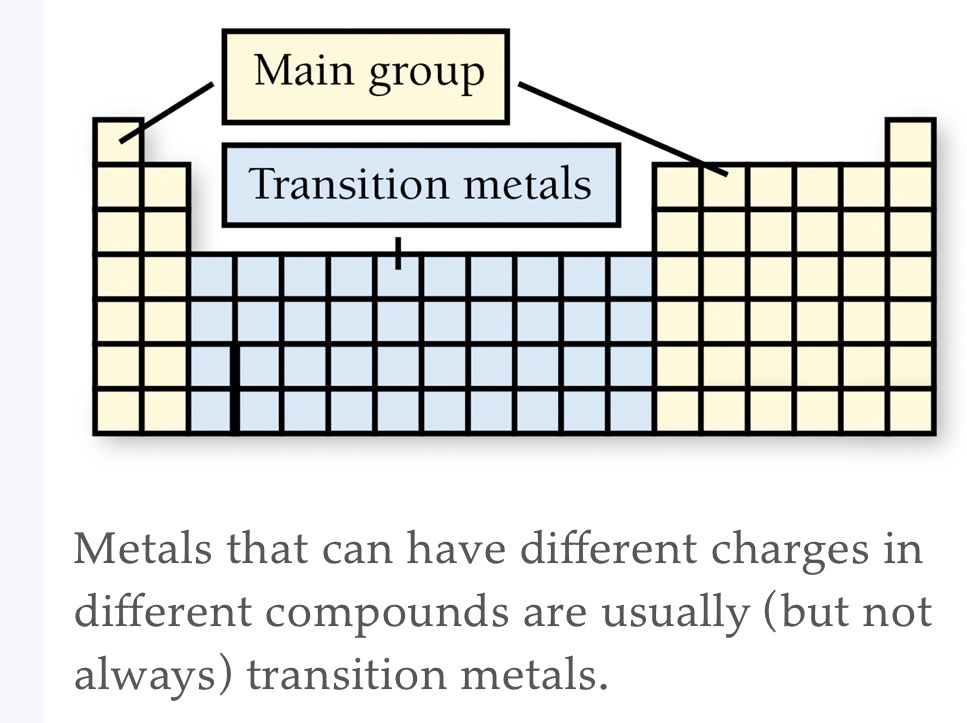
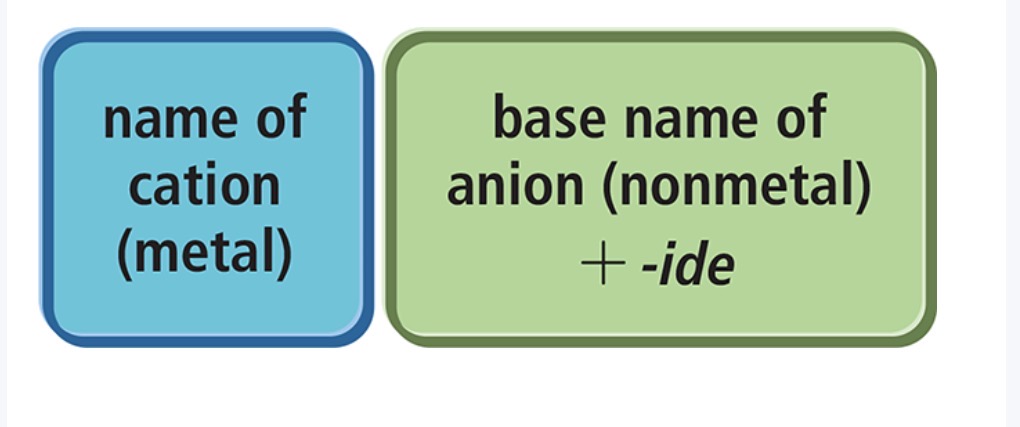
Naming Binary Ionic Compounds: Containg a Metal That Forms Only One Type of Cation (Monatomic atoms)

Naming Binary lonic Compounds Containing a Metal That Forms More Than One Type of Cation
Some metals that form cations with different charges also have an older name.
Common Polyatomic Ions
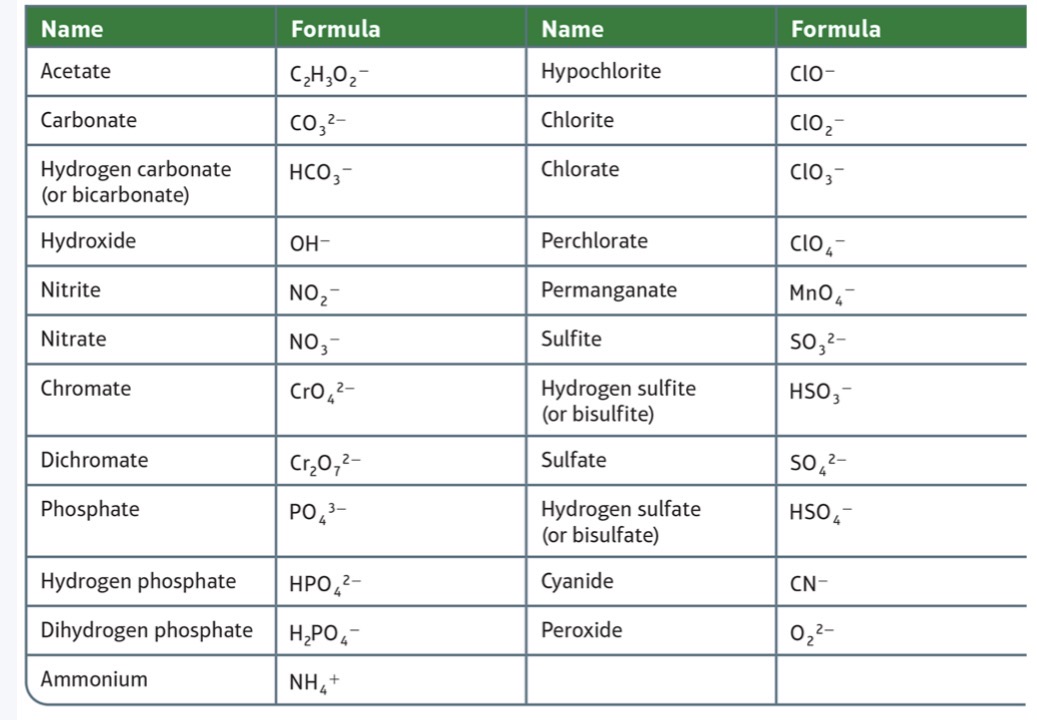
Acetate
C2H3O2^-
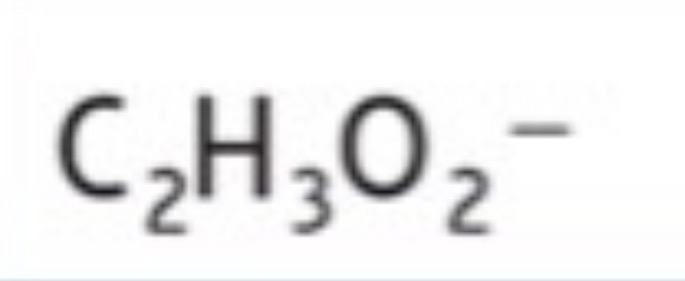
Carbonate
CO3^-2

Hydrogen Carbonate or Bicarbonate
HCO3^-
Hydroxide
OH^-

Nitrite
NO2^-
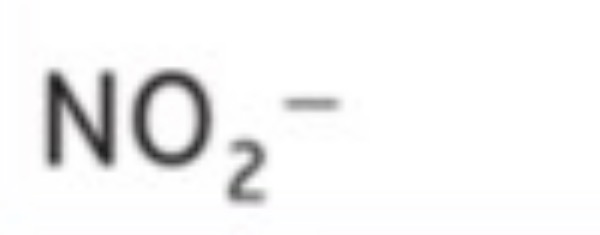
Nitrate
NO3^-

Phosphate
PO4^-3

Hydrogen Phosphate
HPO4^-2
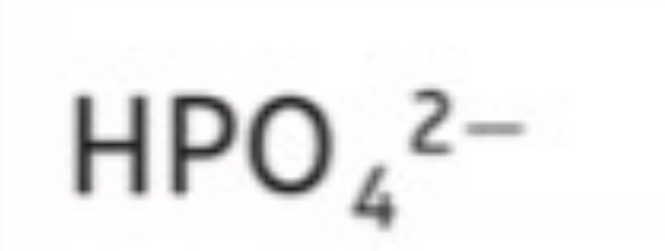
Dihydrogen phosphate (All include O4)
H2PO4^-
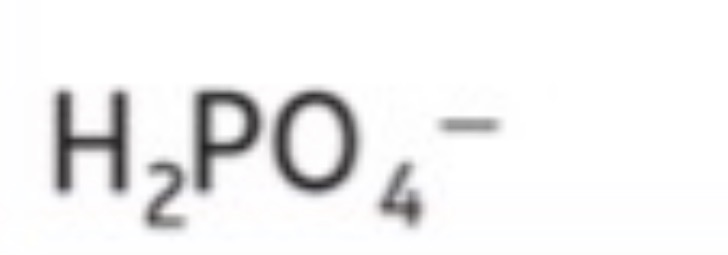
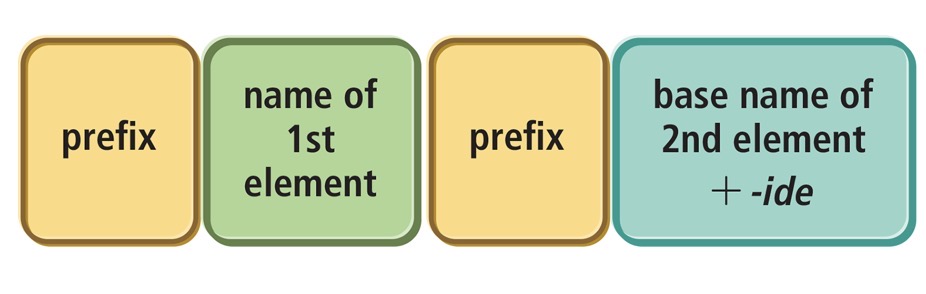
Naming Molecular Compounds (covalent bonds —> nonmetal/nonmetal)
molecular compounds are composed of two or more nonmetals. Molecular compounds use prefixes unlike ionic compounds.
Rules/Steps
Write the element with the SMALLEST group number first.
If both are in the same group then write the one with the larger period number first. (Groups are vertical while periods are horizontal)
If there is only one atom of the first elementin the formula, we normally omit the prefix mono-. For example, we name NO2 according to the first element, nitrogen, with no prefix because mono- is omitted for the first element, followed by the prefix di-, to indicate two oxygen atoms, and the base name of the second element, ox, with the ending ide. Its full name is nitrogen dioxide.
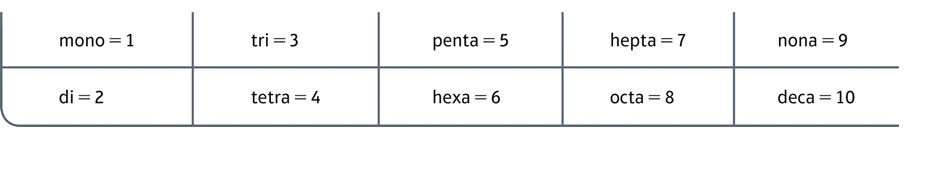
Prefixes
Bonding pair & Lone pair (4.7)
A shared pair of electrons is called a bonding pair. A pair associated with only one atom is called a lone pair(nonbonding electron)
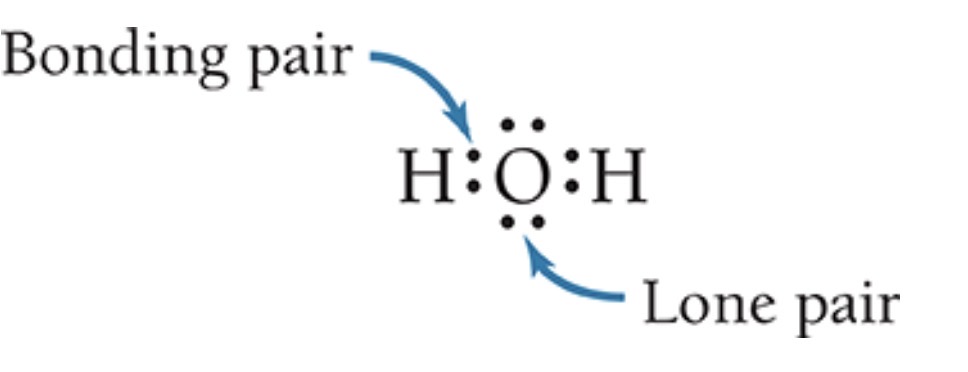
Importance for Lewis structure
The Lewis model account got why covalent bonds are HIGHLY DIRECTIONAL. (Atoms line up a certain way).
for ionic bonds, they are NONDIRECTIONAL and hold together an array of ions.
Intermolecular forces
The interactions between molecules. Weaker than the bonding interactions within a molecule (intra)
Intramolecular forces
Within a molecule.
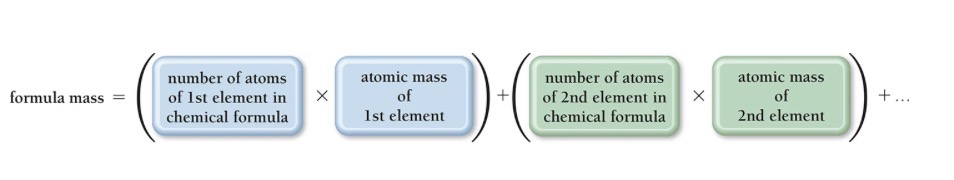
Atomic mass and formula mass
We defined the average mass of an atom of an element as its atomic mass. Similarly, we now define the average mass of a molecule (or a formula unit) of a compound as its formula mass.
Synonymous terms for formula mass
Molecular mass/molecular weight
Formula Mass Calculation
For any compound, the formula mass is the sum of the atomic masses of ALL the atoms in its chemical formula.
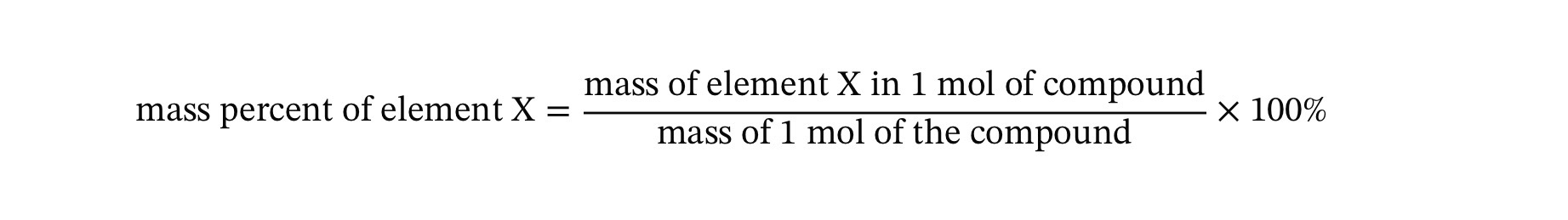
Mass percent or mass percent composition (4.10)
Tells you that element’s percentage of the compounds total mass. (How much of the compound’s total mass comes from that one element.)
Mass percent helps us answer:
“Out of 100 grams of this compound, how many grams are in this element?”
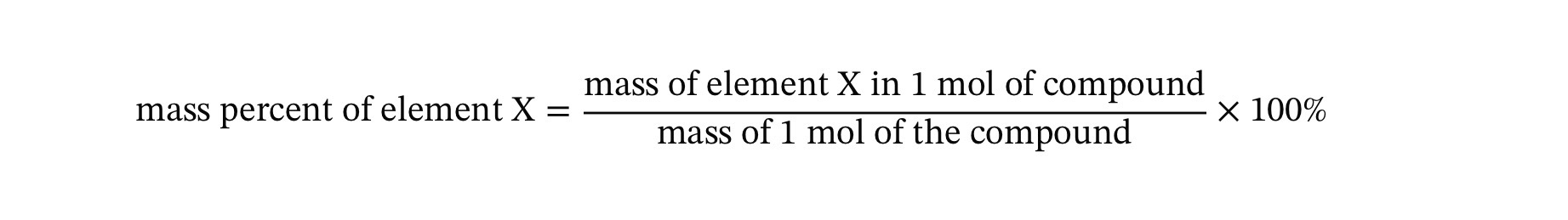
The mass percent composition of an element in a compound is a conversion factor between the mass of the element and the mass of the compound.
Ex: Cl Mass percent in CCl2F2 is 58.64%
Remember: 58.64% means per hundred soo in terms of mass it is 58.64 grams Cl per hundred grams CCl2F2

Conversion Factors from Chemical Formulas
Different way to approach mass percent composition (through mol unit). This also tells us the amount of constituent elements present.
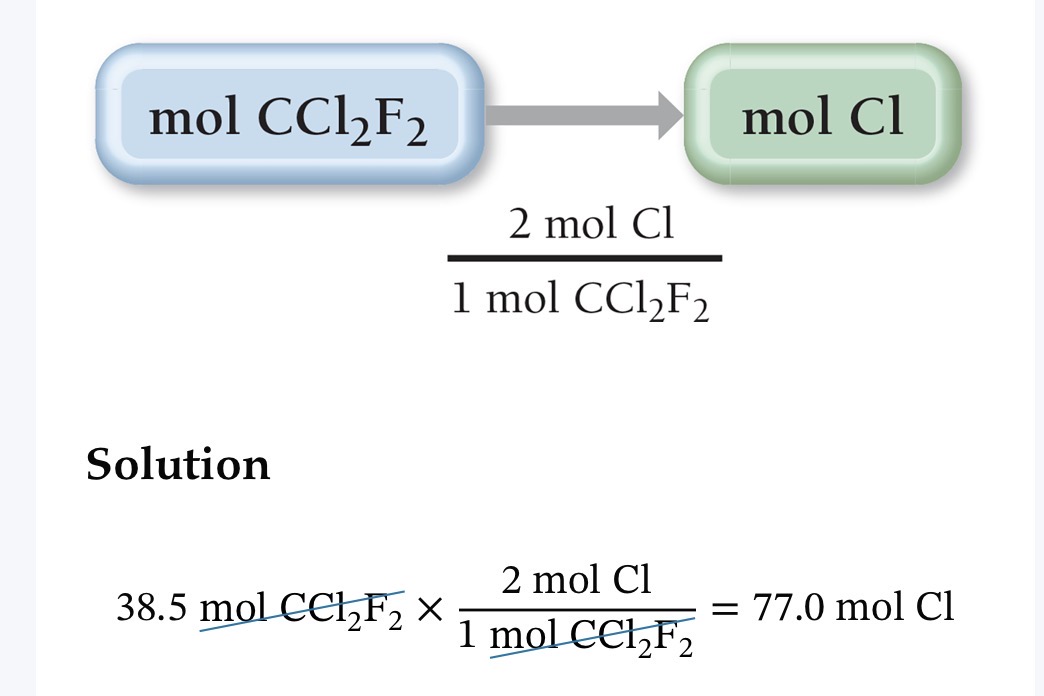
Empirical Formula subscripts

Molar mass (4.11)
Molar mass = empirical formula molar mass x n
n = molar mass / empirical formula
Combustion Analysis (4.11)
Organic compounds (4.12)
Organic compounds are composed of carbon and hydrogen and a few other elements, including nitrogen, oxygen, and sulfur. The key element in organic chemistry, however, is carbon.
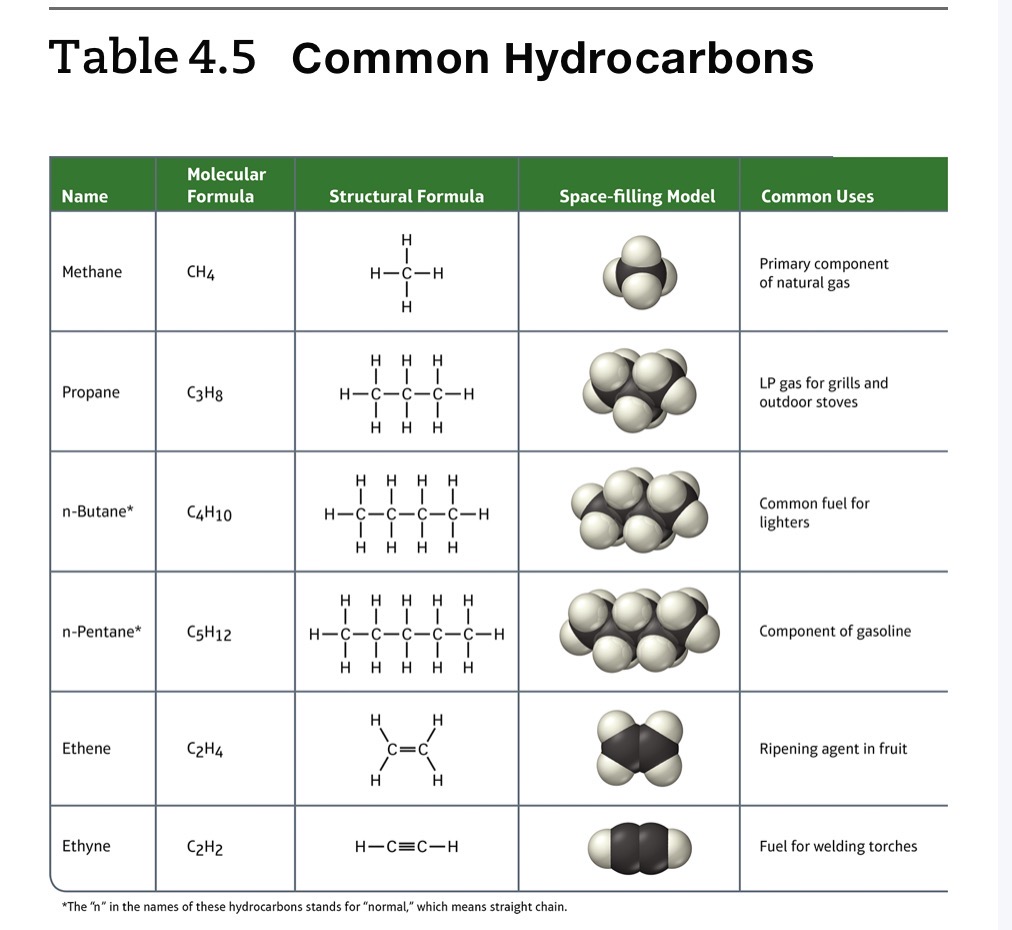
Hydrocarbons
Morphine (5.1)
Morphine can help provide relief from pain. It also is a natural product derived from the sap of the opium poppy.
Polar covalent bond
Is intermediate in nature between a pure covalent bond and an ionic bond. It has a negative pole and a positive pole. It is unequally shared. (NOT IONIC BECAUSE IT DOES NOT INVOLVE TRANSFERS)
Electronegativity
The ability of an atom to attract electrons to ITSELF in a chemical bond (which RESULTS IN POLAR AND IONIC BONDS)
Ex: fluorine is more electronegative than hydrogen
Bond polarity
The degree of polarity in a chemical bond depends on the electronegative difference (sometimes abbreviated as delta EN) between two bonding atoms. THE GREATER the electronegative difference, the more polar the bond.
Nonpolar covalent bond (5.2)
Same electronegativity, electrons equally shared
Large electronegativity
If there is a large electronegativity difference between the two atoms in a one, such as normally occurs between a metal and a nonmetal, the electron from the metal is almost completely transferred to the non metal. The bond is ionic
Intermediate electronegativity
Electronegative difference between two different nonmetal is a polar covalent bond.
Molecular Geometry
This is used to predict the molecular geometry of elements.
Electron geometry is mainly the same but molecular geometry changes
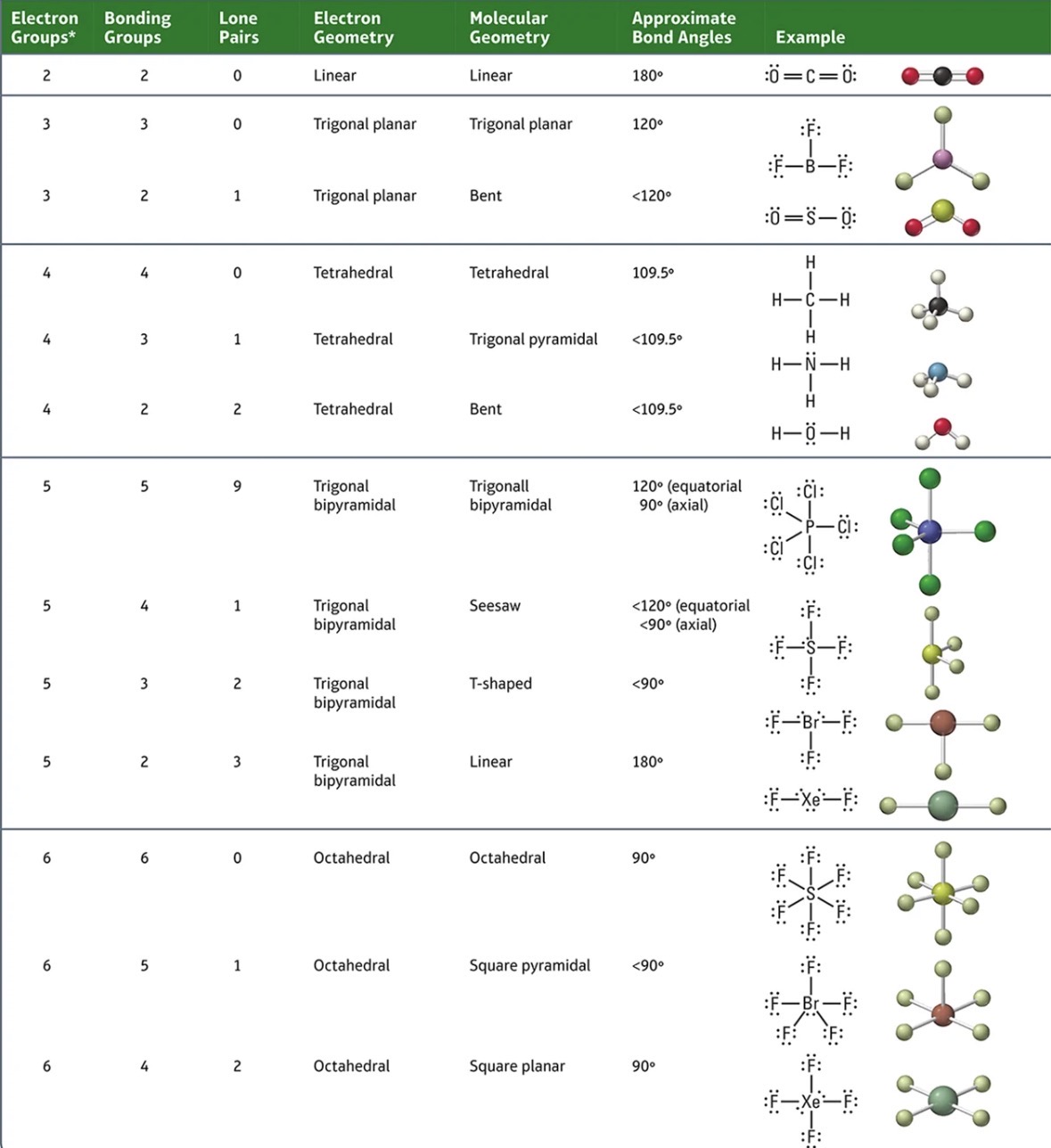
Linear (Strictly one form)
• 2 Electron groups
• 2 Bonding Groups
• Bond angle: 180•

Trigonal Planar (1)
Trigonal Planar and Bent (2)
Tetrahedral (1)
Trigonal Planar