particle model of matter
1/32
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
33 Terms
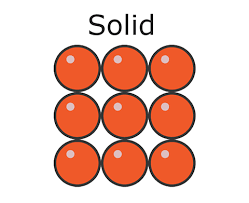
solids
In solids, strong forces of attraction hold the particles close together in a fixed regular arrangement. The particles dont have much energy so they can only vibrate in their fixed positions.
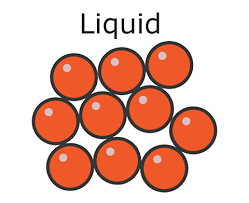
liquids
There are weaker forces of attraction between the particles in liquids. The particles are close together, but can move past each other and form irregular arrangements. They have more energy that particles in a solid and they move in random directions at low speeds
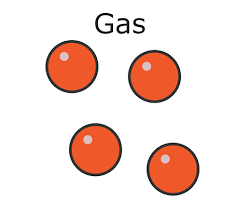
gases
there are almost no forces of attraction between particles in a gas. The particles have more energy than those in liquids and solids and they are free to move and travel in random directions at high speeds.
properties of solids liquids and gases
the density of a substance is generally highest when it is in solid form as the particles are closest together. Liquids tend to be less dense than solids. Gases are the least dense as their particles are spaced far apart
gases and liquids can flow because their particles can move past each other.
Gases are compressible. The particles in a gas are very spread out which means you can squash a gas into a smaller volume, reducing the distance between the particles. The particles in solids and liquids cant really be compressed as their particles are already quite close together
density
the mass per unit of volume
unit of density is kg/m³ or g/cm³
density = mass/volume
density id a property of a material
objects denser than water will sink
objects less dense than water will float
differences in density
Solids (Highest Density): Particles are packed very closely together in a fixed, rigid structure, resulting in high mass per volume.
Liquids (Moderate/High Density): Particles are close together but free to move. They are usually slightly less dense than their solid state, allowing solid metal to sink in liquid metal.
Gases (Lowest Density): Particles are very far apart and move freely, resulting in low mass per volume
exception to density rule
Water: Unlike most substances, water’s density decreases when it freezes. Because its particles spread out in a solid state, ice is less dense than liquid water, allowing it to float
how does temp effect density
Temperature Effects: Increasing the temperature of a substance generally decreases its density because particles move faster and spread apart, increasing the volume.
measuring density of a solid object
First use a balance to measure the objects mass. If its a regular solid like a cuboid, you might be able to measure its dimensions with a ruler and calculate the volume. For an irregular solid, you can find its volume by submerging it in a eureka can of water
a eureka can (or displacement can) is essentially a beaker with a spout. To use one fill it up with water so the water level is just below the start of the spout
place an empty measuring cylinder below the end of the spout. When you put your solid object in, it causes the water level to rise and some will fall out the spout
measure the volume of water that left the can and that is the volume of your irregular solid object
then put it into the formula to find the density
measuring density of a liquid
place a measuring cylinder on a balance and zero the balance
pour 10ml of water into the cylinder and record the total measure and mass. Repeat this process until the cylinder is full
put your results into the density formula and find an avg.
changes of state
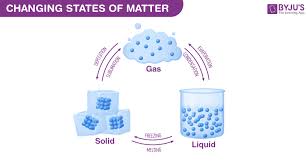
deposition
gas to solid
sublimation
solid to gas
why is a change of state different to a chemical change.
A chemical change creates entirely new substances with different properties through the rearrangement of atoms, and it is typically irreversible
A change of state is a reversible physical process where matter transitions between solid, liquid, or gas phases due to heating or cooling. Particles gain or lose energy, breaking or forming intermolecular bonds, altering their arrangement and freedom of movement. While density changes, mass remains constant.
kinetic energy
due to movement of particles
potential energy
energy between particles
internal energy
defenition - the total amount of kinetic energy and potential energy of all the particles (atoms and molecules) within a system (KE + potential energy = internal energy)
The particles in a system vibrate or move around - they have energy in their KE stores
The energy in systems is stored by its particles (atoms and molecule). The internal energy of a system is the total energy that its particles have in their kinetic and potential energy stores
Heating the system transfers energy to its particles (they gain energy in their kinetic energy stores and move faster, increasing the internal energy. This leads to a change in temp (or state).
The size of the temp change depends on the size of mass of the substance, what its made of (SHC) and the energy input
If the substance is heated enough, the particles will have enough energy in their ke stores to break bonds holding the them together. This means a change if state.
__________________________________
→→→ increasing internal energy
SOLID LIQUID GAS
decreasing internal energy← ← ←
_________________________________
breaking bonds
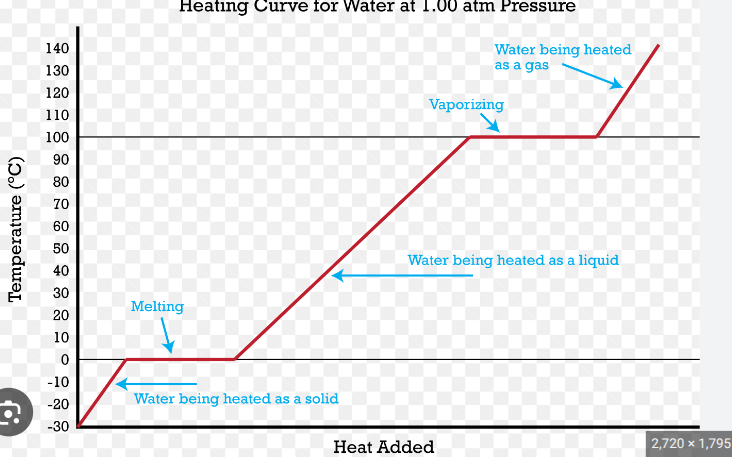
When a substance is melting or boiling, you’re still putting in energy and so increasing the internal energy, but the energy’s used for breaking intermolecular bonds rather than raising the temp.
when a substance is condensing or freezing, bonds are forming between particles, which releases energy. This means the internal energy decreases, but the temp dosent go down until all the substance has turned to liquid(condesing) or solid (freezing)
On the heating graph, potential energy is being used to break bonds on a horizontal line, and KE is being used to heat up the substance on the diagonal.
specific latent heat
definition - the amount of energy required to change the state of 1kg of a material at either its melting point or boiling point.
latent heat of vapourisation - liquid to gas (boiling p)(+condensing)
latent heat of fusion - solid to liquid (melting p)(+freezing)
SLH equations
energy = mass x latent heat
what happens when we heat up a material (SLH)
heat up the material
temp rises
particles have more energy
move around/vibrate more
when the temp reaches melting point
the particles have enough energy to start breaking away and becoming a liquid
the heat energy is used to break bonds
internal energy keeps rising when it melts
but the temp stays constant
what happens to the energy transferred to a substance as it changes state.
When energy is transferred to a substance during a change of state (melting or boiling), it does not increase the temperature. Instead, the energy breaks the intermolecular forces holding the particles together, increasing their potential energy to allow the substance to change state, such as turning from solid to liquid or liquid to gas
what is the difference between specific latent heat and specific heat capacity
Specific Heat Capacity
What it is: The amount of heat needed to raise the temperature of 1 kilogram of a substance by 1°C.
When it’s used: When a substance is heated and its temperature increases (like warming water).
Specific Latent Heat
What it is: The amount of heat needed to change the state of 1 kilogram of a substance without changing its temperature.
When it’s used: During phase changes, like melting ice into water or boiling water into steam, where the temperature stays the same.
what is the motion of molecules of gas
constant, rapid, and random motion, moving in straight lines until they collide with each other or the container walls
temp of gases
if you increase the temp of the gas, the avg speed of its particles increases. This is because the energy is in the particles kinetic stores
heat vs temp
temp is how hot or cold something is, measured in *c
heat is the amount of kinetic energy something has, measured in joules
gas pressure
Gas pressure and temperature are directly proportional - if we heat up a fixed volume of gas, the particles gain energy and move more quickly. This means they hit the walls of their container more frequently, with more force, increasing the pressure.
gas pressure and volume are indirectly proportional - if we decrease volume of container of gas, the particles have less distance to travel between collisions. This means they hit the walls of their container more frequently increasing pressure.
why does collisions with the wall make gas pressure increase
as gas particles move about at high speeds, they bang into eachother and whatever else happens to in the way. When they collide with a surface, they exert a force on it. Since pressure is force per unit area, this means they exert pressure to. In a sealed container, the outward gas pressure is the total force exerted by all the particles in the gas on a unit area of the container walls.
the direction of the net force on the walls of the gas container (or any surface).
yup
gas pressure equation
pressure = force / area
work is the transfer of energy by a force.
yep
what is the effect of doing work on a gas on the internal energy and temperature of the gas
increases both its internal energy and its temperature.
explain how doing work on an enclosed gas leads to an increase in the temperature of the gas.
Because temperature is directly related to the average kinetic energy of these particles, this increased energy leads to a rise in temperature.