DAT Bootcamp: General Chemistry
1/379
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
380 Terms
How to fill electron orbitals?
based on the s, p, d, and f blocks
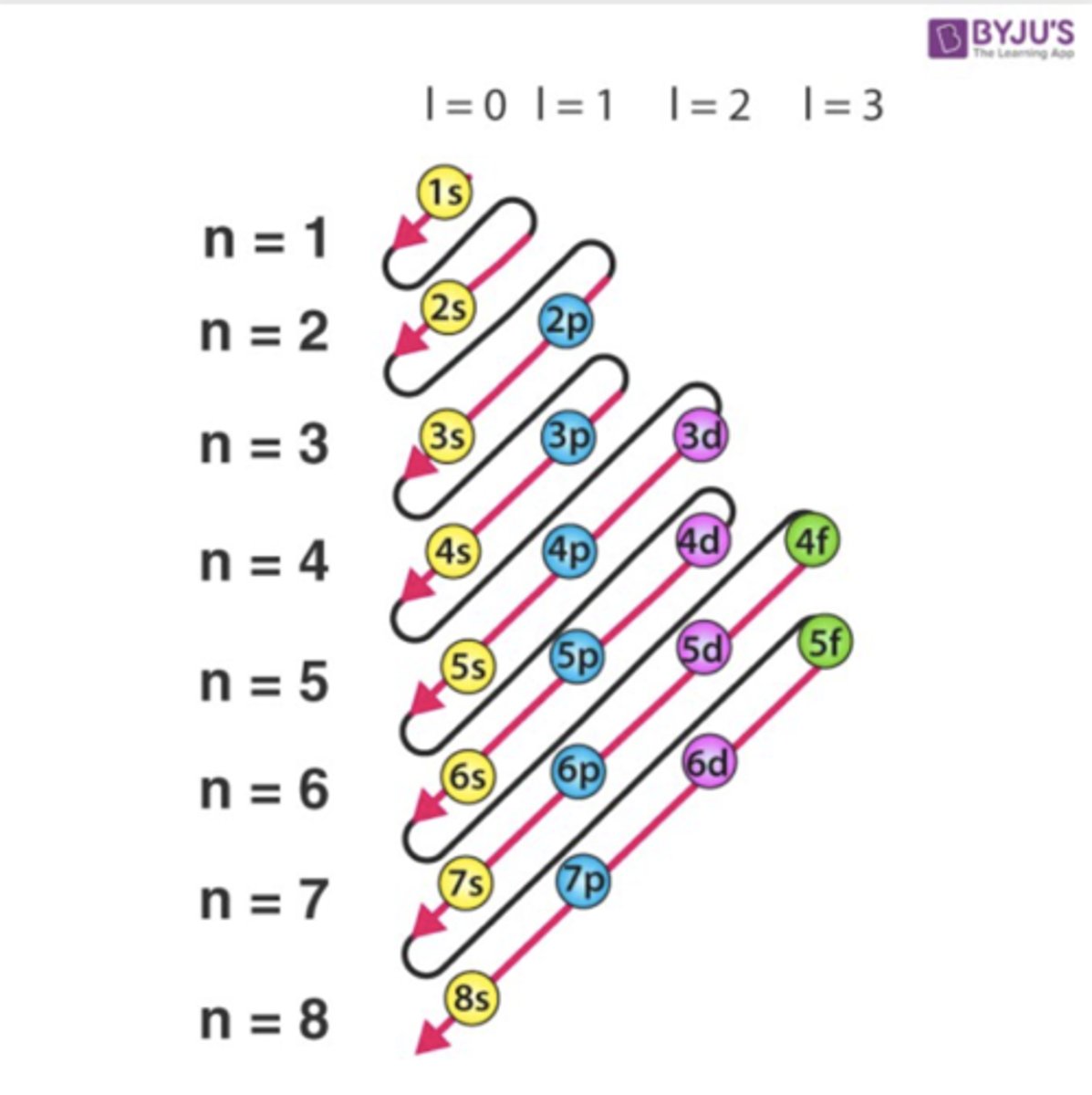
Hund's Rule
states that single electrons with the same spin must occupy each equal-energy orbital before additional electrons with opposite spins can occupy the same orbitals
Aufbau Principle
An electron occupies the lowest-energy orbital that can receive it
The electrons whose orbitals are __________________ away from the nucleus (those with the largest principle quantum number, n) are the atom's valence electrons.
furthest
The ______________ electrons are the highest energy elections and the ones that play a role in chemical reactions.
valence
For elements in the _____ block, the ___ shell electrons are also valence electrons.
d
The higher an electron's principle quantum number (n)...
- the further the distance from the nucleus
- the higher the energy
- the higher the reactivity
What are the five exceptions in electron configuration?
Chromium [Ar] 4s¹ 3d⁵
Molybdenum [Kr] 5s¹ 4d⁵
Cu [Ar] 4s¹ 3d10
Ag [Kr] 5s¹ 4d10
Au [Xe] 6s¹ 4f14 5d10
Cr & Mo move an electron down in order to...
get their electrons unpaired
Cu, Ag, Au move an electron down in order to...
to have it in a lower energy state and be more stable
Paramagnetic
have an unpaired electron
Paramagnetic substances are ______________ to magnets.
attracted
Diamagnetic
all electrons are paired
Diamagnetic substances are slightly ___________ by magnets.
repelled
If an element has an _______ number of electrons then it is paramagnetic.
ODD
If an element has an _________ number of electrons, then it is paramagnetic OR diamagnetic. You have to use the energy diagram to figure it out.
EVEN
Energy of a Photon
hf = hc÷λ
The energy of a photon is directly related to its ______________.
frequency
The energy of a photon is inversely proportional to its ____________.
wavelength
As the energy of a photon increases, the frequency will ________________ and the wavelength will ________________.
increase; decrease
Frequency =
1/λ
N₂ is
diamagnetic
O₂ is
paramagnetic
Octet Rule
refers to the tendency of atoms to prefer to have eight electrons in the valence shell, so that they can behave like noble gases
When atoms gain an electron they become ___________ charged.
negatively
When atoms lose an electron, they become ___________ charged.
positively
Compounds
are molecules comprised of two or more different elements bonded together
Metals v. Nonmetals
helps to determine if an ionic or a molecular compound is formed
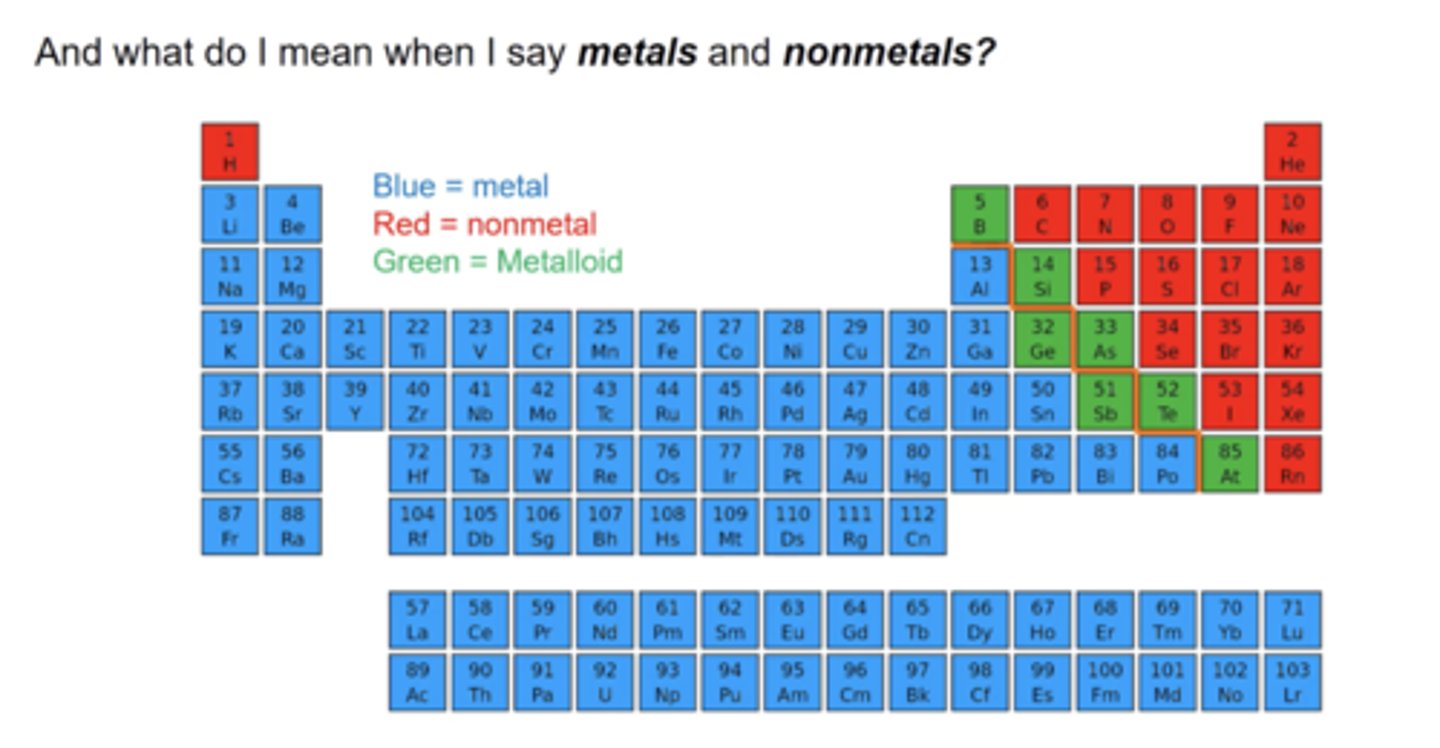
Ionic Compounds
metal + non-metal --> through ionic bonds
(exceptions being the polyatomic ions)
Molecular Compounds
non-metal + non-metal --> through covalent bonding
_____________ determines if the sharing between electrons is equal or unequal.
Electronegativity
___________ Covalent Bonds are molecular compounds with an uneven sharing of electrons caused by a significant different in electronegativity.
Polar
They have partial positive or negative charges
______________ Covalent Bonds occur when nonmetal atoms do not have a significant electronegativity difference between them.
Nonpolar
They do not have partial charges.
Lattice Energy
the energy required to completely separate an ionic compound's cations from its anions

The larger the charges =
the larger the lattice energy
The shorter the bond distance =
the larger the lattice energy
Hydrogen only wants ______ electrons.
2
Beryllium only wants ______ electrons.
4
Boron and Aluminum only want ______ electrons.
6
_________ row and lower can have more than 8 electrons if needed.
Third
Each triple bond is one _____ and two _____ bonds.
σ; π
Each single bond is one _____ bond.
σ
Each double bond is one _____ and one ________ bond.
σ; π
Formal Charge =
(valence electrons) - (one for each dot) - (one for each bond)
A __________ is a column and elements within here have similar properties.
group
A ______________ is a row and elements within here do not necessarily have similar properties.
period
Group 1 = ________ Metals
Alkali
low ionization energies because they want to lose an electron to feel like a noble gas; it is a lot easier for them to shift to the right than the left
- explosive with water
- form ionic compounds
What is the common formula of alkali metals?
M(s) + H₂O(l) --> MOH (aq) + 1/2H₂(g)
- extremely exothermic
Group 2 = ___________ ____________ Metals
Alkaline Earth Metals
- low ionization energies (but not as low as gorup 1)
- Do react with H₂O, but not as violently as group 1
- Generally get more reactive as you go down the group.
Group 7A/17 = ______________
Halogens
- High electronegativities/electron affinities
- They are easily reduced and want to have an extra electron
- Highly reactive with metals
- They are only one valence electron away from full octet
Group 8A/18 = ______________
Noble Gases
- they are inert
D block = _____________ Metals
Transition
- often form brightly colored compounds due to the way that d-orbitals allow electrons to absorb colored light and be promoted to higher energy levels
- multiple oxidation states (ie: iron)
Group 6A = ____________ Group or the Chalcogens
- two forms of molecular oxygen O₂ and O₃.
- O₂ is a good oxidizing agent (oxidant), but O₃ is an even better oxidizing agent
- Metals like to react with oxgen to form metal oxides
Atomic Radius
the distance from its nucleus to the outside of its outermost orbitals
Bond Length =
atomic radius of first element + atomic radius of second element
An increase in the number of energy shells (n) -->
an increase in size
As the effective nuclear charge increases --->
the size of the element decreases
When dealing with atoms of the exact same element, if you give an atom an electron (create an anion) the atom _________ in size.
increases
When dealing with atoms of the exact same element, if you take away an electron from an atom (create an cation) the atom _________ in size.
decreases
Isoelectronic Series
if you have a group of atoms or ions that are from different elements but have the same total number of electrons
Ionization Energy
the amount of energy required to strip an electron from an atom
Why is ionization energy opposite of atomic radius?
The smaller the atom, the closer the electrons are to the nucleus meaning they are being pulled very closely. The larger an atom, the further away electrons are from the nucleus, making it easier to pull them away.
Electron Affinity
the energy produced when an atom accepts an electron; the opposite of ionization energy
Unlike electronegativity, _____________ not fluorine has the highest electron affinity.
chlorine
The __________ gases are excluded from the electron affinity trends.
noble
Be electron affinity is around _______.
0; it has a completely filled 2s subshell
N electron affinity is around _______.
0; it has a half filled 2p subshell
What is the formula for Kelvin?
K = °C + 273
Volume is measured in __________.
liters
1 cm³ =
1 mL = 1cc
What is the formula for Pressure?
P = Force/Area
1 atm =
760 torr = 760 mmHg
Ideal Gas
a theoretical gas whose behavior can be predicted by the ideal gas law.
Ideal Gas Assumptions
1. volume or size is insignificant
2. collisions are elastic
3. average kinetic energy depends only on pressure
Gases behave most ideally at ________ temperatures and ___________ pressures.
high; low
Boyle's Law:
volume is inversely related to pressure
Charle's Law:
volume is directly related to temperature
Avogadro's Law:
volume is directly related to the number of gas molecules
Combined Gas Law:
P₁V₁/nRT₁=P₂V₂/nRT₂
Ideal Gas Law:
enables use to calculate one that we don't know when dealing with PV=nRT
Gas Density =
P(MM)/RT
Dalton's Law of Partial Pressure =
Ptotal = Pa + Pb ...
How to determine one of the partial pressures?
Pa = Xa × Ptotal
Types of Intermolecular Forces:
1. Hydrogen Bonding (strongest)
2. Dipole - Dipole
3. London Dispersion Forces (hydrocarbons, other nonpolar molecules)
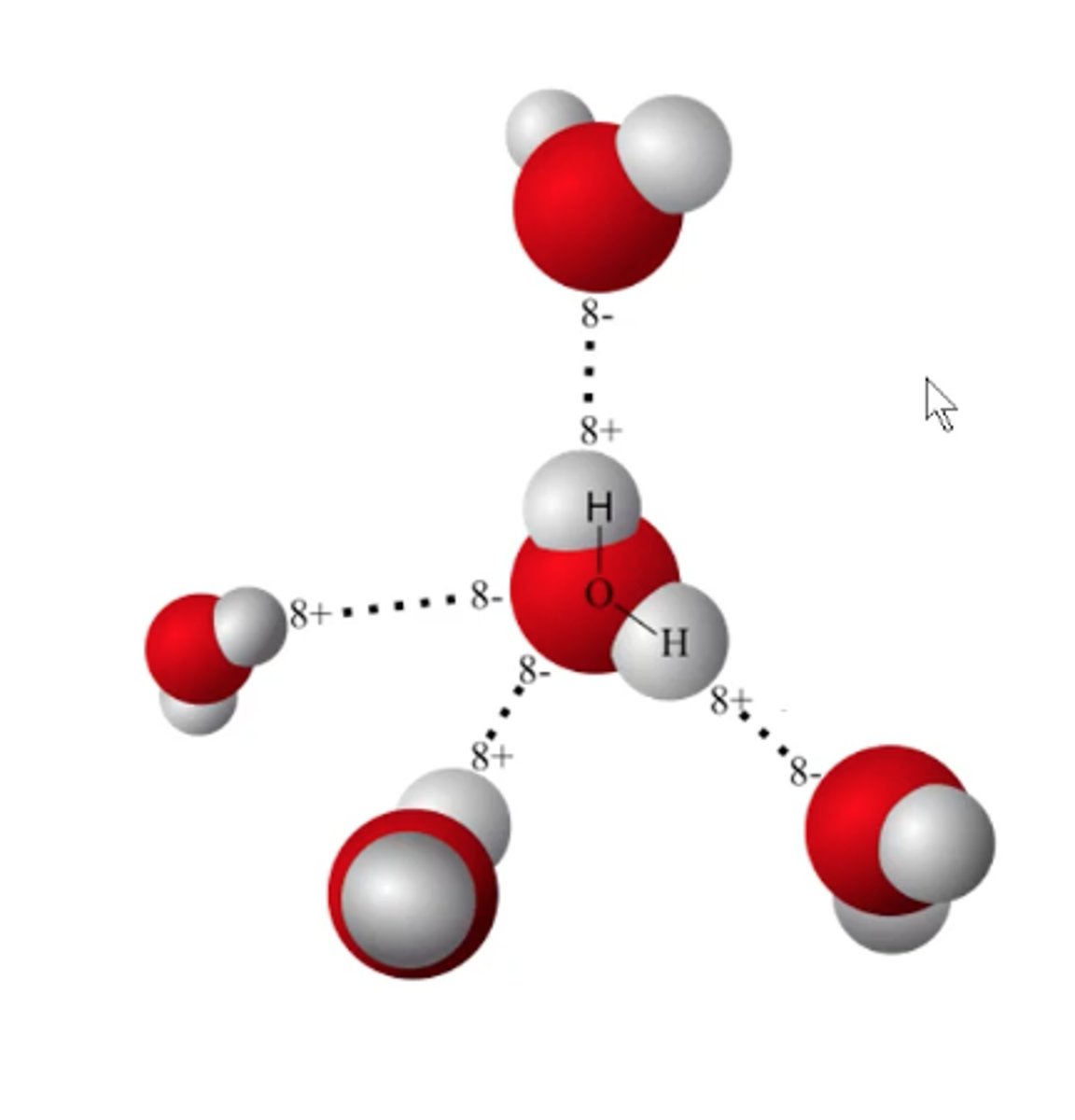
H bonding in H₂O:
partial charges cause them to stack on eachother
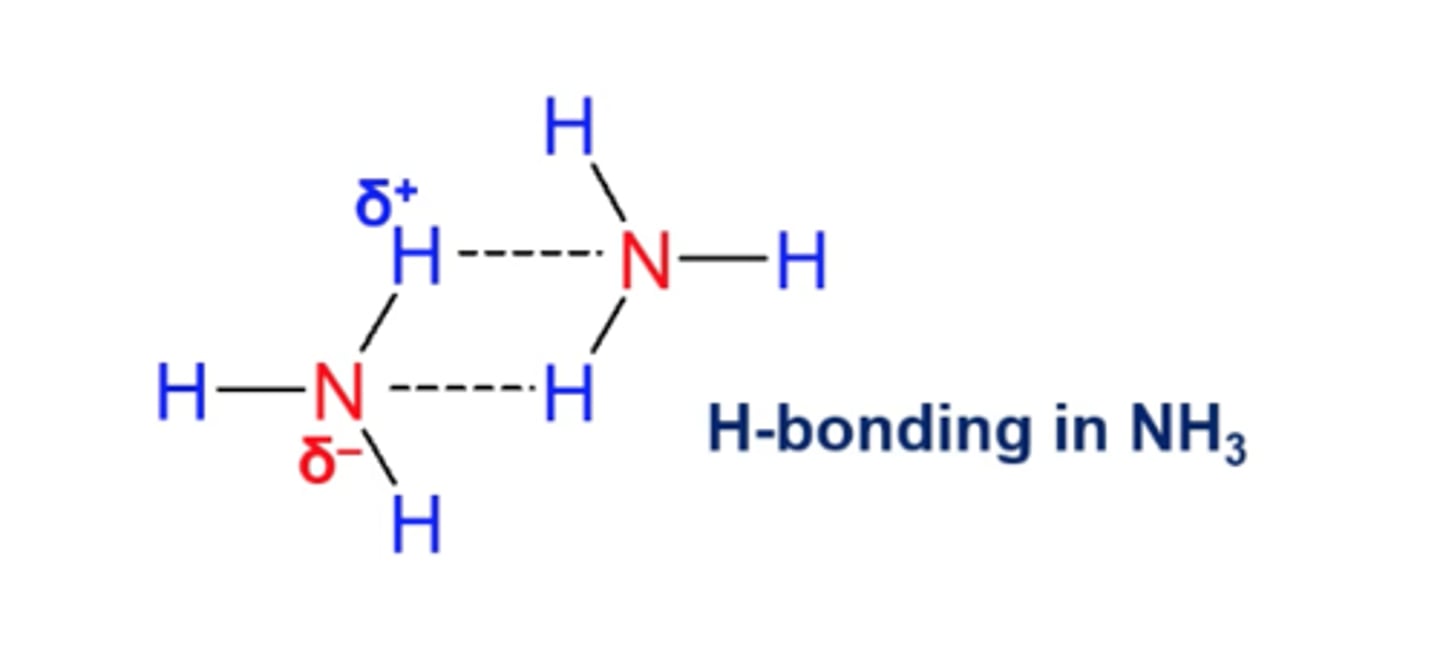
H bonding in NH₃:
partial charges cause them to stick on each other
H bonding in HF:
partial charges cause them to stick together
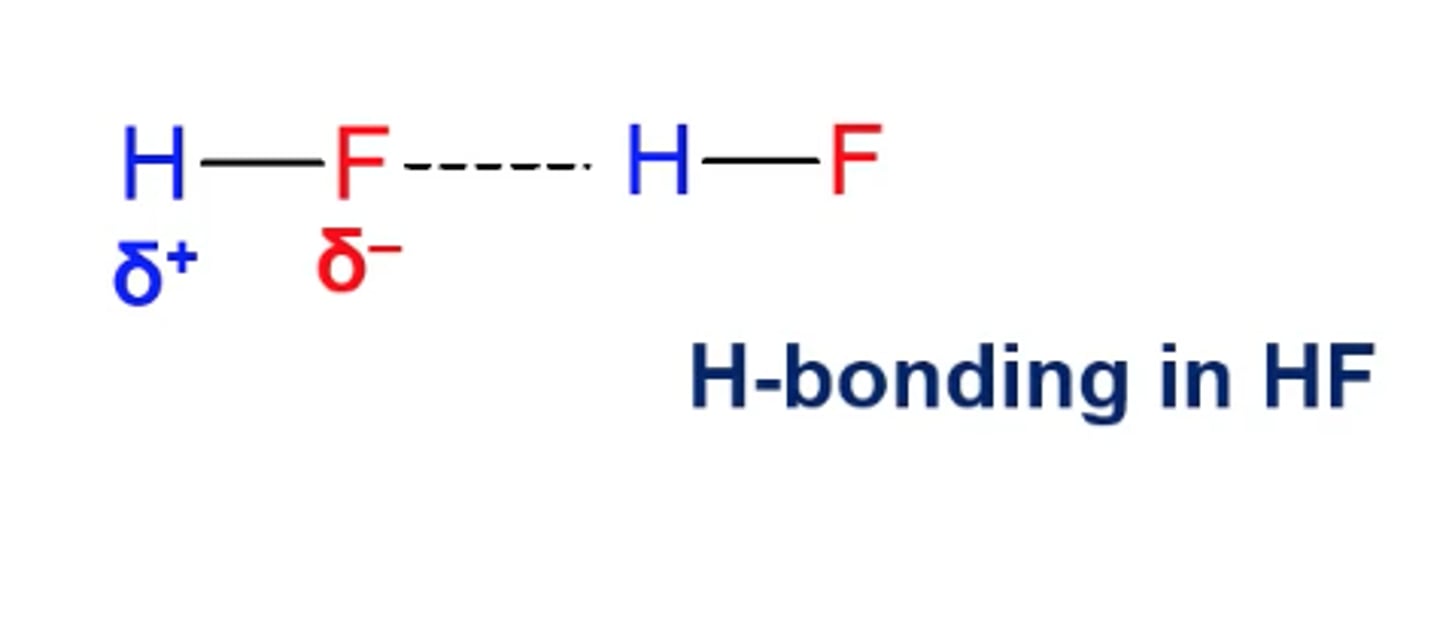
Hydrogen Bonding is strong because of the ______ electronegativity of F, O, N.
high
Dipole Dipole Forces
cause when two bonded atoms have significant electronegativity difference - causes molecule to have partial neg on one atom and a partial positive on another
Dipole Dipole only apply to...
polar, non-ionic molecules that do not have an H bonded to an O, N, F
Dispersion Forces
nonpolar; there is not an uneven sharing of electrons because they have similar electronegativities; they have momentary partial charges from the electrons moving around each other
If you have two molecules with only dispersion forces, the __________ molecule will have a higher B.P.
larger
- it has more dispersion forces
Higher IM forces = ________ B.P.
higher
Higher IM forces = ________ Heat of Vaporization
higher
needs more heat to get molecules to separate and turn into a gas
Higher IM forces = ________ Viscosity
higher
Higher IM forces = ________ Surface Tension
higher
the greater amount of weight the surface can contain
Higher IM forces = ________ Vapor Pressure
lower
Vapor Pressure
the upward pressure exerted by the substance on the atmosphere
Boiling occurs when the pressure exerted by the liquid towards the atmosphere ________ the pressure exerted by the atmosphere down on the liquid molecules.
equals
@ Higher altitudes, there is lower atmospheric pressure and the boiling point __________.
decreases