SNC2DY-a Chemistry Quiz 1: Matter and it's Properties
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
20 Terms
pure substance
A substance made of only one kind of matter and having definite properties.
element
A pure substance made of only one kind of atom
compound
A substance made up of atoms of two or more different elements joined by chemical bonds
physical properties
-malleability, ductility, lustrous/dull, melting point/boiling point, solubility, conduct heat/electricity
chemical properties
-flammability, reactivity with water and acids, toxicity, radioactivity
physical vs chemical change
Physical - does not create a new substance, atoms are not rearranged into different compounds
Chemical - converts one substance into another; atoms are rearranged
periodic table
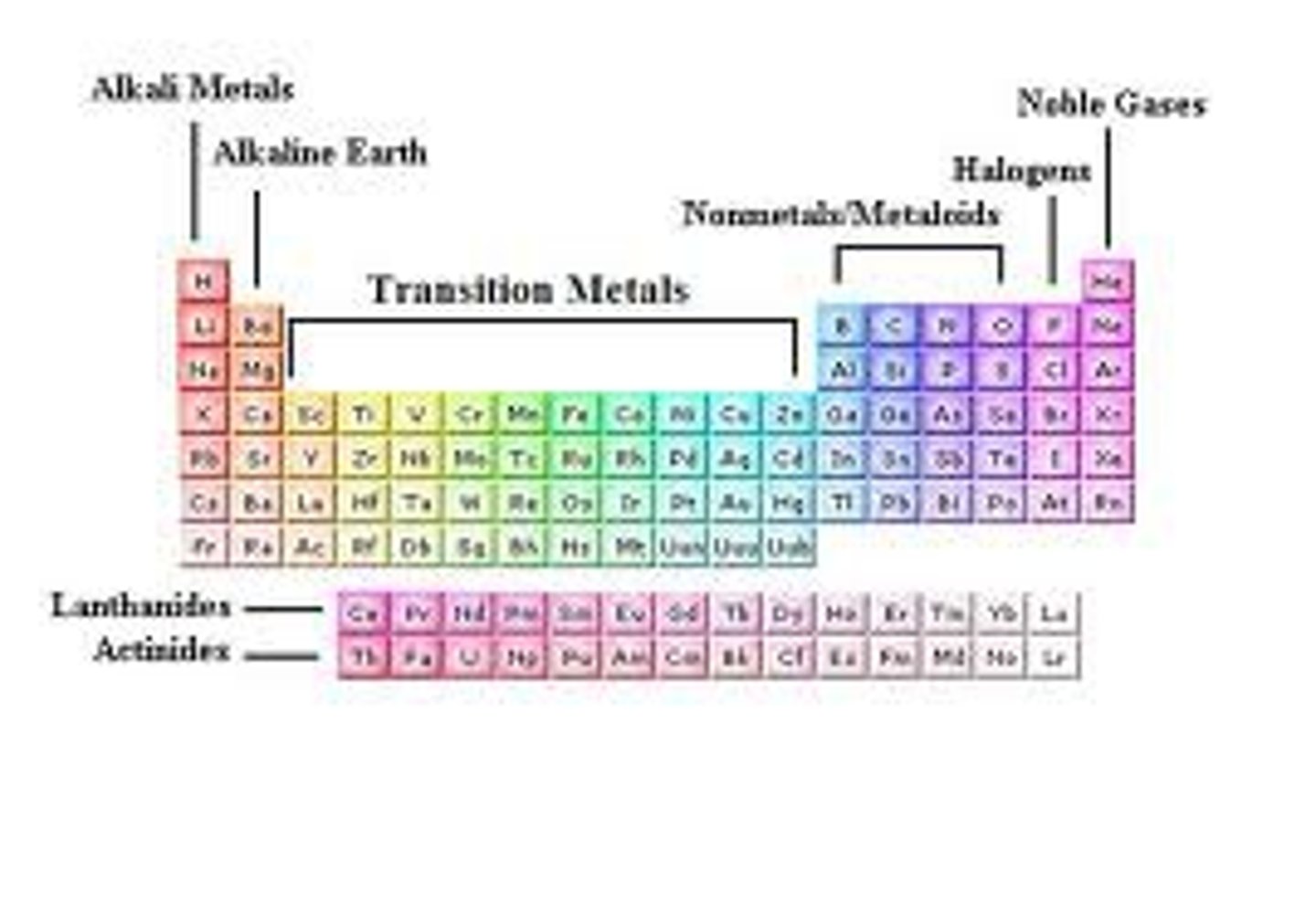
properties of metal
ductile, malleable, conduct electric current well, crystalline structure, high melting points, solids except hg
properties of non metals
dull, brittle, poor conductors, low melting n boiling points, solids or gases except Br
metalloids
-semi conductors moderate boiling/melting, conduction etc
alkali metals
react w water acids halogens oxygen
alkaline earth metals
react w water acids halogens oxygen harder than grp 1
transition metals
majority react with water acids oxygen halogens , precipitates
halogens
strong unpleasant odor can burn skin
noble gases
stable w ful ve shell colorless but when charged emits color
nuclide symbol
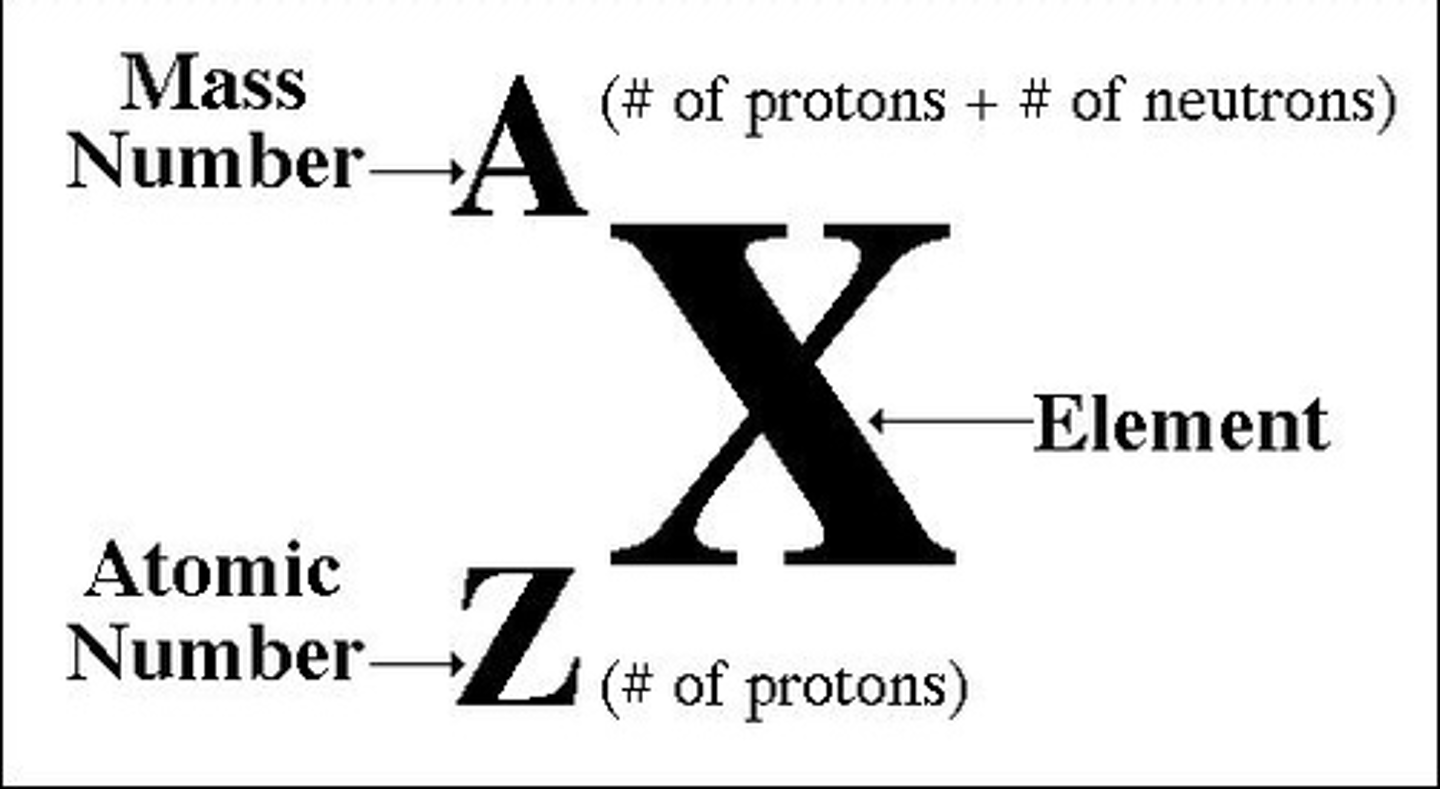
charged ions
gain=negative
lose=positive
BR diagram
-energy levels
-planetary
-greater the distance of the electron the stronger
jumps are called energy transitions
-electron in lowest energy level is called ground state
-excited electron=excited state
iso electronic
same number of electrons (w noble gases usually)
isotopes
Atoms of the same element that have different numbers of neutrons and diff atomic mass
-must have same atomic #