Chemistry - atomic structure and the periodic table
1/253
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
254 Terms
How are particles arranged in solids?
tightly packed in fixed, regular arrangements, held by strong forces that restrict their movement to vibrations in place, giving solids a definite shape and volume
How are particles arranged in liquids?
close together but randomly arranged, allowing them to slide past each other and flow
How are particles arranged in gases?
widely spaced, randomly arranged, and move quickly and freely in all directions, possessing high kinetic energy that overcomes intermolecular forces, allowing them to fill any container they occupy.
What state of matter has the most energy?
gas
What state of matter has the least energy?
solid
What is solid to gas called?
sublimation
What is solid to liquid called?
melting
What is liquid to gas called?
evaporation
What is liquid to solid called?
freezing
What is gas to liquid called?
condensation
What is gas to solid called?
deposition
What are the properties of solids?
- definite shape and volume
- definite melting point
- high density
-incompressible
- low rate of diffusion
What are the properties of liquids?
- they take the shape of a container
- they flow and move randomly
- they cannot be compressed or squashed
- They have a constant density
What are the properties of gases?
- no definite shape
- no definite volume
- particles can expand to fill a container
- can be compressed
Why do solids vibrate at fixed positions?
because their particles are packed tightly together in a rigid, ordered lattice structure and are held by strong intermolecular forces of attraction
Is freezing endothermic or exothermic?
exothermic
Is evaporation exothermic or endothermic?
endothermic
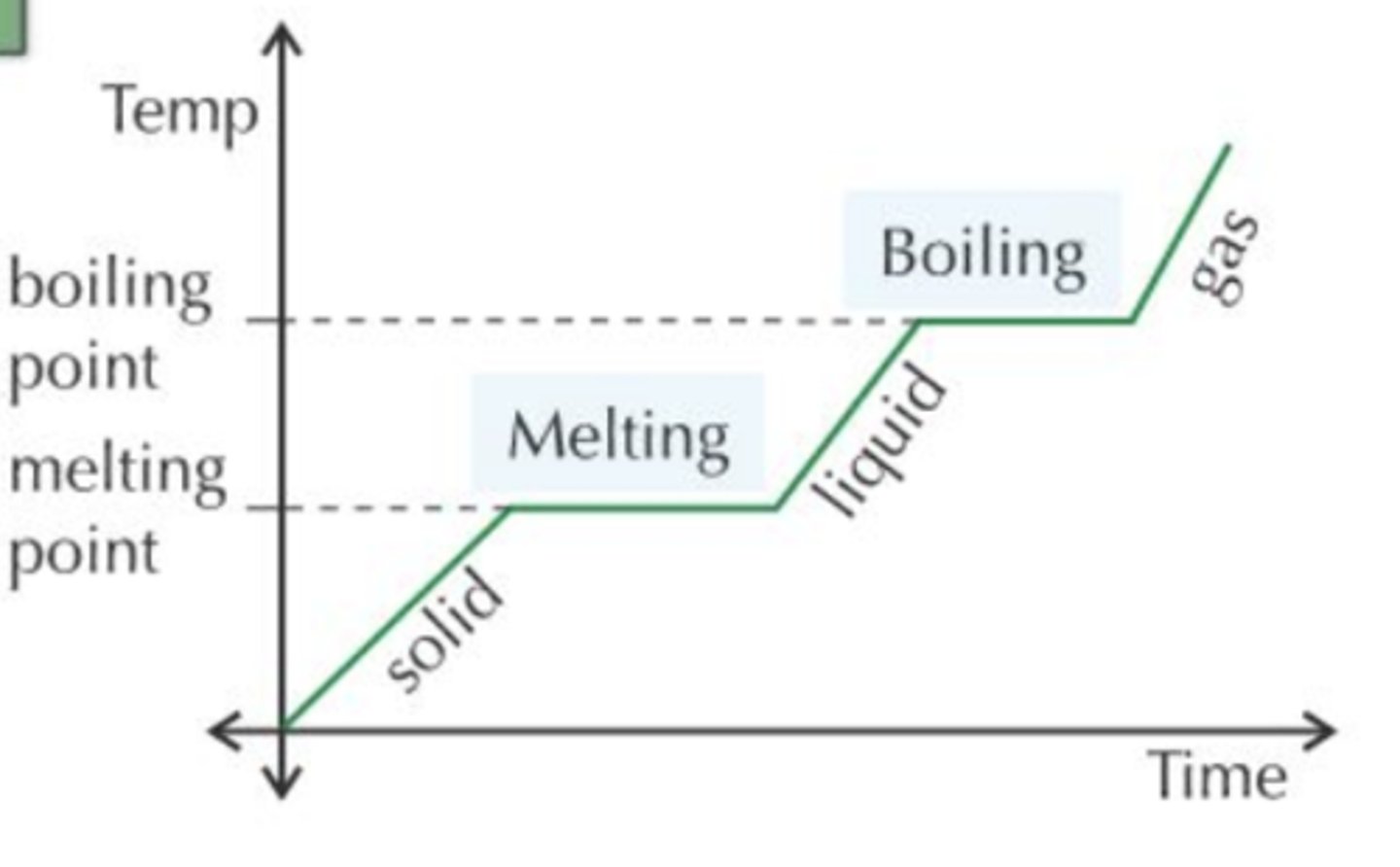
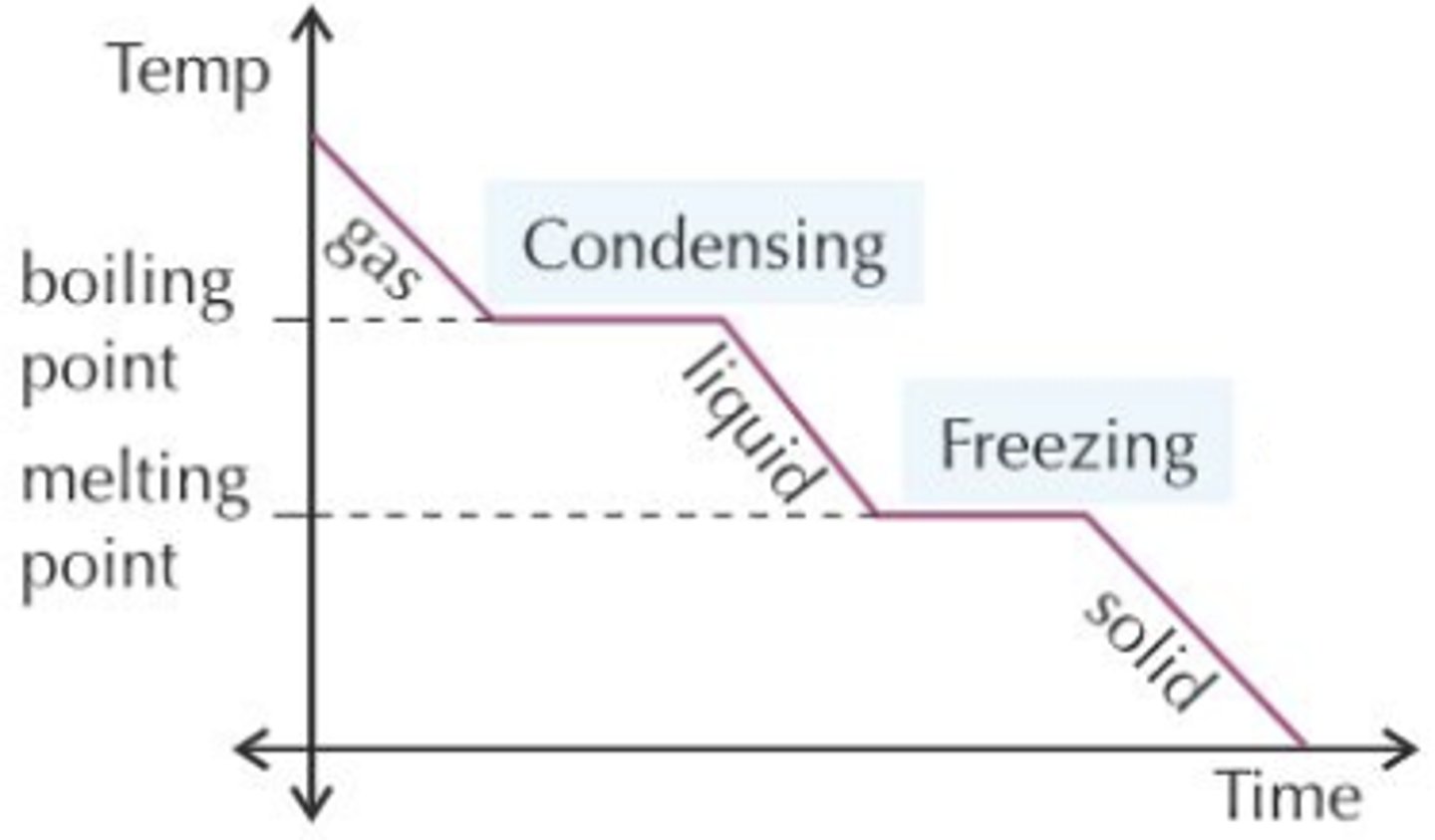
Why does the temperature stay constant when a change of state occurs?
because the energy added or removed is used to break or form intermolecular bonds rather than changing the kinetic energy of the particles
What are physical changes?
- alterations to a substance's state, shape, or appearance without creating a new chemical substance.
- They are usually reversable the chemical composition remains identical, and the total mass is conserved.
What are chemical changes?
processes where atoms rearrange to form new substances with different properties, characterised by the breaking and making of chemical bonds
Heating graph
Cooling graph
What are ions?
Charged atoms
How are positive ions formed?
when an atom loses electrons
What types of atoms form positive ions?
metals
How are negative ions formed?
When an atom gains electrons
What types of atoms form negative ions?
non-metals
What is the dot and cross model?
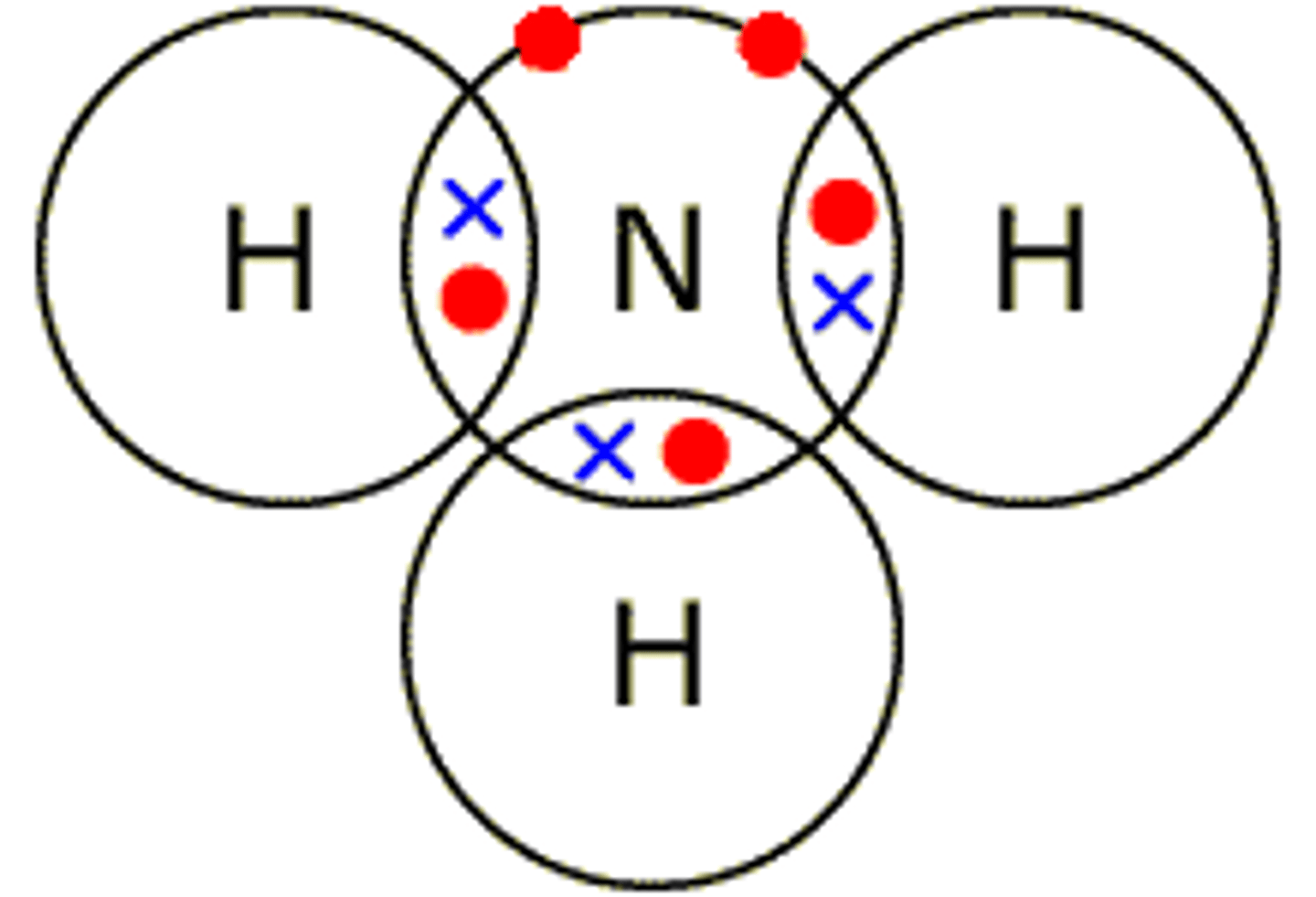
What are the advantages of the dot and cross diagram?
- show the charges
- shows the shape of the molecule
What are the disadvantages of the dot and cross diagram?
- doesn't show the lattice structure
- doesn't show bonding between the particles
What is the stick and ball model?
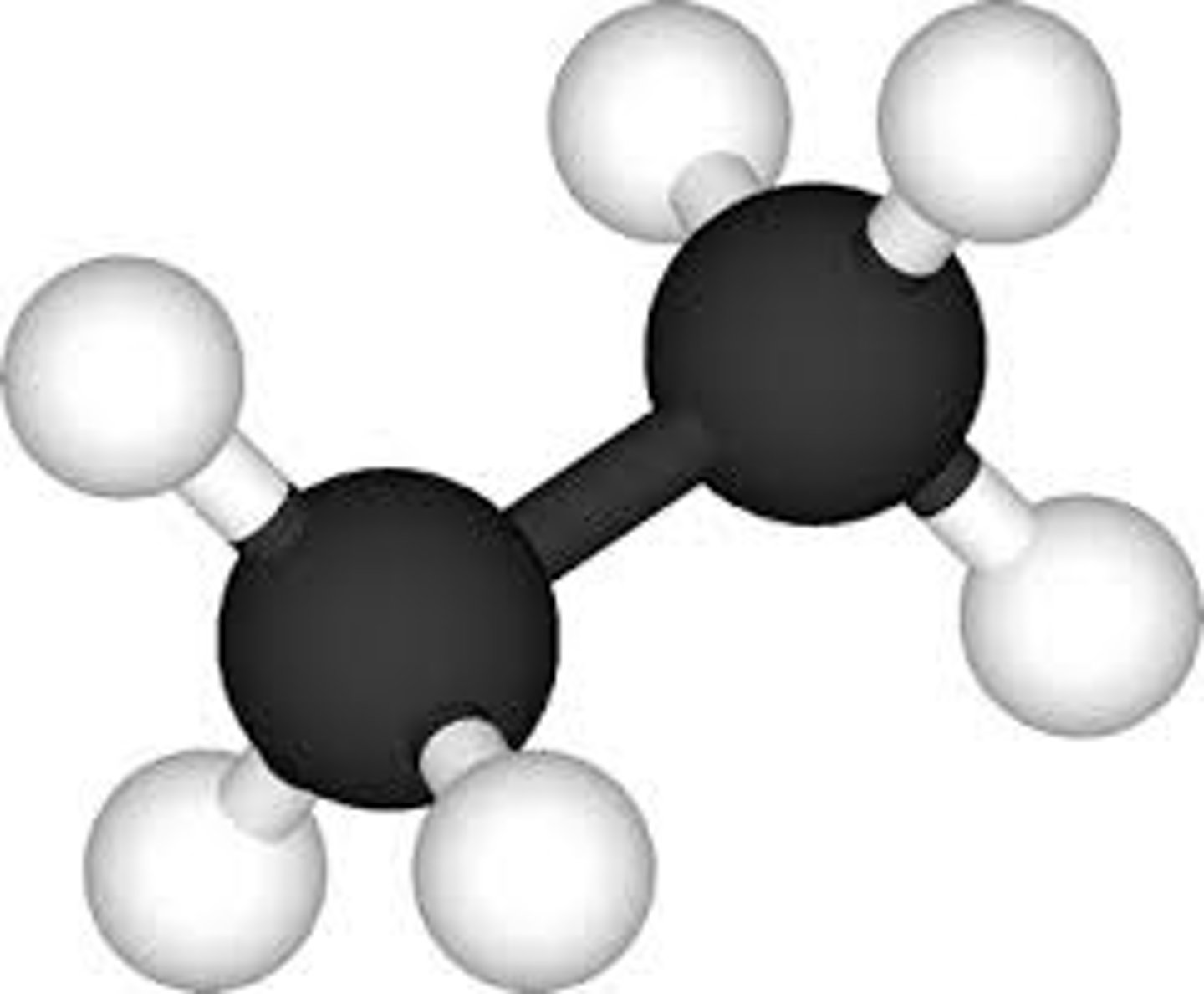
What are the disadvantages of using a ball and stick model?
- It doesn't show how bonds are formed
- Bonds are forces, not sticks
What are the advantages of using a ball and stick model?
- shows the relative sizes of the atoms
- represents forces between atoms
What is the 2D diagram?
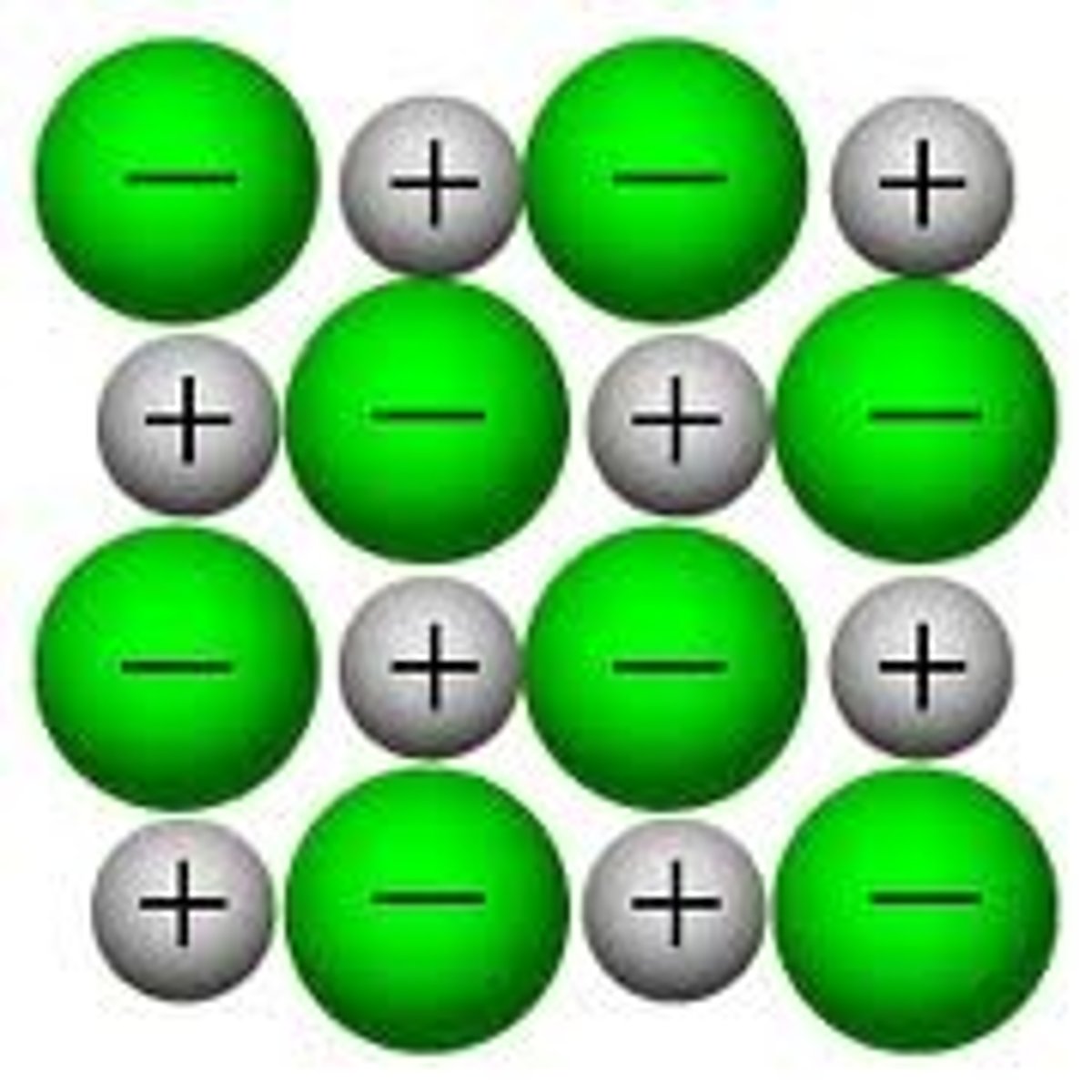
What are the disadvantages of using the 2D model?
Doesn't show how the ions were formed or more than one layer
What is the 3D model?
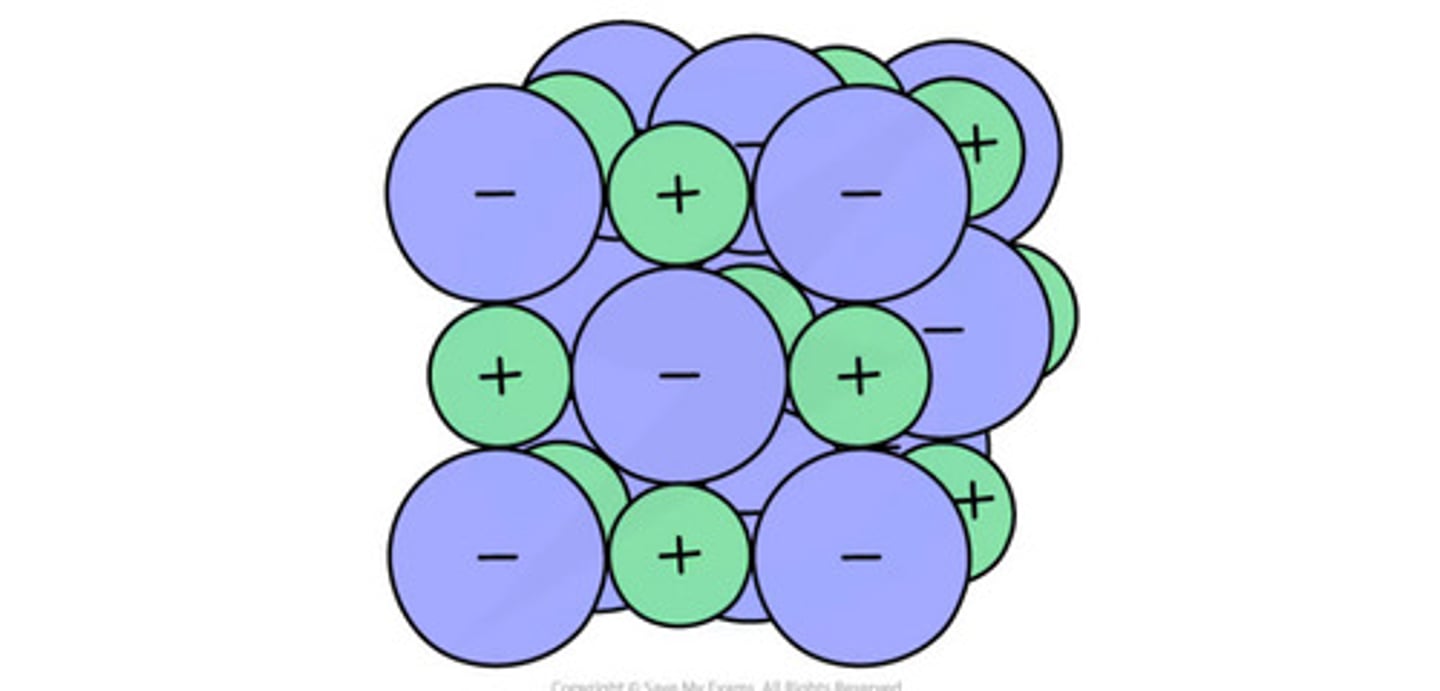
What are the disadvantages of using the 3D model?
- doesn't show charges
- there are no spaces between ions
What are the disadvantages of using the space-filling model?
- doesn't show how the bonds are made
- doesn't show which elements are present
What is a space-filling model?
What types of atoms are in ionic bonds?
metals and non-metals
Why do atoms try to have a full shell of electrons?
to achieve a stable electron configuration. They become stable and unreactive
Why do atoms bond?
To become more stable
What is metallic bonding?
the chemical bonding that results from the attraction between metal atoms and the surrounding sea of electrons
What structure do metals have?
Giant metallic lattice
What is a lattice structure?
a giant, three-dimensional, repeating arrangement of millions of atoms, ions, or molecules bonded together in a fixed, orderly pattern held together by strong forces.
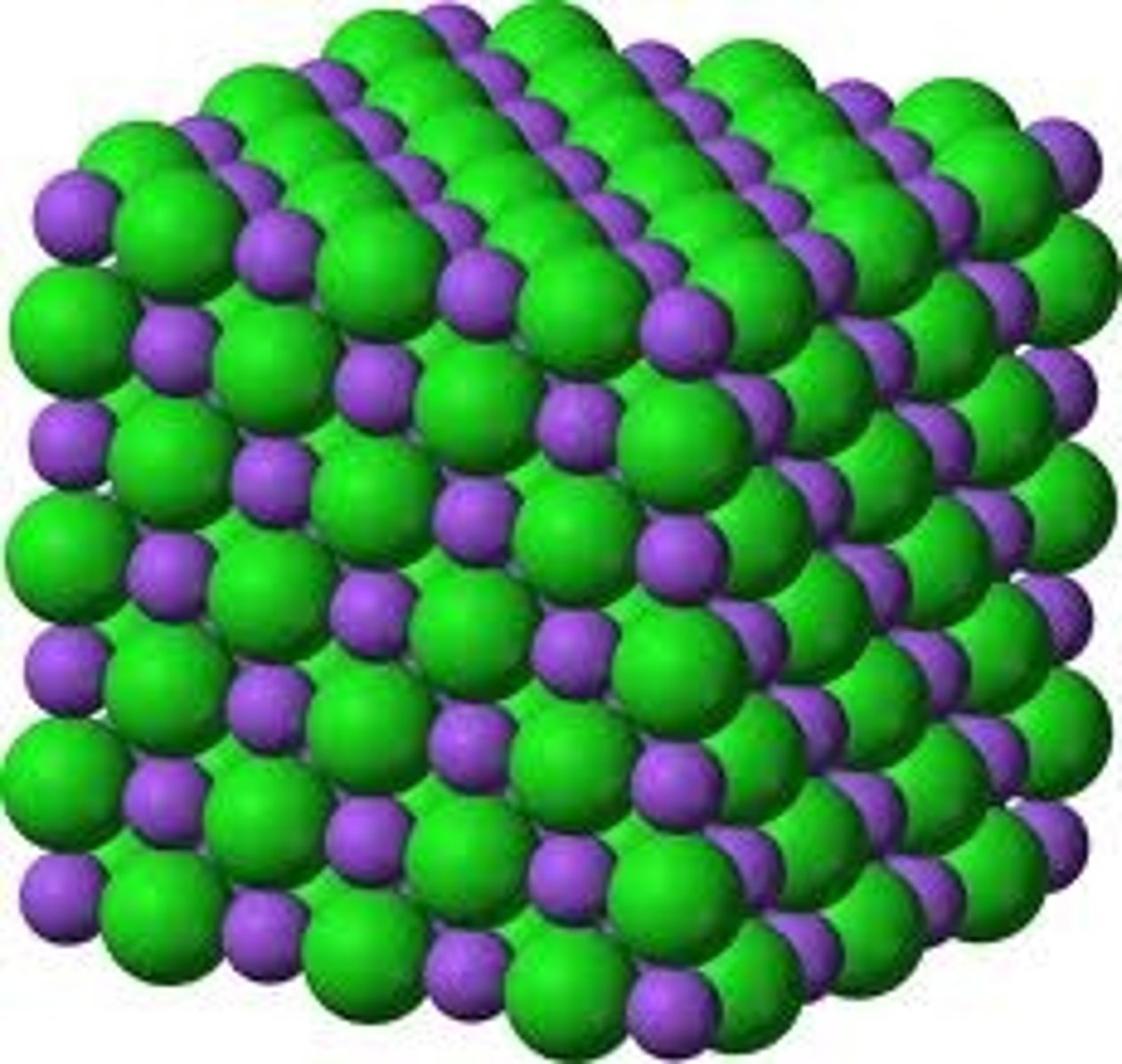
What type of structure does metallic bonding form?
Giant metallic lattice
How does metallic bonding form?
1. The outer electrons from the metal atoms delocalize, forming positive metal ions
2. The electrons form a sea of delocalized electrons surrounding the positive ions
3. The strong electrostatic force between the positive metal ions
What does delocalised mean?
free to move around and not bound to an atom
What are the properties of metals?
- high melting/boiling points
- good conductors of electricity/heat
- malleable
- shiny and dense
Why do metals have high melting and boiling points?
The strong electrostatic attraction between the positive ions and delocalised electrons requires a lot of energy to break
Why are metals good conductors?
The delocalized electrons are charged and are free to move through the structure, allowing electricity and heat to pass through easily.
What is an alloy?
a mixture of two or more elements, at least one of which is a metal
Why are metals malleable?
Metals form layers that can slide over each other without breaking bonds. So pure metals can be hammered into shape and stretched into wires
Why are alloys less malleable?
the different sized atoms distort the regular structure. This makes it more difficult for the layers to slide past each other
What does malleable mean?
can be hammered into shape
What does ductile mean?
can be drawn into wires
Why are metals shiny?
Free electrons in metals vibrate when light strikes them, thereby reemitting the light as reflected light.
What types of atoms are in metallic bonding?
metals
What types of atoms are in ionic bonding?
metals and non-metals
What is ionic bonding?
Strong electrostatic attraction between oppositely charged ions
What is electrostatic force?
The force that charged particles exert to push or pull on each other.
How does ionic bonding work?
1. A metal atom loses electrons to form an positivley charged ion
2. The electron is transferred to a non-metal, forming a negative ion
3. These ions have full outer shells, making them more stable
4. The positive and negative ions attract each other strongly, forming an ionic bond
5. These bonds hold the ions together in a giant lattice structure
What structure do ionic compounds form?
Giant ionic lattices
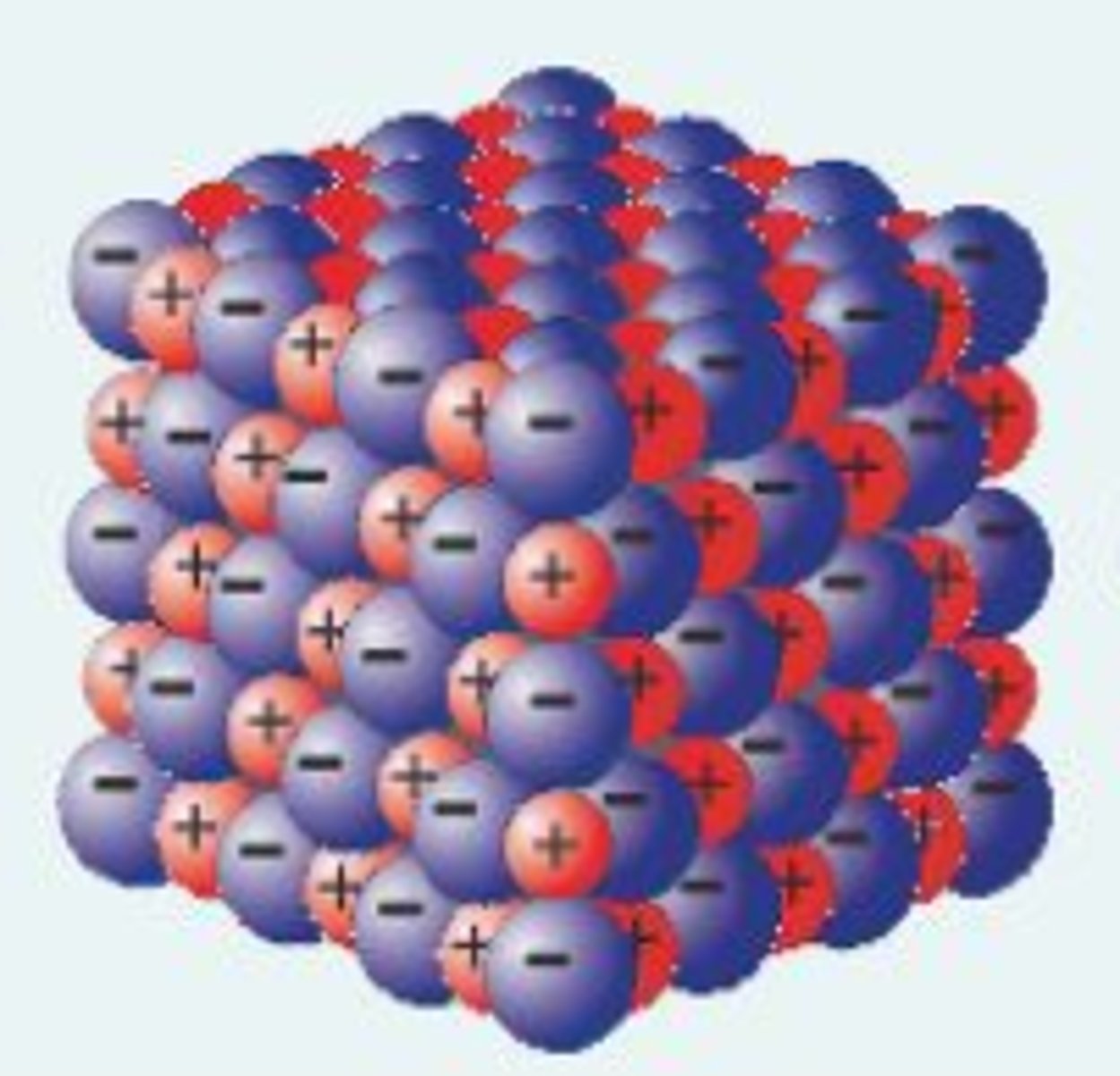
What ions do group 1 form?
+1 ions
What ions do group 2 form?
+2 ions
What ions do group 3 form?
+3 ions
What ions do group 4 form?
Group 4 generally don't form simple ions. But when they do, the heavier metals at the bottom of the group (Tin and Lead) typically form +2 or +4 ions
What ions do group 5 form?
3- ions
What ions do group 6 form?
2- ions
What ions do group 7 form?
1- ions
What are anions?
negatively charged ions
What are cations?
positively charged ions
What are the properties of ionic compounds?
-High melting and boiling points
-Don't conduct electricity when solid
-Do conduct electricity when molten or in solution
- soluble in water
Why do ionic compounds have high melting points?
Strong electrostatic attraction between the positive and negative ions requires a lot of energy to overcome.
Why can't ionic compounds conduct when solid?
When in solid form, the ions can't move, so electricity can't flow. However, when molten or dissolved, the ions are free to move and carry a charge
Why are ionic compound soluable in water?
Many ionic compounds dissolve in water because the water molecules surround the ions
What is a solution?
a homogeneous mixture of two or more substances (a mixture of solute and solvent)
What is a solute?
the substance that is dissolved
What is a solvent?
the substance in which the solute dissolves
What are covalent bonds?
a chemical bond that involves the sharing of electron pairs between atoms
What types of atoms form covalent bonds?
nonmetals
How does covalent bonding work?
- Non-metals share valence electrons to gain a full outer shell of electrons
- Each pair of shared electrons is a covalent bond
- The atoms are held together strongly by the electrostatic attraction between the positive nucleus and the negatively charged bonding region
What structure do covalent bonds form?
simple molecular structures and giant covalent structures
How many covalent bonds are in group 5 molecules?
3 covalent bonds
How many covalent bonds are in group 6 molecules?
2 covalent bonds
How many covalent bonds are in group 7 molecules?
1 covalent bond
What are intermolecular forces?
forces of attraction between molecules
Why do covalent bonds have a low melting point (in molecules)?
The intermolecular forces between the molecules are weak and don't need a lot of energy to overcome
Why can't covalent bonds conduct?
because they have no free, charged particles to carry charge
What are allotropes?
substances made from the same element but with different structures in the same physical state
What are giant structures?
Structures made from many atoms each joined to adjacent atoms by strong covelant bonds
What are the allotropes of carbon?
diamond, graphite, graphene, fullerenes
Are intermolecular forces weak or strong?
very weak
What is the strongest type of bonding?
covalent
What is the weakest type of bonding?
intermolecular forces
What are the properties of covalent bonding?
- Low melting and boiling points because of the weak intermolecular bonds that act between the molecules
- They don't conduct because they have no free charged particles to carry charge
Why do intermolecular forces happen?
because of electrostatic attractions between opposite partial or temporary charges on neighboring molecules
What are the properties of intermolecular forces?
-Weak forces between molecules
-Not as much energy needed to overcome
-Molecules are quite separated
-Higher melting/ Boiling point as molecules get bigger
Why do intermolecular forces increase with a molecule's size?
because they have larger molecules, have more electrons and a greater surface area.
What are simple covalent molecules?
substances made up of individual molecules which are held together by intermolecular forces