Chemistry - test 3 - covalent molecules
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Molecular substances
molecules are usually made up of only non-metals (opposed to an ionic compound)
properties of molecular substances
have low melting and boiling points
are non-conductive
have low hardness
Covalent bonds
as non-metals have similarly high electronegativities, they ‘share’ electrons.
a covalent bond is the result of simultaneous attraction of the two positive nuclei to the same shared pair of electrons.
results in each atom gaining a full octet in its valence shell.
INTRAmolecular forces
exist between the atoms in a molecule.
strong, due to electrostatic attraction between nucleus and electrons.
broken with large amounts of energy.
INTERmolecular forces
between molecules of a covalent substance.
generally weaker than intramolecular forces.
broken with small amounts of energy.
Bonding vs lone pairs
bonding pairs involve shared electrons
a lone pair has no shared electrons
VSEPR theory
Valence Shell Electron Pair Repulsion theory- states that electron pairs repel each other as they are negatively charged
leads to predictable shapes with consistent and specific bond angles
Linear molecules
have one single bond or two double bonds
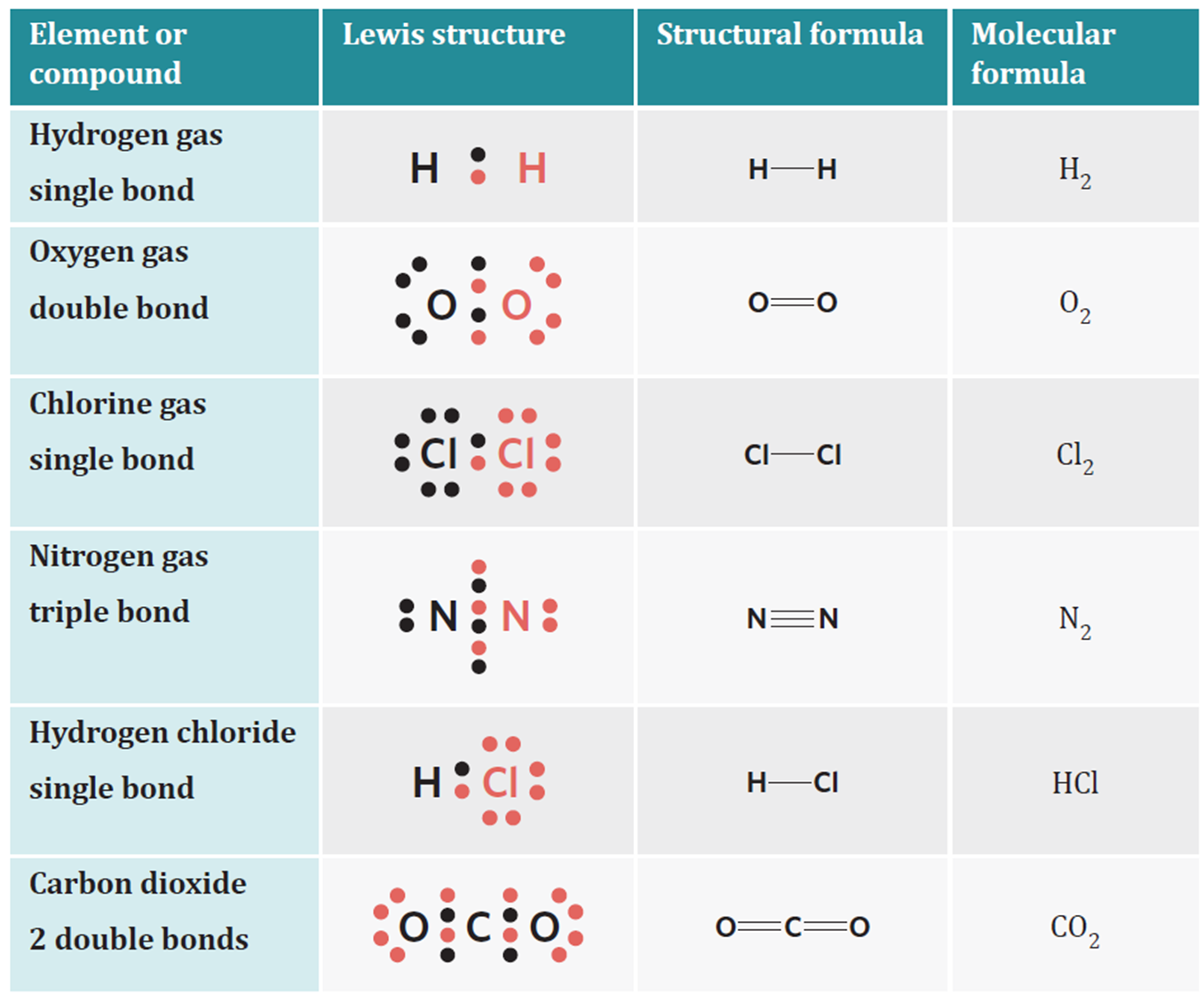
Bent molecules
two single covalent bonds pointing outwards next to each other

pyramidal molecules
have three bonding pairs and one lone pair
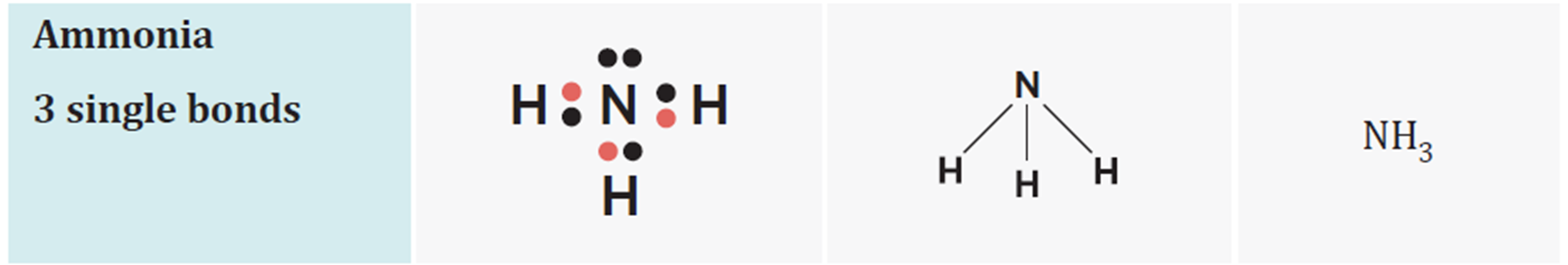
tetrahedral molecules
no lone pairs, all single bonds
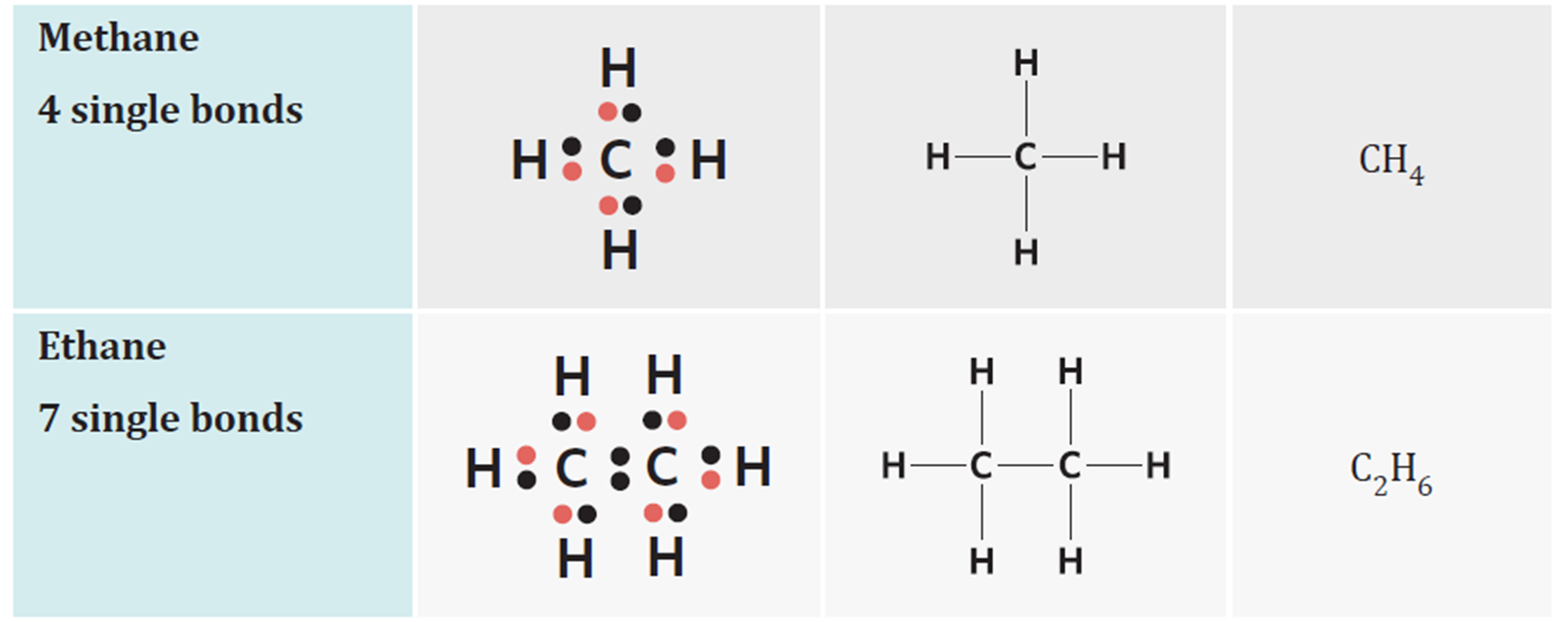
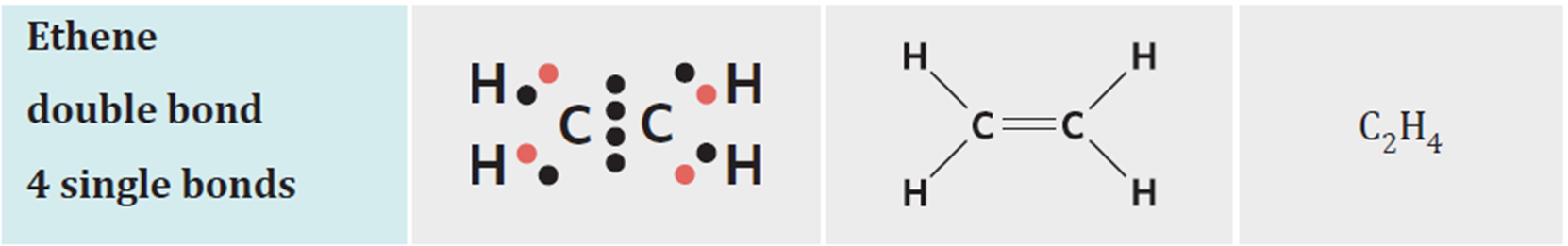
trigonal planar molecules
one double bond, two lone pairs
molecular polarity
atoms with higher electronegativities pull electrons closer, meaning they have a slight negative charge.
polar bonds occur when the difference in polarity of atoms is between 0.5 and 2
polarity and symmetry
if a molecule with polar bonds is symmetrical, the pull is equal in both directions, so it will be non-polar overall
determining polarity
identify atoms involved in the molecule
using electronegativity values on the periodic table, determine electronegativity difference
use VSEPR theory to draw shape
if polar, add dipole symbols (δ+ or δ-)
if the charges are not symmetric, the molecule is polar
Intermolecular forces: Hydrogen bonding
strongest of intermolecular forces
ONLY occurs when hydrogen is bonded to flourine, oxygen or nitrogen due to electronegativities
Dipole-Dipole interactions
same as hydrogen bonding, but for molecules not including F, O, N
positive dipoles of polar molecules are attracted to negative dipoles of other polar molecules
Dispersion forces
Weakest of intermolecular forces
instantaneous dipoles cause a slight charge on one side
all covalent molecular substances have dispersion forces
boiling and melting points increase as molecules become larger
Allotropes of carbon - diamond
carbon atoms are bonded to four other carbon atoms in a tetrahedral, covalent network lattice
non-conductive but high thermal conductivity
very high sublimation point
graphite
layer lattice of each carbon atom bonded to three others
fourth valence electron is delocalised
conducts heat and electricity
hardness depends on directionality- can be soft and slippery
high sublimation point
chromatography
used to separate and thus identify components of a mixture by polarity, weight and size
involves a stationary phase and a mobile phase; one is polar, one is non polar
a sample is added to the origin of the stationary phase
the mobile phase moves along stationary phase
different compounds in the sample will desorb (up) into the mobile phase at different rates
polar compounds move further in a polar solvent
non-polar compounds will not move as far
retention factors
the ratio of a compound’s distance travelled to the distance travelled by the solvent
both are measured from the origin