Important Terms
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
physical property
something you can observe about a substance without changing what it is. If you describe it, it stays the same thing
Chemical Property
ability of a substance to change into a new substance.
Homogeneous Mixture
everything is mixed so perfectly that you can't see the individual parts. It looks the same from top to bottom
Heterogeneous Mixture
you can actually see the different "chunks" or layers. It is not mixed evenly.
Element
Only one type of atom present.

Compound
Two or more different types of atoms chemically bonded
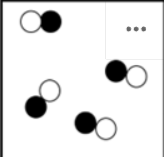
Mixture
Different types of atoms or molecules present that are not bonded to each other.

Molecular Formula
Count every single atom you see in the drawing C4H6
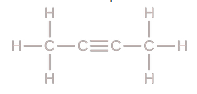
Empirical Formula
molecular formula and divide by the largest common factor C2H3
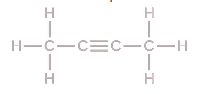
Photon Emission
Occurs when an electron drops from a HIGHER energy level to a LOWER energy level.
Covalent Naming Rules
Non-metal + Non-metal.
Use Greek Prefixes
DONT BALANCE out charges
Molecule
Two or more atoms held together by covalent bonds
λ
Wavelength
ν
frequency (s^-1) or Hz
Atomic Mass Formula
(mass x abundace) + (mass x abudnace) = element amu
and change abundance to 1-x when looking for abundace and there is no other abundance
How to find neutrons
(mass number - atom number)
Isotopes
Same: Atomic Number (Protons)
Different: Mass Number (Protons + Neutrons)
How is an anion formed?
When atom gains electrons
What are cations
Metals who form a positive charge
What are anions
Non metals who form a negative charge
How are photons gain
When energy is added to an atom
Quantitive
Number and units
Qualitative
Physical description