lab 2
1/87
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
88 Terms
what is a key requirement for survival
is the ability of cells to control the movement of substances (ions, nutrients, proteins, gases, and wastes) across the plasma membrane between the intracellular and extracellular fluids
key characteristic of the plasma membrane
semipermeable; it allows some substances to pass through while preventing others from crossing
how does selectivity help support homeostasis?
helping maintain appropriate cell volume, ion balance, nutrient availability and waste removal
what is the plasma membrane made from
phospholipid bilayer with numerous embedded proteins
characteristics of phospholipids
hydrophilic head and hydrophobic tails
forming a barrier to many water soluble substances
can small non-polar molecules and lipid soluble cross the membrane?
easily can cross
can large polar molecules and ions cross the membrane?
cannot freely pass and require transport proteins
can water pass through the membrane?
molecules move either directly across or using aquaporins
two main forces that influence movement of ions and molecules across cell membranes
concentration gradient
electrical gradients
concentration gradients
differences in solute concentration between two areas
molecules tend to move from higher to lower concentration
electrical gradients
differences in total number of positive and negative electrical charges on either side of the membrane
opposite charges (positive-negative) attract, and like charges (negative-negative or positive-positive) repel
what do the chemical and electrochemical gradients form?
electrochemical gradient; important for ion movement
Movement from areas of high concentration to low concentration is considered movement ____ a gradient
down
movement from areas of low concentration to high concentration is considered movement____ a gradient
against
does movement down a gradient require energy?
no
does movement against a gradient require energy?
yes, usually ATP
two main types of membrane transport
passive and active
passive processes
involve movement ions or molecules across the cell membrane from areas where they are more highly concentrated to areas where they are less concentrated
no energy required; relies on constant movement of ions/molecules
equilibrium
point where concentrations are even
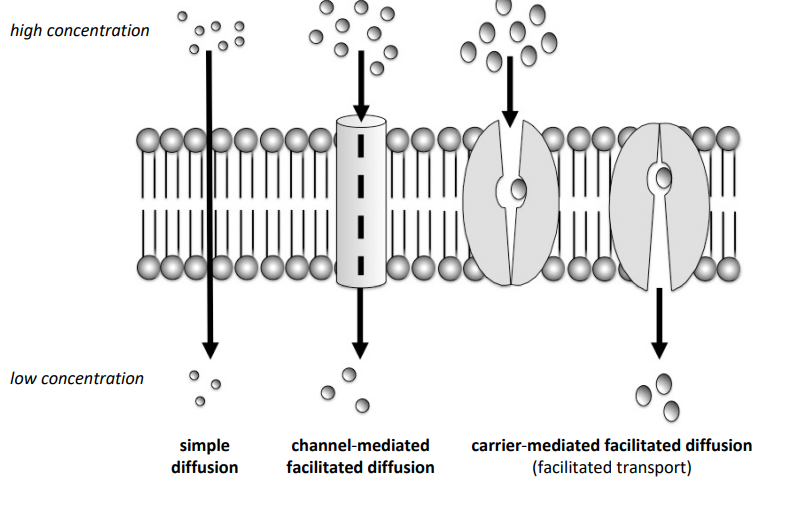
What is simple diffusion?
Movement of molecules directly through the phospholipid bilayer from high → low concentration without using membrane proteins or energy.
What types of molecules use simple diffusion?
Small non-polar or lipid-soluble molecules, including:
O₂
CO₂
fatty acids
steroid hormones
fat-soluble vitamins (ex: vitamin D)
What small polar molecule can sometimes move through the membrane by simple diffusion?
water
What is facilitated diffusion?
Passive movement of polar molecules or ions across the membrane through membrane proteins from high concentration to low concentration. No ATP required.
Which substances usually require facilitated diffusion?
Polar molecules and charged particles such as glucose and ions (Na+, K+, Cl−, Ca2+).
What two types of proteins allow facilitated diffusion?
Channel proteins – create a pore for molecules or ions to pass through.
Carrier proteins – bind to a molecule and move it across the membrane.
What is the difference between simple diffusion and facilitated diffusion?
Simple diffusion: molecules move directly through the phospholipid bilayer.
Facilitated diffusion: molecules move through membrane proteins.
What is osmosis?
Net movement of water across a selectively permeable membrane from high water concentration (low solute) to low water concentration (high solute).
Why does osmosis occur?
Many solutes cannot cross the membrane easily, so water moves to balance differences in solute concentration.
Why is osmosis important in physiology?
Movement of water can change cell volume (cells may swell or shrink).
does active transport require energy?
yes; supplied by the high energy bonds found in ATP
types of active transport
primary active transport
secondary active transport
vesicular transport
What is primary active transport?
Movement of ions or molecules across the membrane against their concentration gradient using a carrier protein and energy.
What direction do substances move in primary active transport?
From low concentration to high concentration.
What type of protein is used in primary active transport?
Carrier proteins called pumps that bind solutes and move them across the membrane.
What is secondary active transport?
Movement of a substance against its concentration gradient using energy from an ion gradient created by primary active transport.
Does secondary active transport use ATP directly?
No. It uses energy stored in an ion gradient.
Example of secondary active transport?
Na⁺ moving into the cell down its gradient drives glucose into the cell against its gradient (such as in intestinal cells).
What is vesicular transport?
Movement of very large particles or large numbers of molecules across the membrane using membrane-bound vesicles.
What is endocytosis?
Substances enter the cell when the cell membrane forms a vesicle around them.
What are the two types of endocytosis?
Phagocytosis – “cell eating”, large particles enter the cell.
Pinocytosis – “cell drinking”, water and dissolved solutes enter the cell.
What is exocytosis?
Substances leave the cell when a vesicle fuses with the cell membrane and releases its contents into the extracellular fluid.
What happens at equilibrium in diffusion?
Solutes become evenly distributed across the membrane.
Can all molecules diffuse across the membrane if a concentration gradient exists?
No. Some molecules require a protein channel or carrier to cross the membrane.
How does the steepness of a concentration gradient affect diffusion?
A steeper gradient causes more molecules to move from high to low concentration.
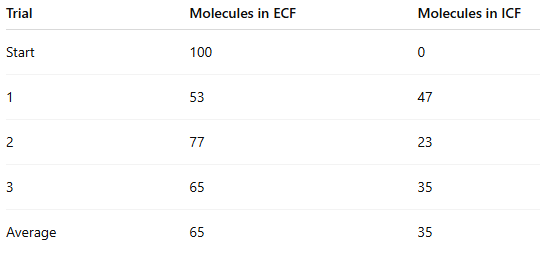
Which side had the highest concentration at the start?
ECF (100 vs 0)
At the end of the experiment, how many beads moved from ECF to ICF?
100 − average final ECF
100 − 65 = 35 beads
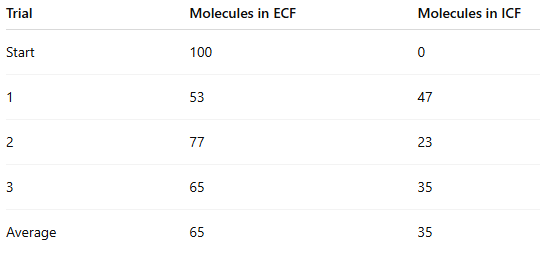
Did the ECF have higher or lower concentration at the end compared to the beginning?
Lower
(100 → 65)
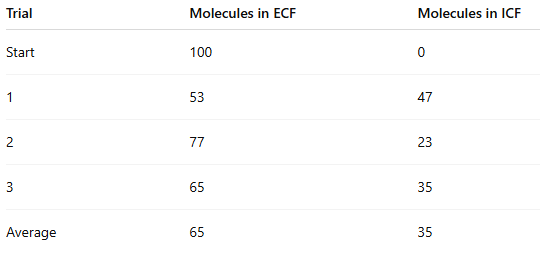
Did the ICF have higher or lower concentration at the end compared to the beginning?
Higher
(0 → 35)
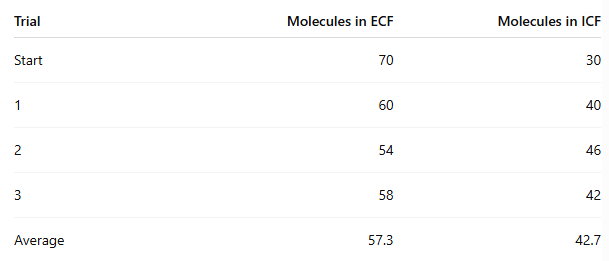
Which side had the highest concentration at the start?
ECF
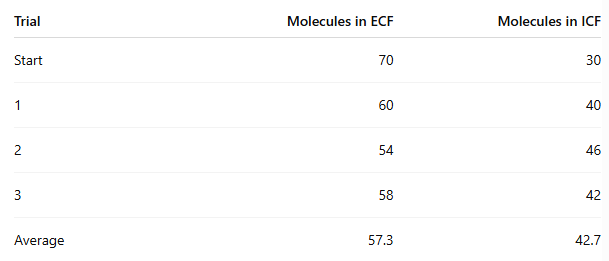
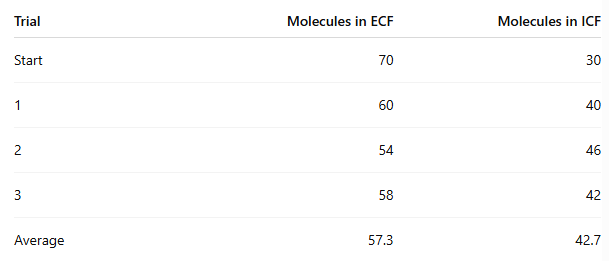
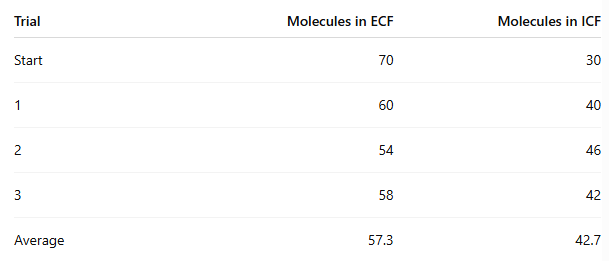
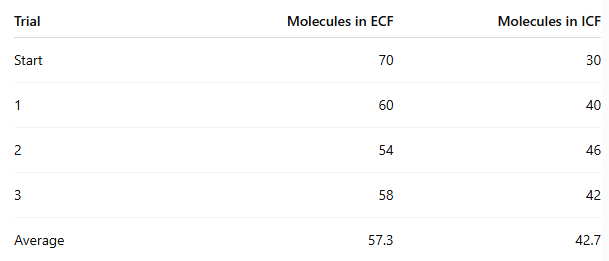
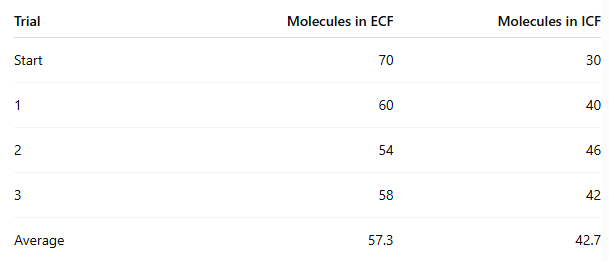
At the end of the experiment, how many molecules moved from ECF to ICF?
70 - 57.3 = 12.7 molecules on average
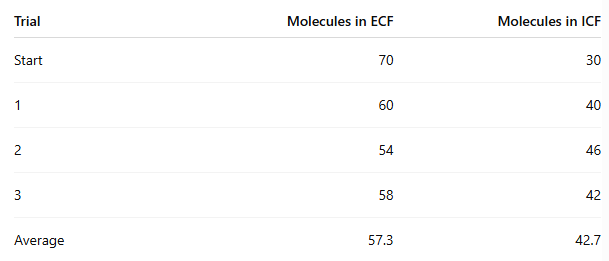
In which procedure was the concentration gradient steeper, and did it affect movement?
Procedure 5 (100/0) was steeper than Procedure 6 (70/30)
Yes, the steeper gradient caused more beads to move
Procedure 5: about 35 moved
Procedure 6: about 12.7 moved
What would happen if 70 beads were in the ICF and 30 in the ECF?
Net movement would go from ICF to ECF
Beads would move from the higher concentration side to the lower concentration side until equilibrium was approached
If carrier proteins actively transported beads against their gradient, what would happen?
Beads would be moved from lower concentration to higher concentration
This would make the distribution less even
One side could build up a higher concentration instead of moving toward equilibrium
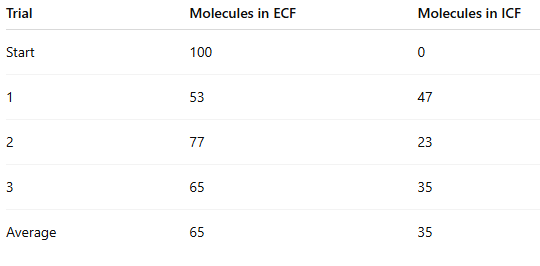
Why is using the average of 3 trials better than using only the first trial?
It reduces random error
It gives a more reliable result
One trial could be unusually high or low by chance
After equilibrium is established, do molecules keep moving through open channels?
Yes
Molecules continue to move both directions
Does that movement substantially change the number on either side?
No
At equilibrium there is no net movement, so the numbers stay about the same overall
if you keep shaking the container beyond the 15 seconds, after equilibrium is established, do ions/molecules continue to move through the open membrane channels? Does this movement substantially change the number of molecules on either side of the membrane?
movement will still occur
no net movement
In osmosis, water moves from what type of solution to what type?
From a solution with low solute concentration (high water concentration) to a solution with high solute concentration (low water concentration).
What gradient actually drives osmosis?
The water concentration gradient (their own concentration gradient), not the solute gradient.
How can water cross the cell membrane during osmosis?
Directly through the membrane or through protein channels called aquaporins.
what is the direction of osmosis determined by?
by the osmotic pressure on either side of a semipermeable membrane
What is osmotic pressure?
The pulling force created by a difference in solute concentration across a semipermeable membrane that draws water toward the side with more solute.
How does solute concentration affect osmotic pressure?
More solute (less water) = higher osmotic pressure = stronger pull on water.
What is the osmotic pressure of pure water?
Zero, because it contains no solutes.
When will there be no net movement of water between two solutions?
When the solutions have equal solute concentrations and equal osmotic pressures (isotonic).
can’t cross the membrane
any solutions containing solutes will have a
greater osmotic pressure than water
ex. ECF has total solute concentration equivalent to 0.9% solution of NaCl and the ICF also has a solute concentration equivalent to 0.9 NaCl, then osmotic pressure of the two solutes?
are equal and there will be no net movement
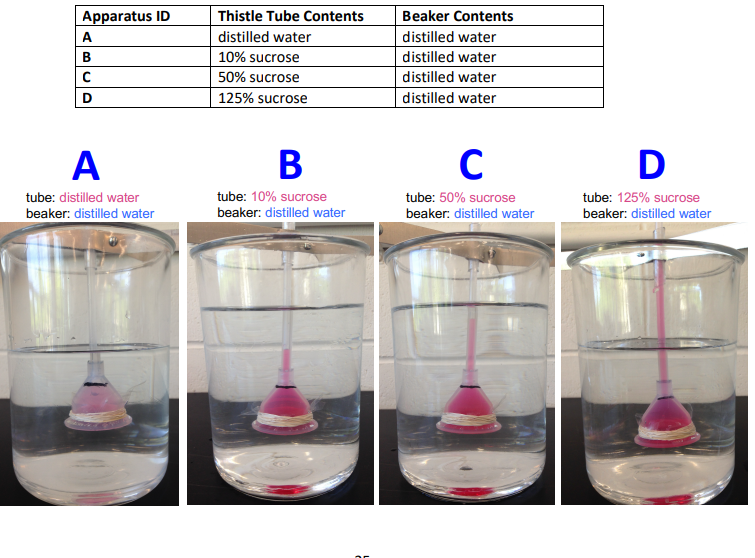
for each apparatus did net osmosis occur?
A: no; both sides are distilled water, equal solute concentration
B: yes; tube has more solute than beaker. water moves from distilled water in the beaker into 10% sucrose tube
C: yes; tube has even more solute than the beaker, water moves into the tube
D:yes; tube has the highest solute concentration, water moves into the tube strongly
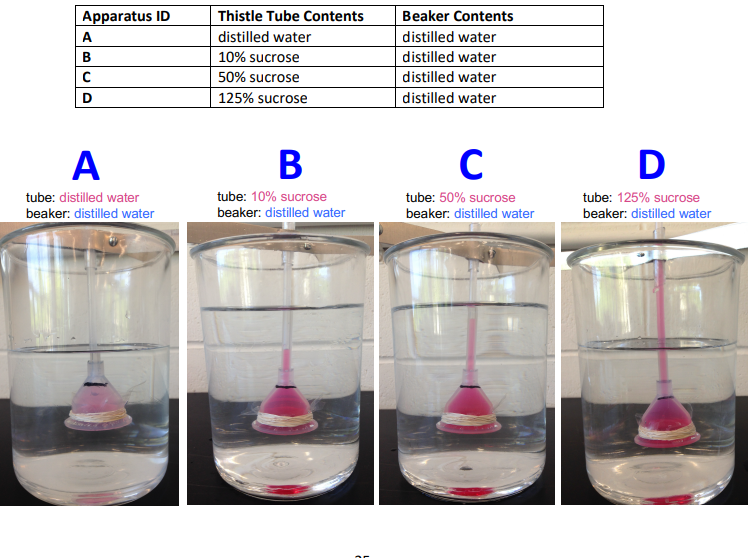
in apparatus A, are water molecules moving across the membrane at all?
yes
water will move randomly in both directions across the membrane
but both sides are the same, movement is equal both ways, so there is NO NET OSMOSIS (ISOTONIC)
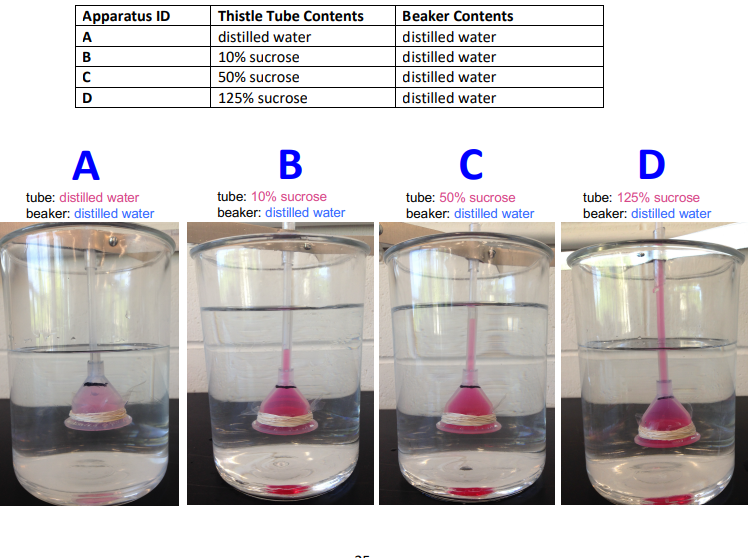
which apparatus showed the greatest change in the position of the meniscus? which showed the least?
greater change: D; greater difference in solute concentration, the greater the osmotic pressure difference
least change: A; no concentration difference, no net change
osmotic pressure in lecture notes
minimum pressure required to stop inward flow of a pure solvent (water) across the semipermeable membrane into a more concentrated area
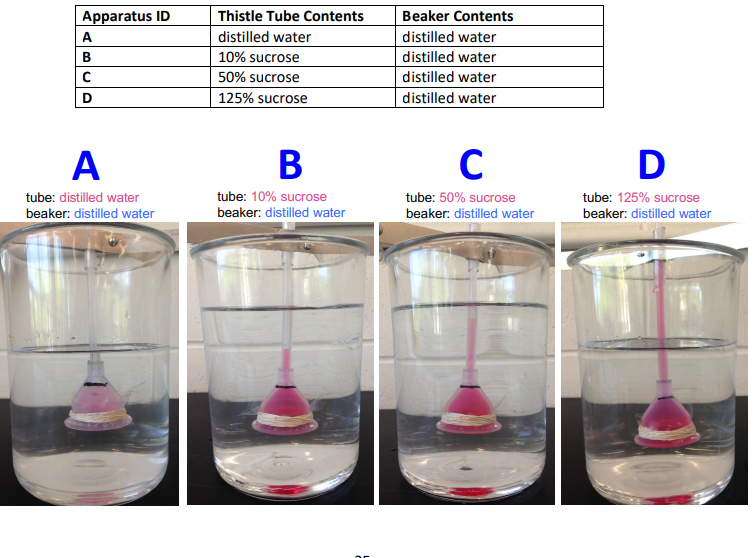
Which thistle tube had the greatest osmotic pressure? Which had the least?
Greatest osmotic pressure: D (125% sucrose)
Least osmotic pressure: A (distilled water)
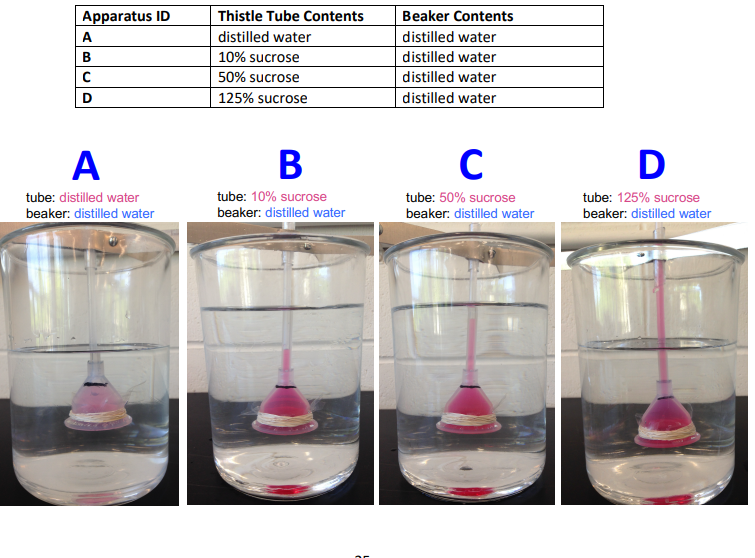
How does osmotic pressure relate to sucrose concentration?
Higher sucrose concentration = higher osmotic pressure
More solute means a stronger pull on water
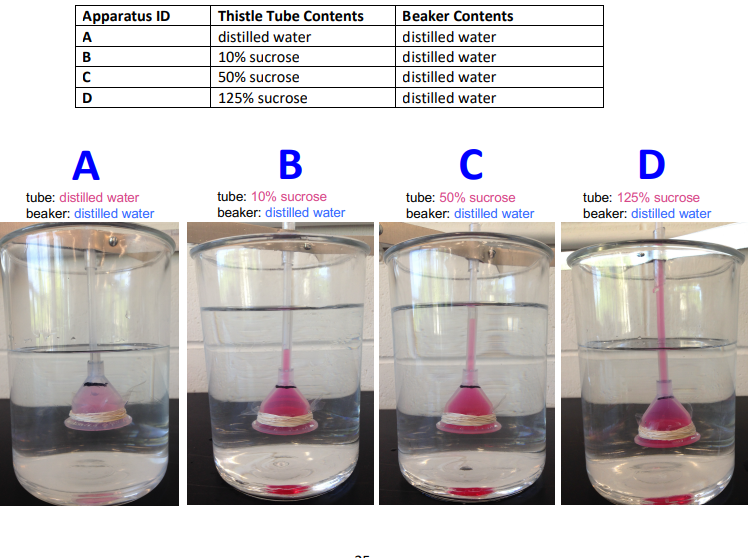
How did the osmotic pressure of the beaker compare to the tube in each case?
A:
Beaker and tube had equal osmotic pressure
No net water movement
B:
Tube had higher osmotic pressure than beaker
Water moved into tube
C:
Tube had much higher osmotic pressure than beaker
More water moved into tube
D:
Tube had the highest osmotic pressure compared with the beaker
Greatest water movement into tube
compared to animal cells, plant cells
have cell wall that provides and extra layer of protection from fluctuations in the environment or internal conditions
what happens when an animal cell vs a plant cell is placed in a hypoosmotic solution?
red blood cell will swell and burst
plant cell will become turgid (swollen) against the cell wall
what happens when an animal cell vs a plant cell is placed in a hyperosmotic solution?
animal cell will crenate (also shrink)
plant cell, membrane will shrink away from the cell wall
toncity
described how a solution (ECF) affects the size/volume and shape of a cell based on the concentration of solutes it contains (and the permeability of the cell membrane to the solute)
isotonic solution
ECF and ICF have equal osmotic pressure
no net movement of water movement
no overall or net change in volume on either side
cell volume remains stable
hypotonic solution
ECF has lower osmotic pressure (higher water concentration, lower solute) than the ICF
water molecules will enter down their concentration gradient and the cell will swell
cell membrane ruptures (lysis)
hypertonic solution
ECF has higher osmotic pressure (lower concentration, higher solute) compared to the inside of the cell (ICF)
water molecule will exit the cell down their concentration gradient and the cell will shrink
cell dehydrates
is 0.9% NaCl hypotonic, isotonic, or hypertonic to the purple onion cells?
isotonic
5% solution on the purple onion cell
shape maintains due to cell wall but the contents shrivel and shrink
hypertonic solution
is distilled water hypotonic, isotonic, or hypertonic to the onion skin cells?
hypotonic
Explain the observations of the thistle tube experiment using the concepts of osmosis, osmotic pressure, and tonicity.
Osmosis: water moves across a semipermeable membrane from low solute (high water) → high solute (low water).
The sucrose solutions in the thistle tubes had higher solute concentration than the distilled water in the beaker.
Because of this, the tube solutions were hypertonic relative to the beaker.
Water moved from the beaker into the tubes by osmosis.
Higher sucrose concentration → higher osmotic pressure → stronger pull on water.
This caused a larger rise in the meniscus in tubes with higher sucrose concentrations.
In apparatus A both sides contained distilled water (isotonic), so water moved both directions equally and there was no net osmosis.
all of the following factors that influence whether or not a net movement of solute will occur across a cell membrane.
a) The concentration of electrically neutral (uncharged) solutes in the solution and the cell.
b) The permeability of the membrane to the solute.
c) The tonicity of the cell.
d) The concentration of the solute on either side of the membrane. e) The osmotic pressure of the solution the cell is placed in.
permeability of the membrane to the solute
the concentration of the solute on either side of the membrane
After competing in a water chugging contest, a 28-year old woman is brought to the emergency room with signs of osmotic cerebral edema (swelling of the brain due to movement of water from the blood into the intra- and extracellular spaces in the brain). Which of the following IV (intravenous) fluids would the doctors/nurses likely administer to this patient in order to reduce the swelling of the brain cells? Explain your selection with reference to tonicity.
a) 0.3% saline b) distilled water c) 0.1% NaCl d) 3% NaCl e) 0.9% saline
3% NaCl
Imagine you are stranded on deserted island with no access to fresh water, only seawater. After two days without drinking water, you decide to drink a liter of seawater to quench your thirst. Based on your understanding of osmosis and the knowledge that seawater is an ~3.5% solution of NaCl:
a. Predict what will happen to the cells in your body after drinking the seawater. Will water move into or out of your cells?
b. Why does drinking seawater ultimately lead to dehydration rather than rehydration, even though it is made up of mostly water?
a: water moves out of cell as seawater is hypertonic relative to body cells, cell will shrink
b: water is pulled out of cells by osmosis causing the cells to lose water; kidneys must also use extra water to remove the excess salt in the urine; because more water is lost than gained, the body becomes more dehydrated over time