BIOC PART C
1/79
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
80 Terms
glucose→ glucose-6-phosphate (hexokinase, or glucokinase in liver) ATP→ADP
fructose→ fructose-1,6-bisphosphate (phosphofructokinase-1) ATP→ADP
phosphenolpyruvate→ pyruvate (pyruvate kinase) 2ADP→2ATP
3 most exergonic steps glycolysis (and enzymes)
pyruvate→phosphenol pyruvate (many enzymes)
fructose-1,6- bisphosphate → fructose 6- phosphate (fructose 1,6 bisphosphatase) H2O→Pi
glucose-6 phosphate → glucose (glucose 6 phosphatase) H2O→Pi
3 most exergonic steps of gluconeogenesis (and enzymes)
pyruvate enters mitcochondria*
pyruvate→ oxaloactetate (pyruvate caboxylase) +CO2, 2ATP→2ADP
oxaloacetate→ malate (mitochondrial malate dehydrogenase) NADH+H→NAD+
malate exits the mitochondria*
malate→oxaloacetate (cytosolic malate dehydrogenase) NAD+→ NADH+H
oxaloacetate→ phosphenol pyruvate (phosphenol pyruvate carboxylase) -CO2, 2GTP→2GDP
explain all the steps of step one gluconeogenesis (pyruvate to PEP)
IV: fructose-6-phosphate
I: glucose-6-phosphate
what inhibits glucokinase (hexokinase IV)?
what inhibits hexokinase I?
F-6-P→F1,6BP (PFK-1)
what is the committed step in glycolysis?
activates: (x100) F26BP, AMP, ADP
inactivates: citrate, ATP
what inhibits step 3 glycolysis?
what activates step 3 glycolysis?
(F-6-P→F1,6BP (PFK-1))
activates: F1,6BP (product of step 3)
inactivates: long chain FA, alanine, ATP, acetyl-COA
what activates step 10 glycolysis?
what inactivates step 10 glycolysis?
(PEP→pyruvate (PK))
pyruvate kinase (PK) is active when glucagon is low in liver (allows glycolysis to proceed)
in liver when glucagon is released it activates PKA which inactivates pyruvate kinase. when is pyruvate kinase active? (step 10 glycolysis)
activates: acetyl COA
inactivates: ADP
what activates step 1 gluconeogenesis?
what inactivates step 1 gluconeogenesis?
(pyruvate→PEP (many enzymes))
activates: ?
inactivates:AMP, F26BP(x10)
what activates step 8 gluconeogenesis?
what inactivates step 8 gluconeogenesis?
(F1,6BP→F6P (F1,6BPase))
glycolysis, gluconeogenesis
in general low energy molecules activate __________ and high energy molecules activate ________
low, high
______ energy molecules favour glycolysis, ______ energy molecules favour gluconeogenesis to occur
low energy molecules include: ADP, AMP, F16BP, F26BP
high energy molecules include: ATP, acetyl-COA, long chain FA, alanine, citrate
low energy molecules include:
high energy molecules include:
sum of chemical transformations (catabolism+anabolism)
metabolism=
degrade carbs, fats, proteins
produce high energy ATP, NADPH, NADH, FADH2
byproducts of CO2,H2O,NH3
describe catabolism (what is used, made and byproducts)
use sugar, amino acids, FA, nitrogenous bases
produce proteins, polysacchardies, nucleic acids
byproducts of ADP, HPO2-, NAD+,FAD, NADP+
describe anabolism (what is used, made and byproducts)
acetylCOA, biomolecules
catabolism coverges to _______, anabolism diverges to _______
F, rate limiting steps are often far from equilibrium. the substrate can be in excess and teh pathway is still slowed down by rate limiting enzyme
rate limiting steps are in equalibrium (T/F)
when products have less energy that substrate and deltaG is negative
gibbs free energy is the ability to do work in a system when is a reaction spontaneous?
increased stability (lower energy products)
increased entropy
energy release to power cell
coupling with nonspontaneous reactions to drive processes forward
why is negative deltaG favoured?
equalibrium
non spontaneous (endergonic)
spontaneous (exergonic)
deltaG=0
deltaG>0
deltaG<0
Ea
enzymes decrease ____ of rxn
deltaG= deltaG ‘ +RT ln product/reactants
deltaG=
ln small/large (small product with large concentration of reactants)
deltaG is negative when
I: max activity in the muscle (only inhibited if G6P concentration high)
IV: normally low glucose affinity, low activity, responds as glucose concentration increases (inhibited by F6P)
hexokinase I is normally at _______ activity
hexokinase IV is normally at ________ activity
glucokinase has a much higher K0.5 than hexokinase
glucokinase K0.5=10
hexokinase K0.5=5
higher k0.5 = lower affinity for glucose
does hexokinase or glucokinase have higher K0.5
in the nucleus. only moves into cytosol to beocme active when glucose concentrations increase
where is hexokinase-IV (glucokinase) found when glucose concentrations are low/ F6P concentrations are high?
synthesis of glucose form non hexose precurors (lactoc acid, glycerol, gluconeogenic AAs)
define gluconeogenesis
gluconeogenesis consumes NADH (glyceraldehyde 3-phosphate dehydrogenase) and the liver mitcochondria degrades FA in gluconeogenesis that produces a lot of NADH.
the way to get the NADH out of the mitcochondria so it can be used in gluconeogenesis is to have it carried out by malate intermediate, then go back to oxaloacetate in the cytoplasm to relase the NADH there (bring it out of mitochondria into cytoplasm)
why does oxaloacetate→malate→oxaloacetate in the first step of gluconeogenesis (whats the point?)
when lactate is feeding gluconeogenesis (not FA breakdown in mitochondria) as lactate dehydrogenase (LDH) produces NADH right in the cytoplasm already (dont need all those steps to get it out of the mitochondria)
when do the multi enzyme steps in step 1 gluconeogenesis not need to take place?
L-malate
is malate in the first step of gluconeogenesis L or D malate?
glycolysis, gluconeogenesis
F26BP is a low energy molecule so it activates ______ and inactivates ______
PFK2 and FBPase2
F26BP production is regulated by a bifunctional enzyme of _______ and _____
not produced (gluconeogenesis occurs)
when FBPase2 is active F26BP is _________
produced (glycolysis occurs)
when PFK2 is active F26BP is _______
phosphoprotein phosphatate (PP), insulin and xylulose
(IN Muscle)inactive PFK2, active FBPase2 → active PFK2, inactive FBPase2
occurs by enzyme _______ which is activated by______
PFK2, glycolysis (inhibit gluconeogenesis)
insulin and xyulose activate phosphoprotein phosphatase enzyme,
from here ______ enzyme is activated, producing F26BP which enourages _________
PKA, glucagon,epinepherine and cAMP
(LIVER)active PFK2, inactive FBPase2 → inactive PFK2, active FBPase2
occurs by enzyme _______ which is activated by______
FBPase2, gluconeogenesis (inhibits glycolysis)
glucagon and cAMP activate PKA enzyme,
from here ______ enzyme is activated, blocking F26BP production which enourages _________
gluconeogenesis
glycolysis
in summary of the last cards regarding F26BP regulation (not rxn just flow chart)
glucagon/cAMP→PKA→FBPase2→decrease F26BP→ activates _______
insulin/xyulose5P→PP→PFK2→increase F26BP→ activates ______
Ser32
in liver isozyme of FBPase2/PFK2, phosphorylation of ________ activates FBPase2
Ser406 and Thr475
In cardiac isozyme of FBPase2/PFK2, phosphorylation of _____ and _____ activates PFK2
glucagon, inhibits (inhibiting PK inhibits glycolysis)
pyruvate kinase (step 10 glycolysis) is allosterically activated by F16BP and inhibited by high energy molecules.
however the liver isozyme of pyruvate kinase is phosphorylated by PKA in response to _________ which __________ the pyruvate kinase to reserve sugar for organs that need it
cytoplasm
parallel to glycolysis
where does the pentose phosphate pathway take place?
does pentose phosphate pathway run parallel to glycolysis or against glycolysis
oxidative: produce 2NADPH (used for reductive biosynthesis) and ribulose-5-phosphate (for nucleotide synthesis)
non-oxidative: replenishes glucose-6-phosphate and glycolytic intermediates, source of xyulose-5-phosphate (activates PFK2 and F26BP production which increases glycolysis)
what are the two main phases of the pentose phosphate pathway and what are the products/ purposes of these phases?
FBPase2 active(-P)
PFK2 active(-OH)
when PFK2/FBPase2 enzyme is phosphorylated the active domain is _________
when PFK2/FBPase2 enzyme is dephosphorylated the active domain is _________
(IN LIVER)
fructose 6 phosphate→ fructose 2,6 bisphosphate by phosphorylation (on 2nd position)
how does PFK2 "“make” F26BP
glucose-6-phosphate → 6-phosphogluconate (NADP+ →NADPH)
NADPH used in GSSG→ 2GSH reaction by glutathione reductase, and recycled back to NADP+
6-phosphoggluconate →ribulose-5-phosphate (-CO2, NADP+ →NADPH)
NADPH used in reductive biosynthesis of FA, stearols, etc…
list the steps of the pentose phosphate oxidative pathway from glucose-6-phosphate to ribulose5-phosphate
isomerase and epimerase
In the non oxidative phase, which two types of enzymes inter convert xyulose-5-phosphate and ribulose-5-phosphate?
transketolase (2) and transaldolase (3)
which two enzymes allow of the transfer of 2/3 carbons between sugar phosphates allowing rearrangemnet of 5C molecules to 6C molecules (glycolytic intermediates)?
3-7C
transketolase and transaldolase allow interconversion of sugars between ___C and ____C
(6) 5C sugars→ (5) 6C sugars
the net reaction in PPP is (6) 5C sugars→ ______
3 × 5C=15C
(rearrangements by transketolases and transaldolases)
result: 2 fructose-6-phosphate + 1 glyceraldehyde-3-phosphate
(net 6C+6C+3C=15C conserved)
starting from 1 ribulose-5-phosphate and 2 xyulose 5 phosphate molecules, what are products of PPP
gluconeogenic steps
final glyceraldehyde-3-phosphate can be converted into glucose-6-phosphate by
inhibited by NADPH (neg feedback as NADPH is produced in steps of pathway)
stimulated by NADP+ (pathway needs to make more NADPH)
glucose-6-phsophatase dehydrogenase (glucose-6-phosphate→ 6-phosphogluconolactone rxn in PPP) is inhibited by ______ and stimulated by _____
(regulated by redox stste of the cytosol)
a polymer of glucose stored for quick energy (have about a day supply)
what is glycogen?
regulate blood glucose
glycogen is metabolized in the muscle and liver by glycolysis, the liver also uses glycogen to __________
glucose-6-phosphate→glucose-1-phosphate (phosphoglucomutase)
(note glucose-1,6 phosphate intermediate)
glucose-1-phosphate +UTP→ UDP-glucose +PPi (UDP glucose pyrophosphatase)
the precursor of glycogen synthesis is UDP glucose, how is this produced from glucose-6-P?
activated sugar donor
UDP glucose is the __________ for glycogen synthesis to start
>4
for UDP-glucose to be added to a glycogen chain how long must the glycogen chain be
UDP-glucose + non-reducting end of glycogen chain→ UDP + longer glycogen chain
how does UDP glucose become part of glycogen?
the breakdown of glycogen
glycogen phsphorylase a + Pi cleaves off non reducing end and removes a glucose-1-phosphate from the glycogen
(reverse of glycogen synthesis)
what is glyconeogenesis? what enzyme is used?
reduce cellular osmotic pressure in cell to take in more glucose
why store glucose as glycogen?
to be the phosphate on glucose-1-phosphate and save energy (no ATP needed)
why is Pi used to cleave glucose-1-phosphate off from glycogen?
phsophorylation (makes glycogen synthase b- inactive)
gkycogen synthase kinase 3 (GSK3) is inactivated by _______
phsophorylation done by glycogen phosphorylase kinase b
glycogen phosphorylase is activated by ______ done by _______________ enzyme
dephosphorylation done by glycogen phosporylase a phosphatatse
glycogen phosphorylase is inactivated by ________ done by ____________ enzyme
PKA
glycogen phosphorylase kinase b is mediated by ______
phosphorylase a phosphatase
glycogen phsophorylase is a glucose sensor, glucose binding has an allosteric effcet on glycogen phsophorylase a and favours its dephosphorylation to glycogen phosphorylase b by enzyme _________ to produce glycogen phoshorylase b
enzyme that breaks down glycogen into glucose-1-phosphate
want it active when glucose is LOW, and inactive when glucose is already HIGH
what is glycogen phosphorylase? what it do and when u want it active>
inhibits catalytic activity and it exposes the phosphate groups on the serine residues, making them accessible. these accessible phosphate groups can the be better removed by protein phsophorylase 1 (making it dephsophorylated) now it becomes glycogen phosphorylase b.
how does glucose binding start shifting glycogen phsophorylase a to glycogen phsophroylase b?
inactivates (a→b)
high glucose ______ glycogen phosporylase
glycogen synthase a →glycogen synthase b ( by GSK3)
glycogen synthase b→ glycogen synthase a (by PP1)
glycogen synthase a →glycogen synthase b ( by ____________)
glycogen synthase b→ glycogen synthase a (by ____________)
glycogen phosphorylase a →glycogen phosphorylase b ( by PP1 )
glycogen phosphorylase b→ glycogen phosphorylase a (by phsophorylase kinase )
glycogen phosphorylase a →glycogen phosphorylase b ( by _________)
glycogen phosphorylase b→ glycogen phosphorylase a (by _________ )
glycogen phosphorylase active when: the cell needs energy (low glucose) and functions to break down glycogen
glycogen synthase active when: energy levels are high (high glucose) and functions to add glucose to store as glycogen
glycogen phosporylase is active when _____________ and functions to __________
glycogen sythase is active when _____________ and functions to __________
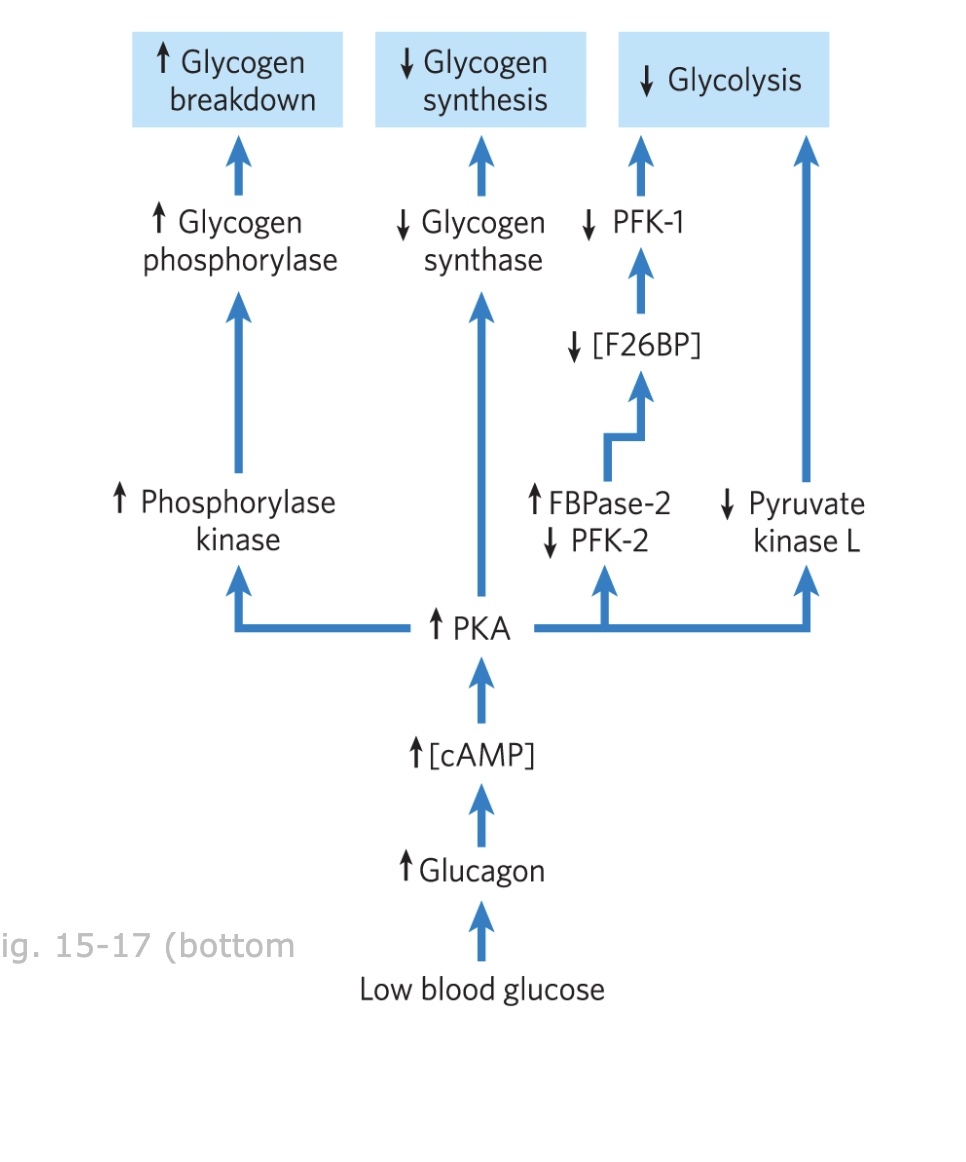
increase glycogen breakdown
decrrease glycolysis and glycogen synthesis
PKA regulation ensures that when blood sugar is low, the liver increases __________ and decreases ___________ and ____________ so that sugar can go to tissues that need it more
muscle: increased glycogenolysis and glycolysis (glycogen→pyruvate)
liver: increased glycogenolysis and gluconeogenesis, decreased glycolysis
what does epinephrine signal to occur in muscle?
what do epinephrine and gluacgon signal to occur in liver?
Myocytes lack glucagon receptors
Muscle pyruvate kinase is not phosphorylated by PKA
Muscle lacks gluconeogenic enzymes
Muscle lacks a key enzyme for glucose export
→ Muscle uses stored glycogen and glucose for itself
why does muscle not do gluconeogenesis? why are muscle and liver metabolisms different?
muscle: decreased glycogenolysis and increased glycolysis (pyruvate→glycogen)
liver: decreased glycogenolysis and gluconeogenesis, increased glycolysis
What does insulin signal to occur in muscle?
What do insulin (and low glucagon) signal to occur in liver?
In muscle:
Epinephrine says "use your glycogen for energy NOW" → glycogenolysis + glycolysis ↑
Insulin says "you have plenty of glucose, store it" → glycogen synthesis ↑, glycolysis ↓
In liver:
Epinephrine/glucagon say "make and release glucose into the blood" → glycogenolysis + gluconeogenesis ↑, glycolysis ↓
Insulin says "blood glucose is high, stop making more" → glycogenolysis + gluconeogenesis ↓, glycolysis ↑
The core logic:
Epinephrine = fight-or-flight → muscles need fuel fast, liver dumps glucose into blood
Glucagon = low blood sugar → liver makes glucose to raise blood levels back up
Insulin = high blood sugar → everyone take up glucose, store it, stop making it
summary to read: