Chapter 20 fr
1/8
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
9 Terms
COOH binded to alkane/
oic + acid at the end
ring + COOH
carboxylic acid at the end
Nitriles or C=/N
can either be attached to alkane or ring just add nitirle at the end
Carboxylic acid
more acidic then phenols and donate H+
low pKa
more acidic/ ewg increases acidity
making carboxylic acid
primary and secondary alkyl groups + KMnO4 leads to carboxylic acid/ primary alochols and aldehydes + CrO3/Nitrile and H3O leads to carboxylic acid/ Grignard + CO2
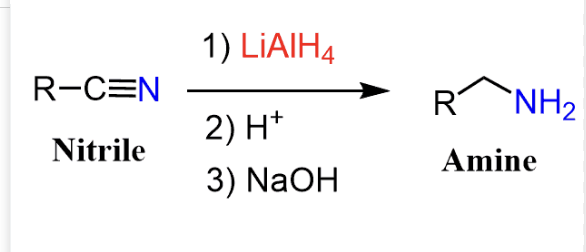
C=/N
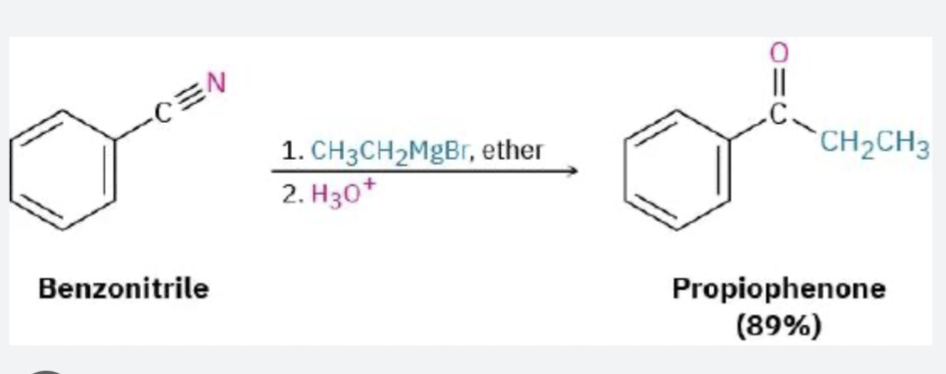
LiAlH4 →leads to amine → fully reduces → R–CH₂–NH₂ (amine)/ grignard leads to ketone Grignard (R–MgBr)
→ adds carbon → after workup → ketone
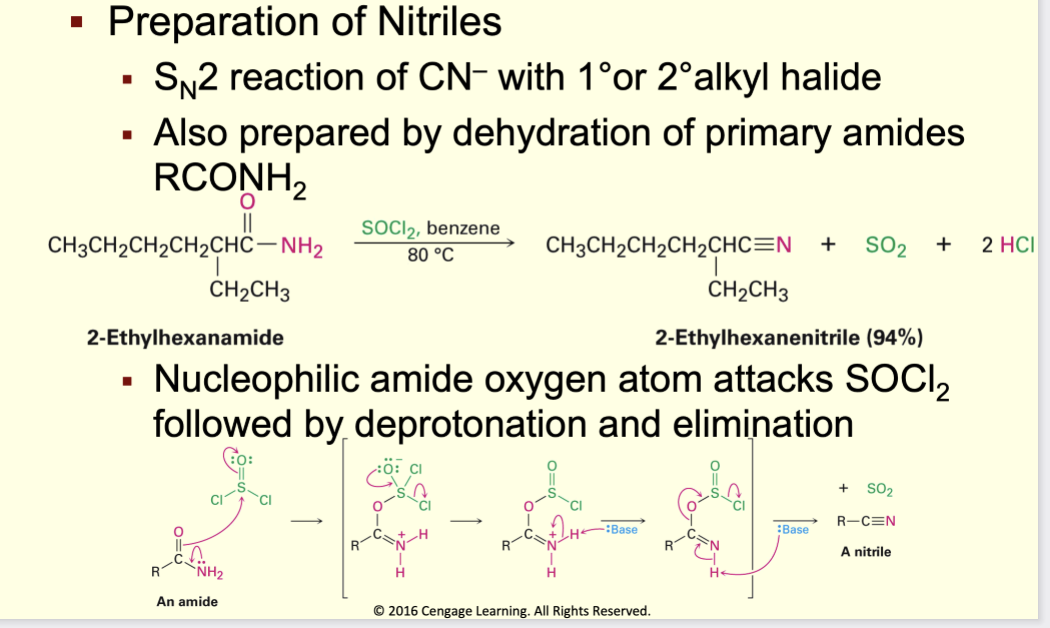
Prepartation on nitrile
sn2 reaction of cn with primary or secondary alkyl halideR–X + CN⁻ → R–C≡N (SN2, primary best)/ or primary amide being dehydratedR–CONH₂ + SOCl₂ → R–C≡N (dehydration)
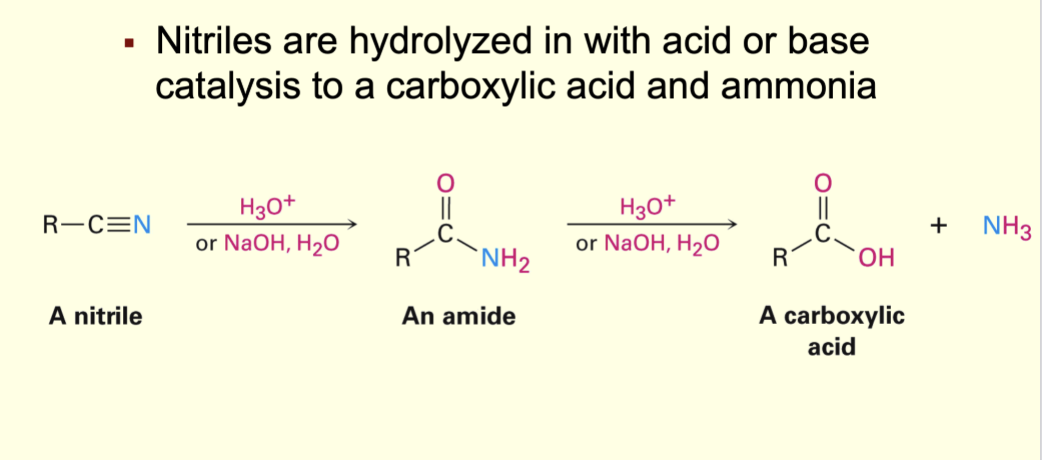
Nitriles to carboxylic acid
hydrolized with acid or base twice