Cell Exam 2 - Imported Set
1/453
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
454 Terms
What is the plasma membrane and what are the functions?
a protein-studded, fatty film so thin that it cannot be seen directly in the light microscope; a living cell is held together by this "container"
separate and protect its chemical components from the outside environment; cell communication, transport of small molecules, and energy generation
What is the structure of the plasma membrane?
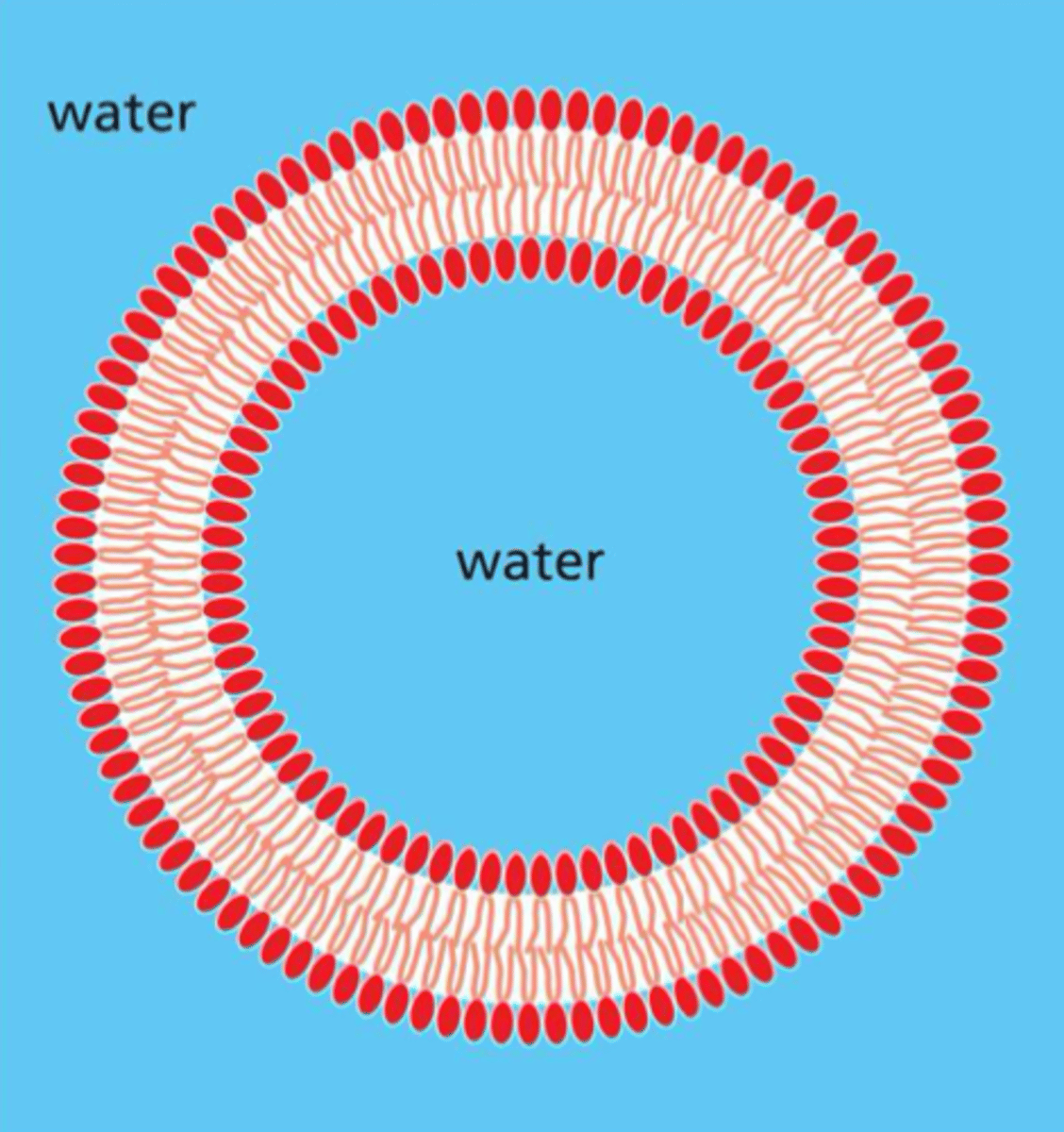
Two layers of phospholipids, with the hydrophobic tails pointing in and the hydrophilic tails pointing out about 5nm (or 50 atoms) thick with proteins inserted throughout
What would happen to the cell if it did not have a cell membrane?
There would be no cell --> --> --> no life
How many membranes do bacteria have?
one - plasma membrane
How many membranes do Eukaryotes have?
two - plasma membrane & internal membranes that enclose individual organelles
Which organelles have single membranes?
ER, golgi apparatus, endosomes, and mitochondria
Which organelles have double membranes?
nucleus and mitochondria
What does it mean to have a double membrane?
internal membrane that incloses our cellular compartments / various organelles
What are the functions of the double membrane?
- allow for organization
- transportation of H2O and other components
- barrier
Nutrients must pass inward and waste products must make their way out, how does the plasma membrane facilitate this?
the membrane is penetrated by highly selective channels and transporters -- proteins that allow specific, small molecules and ions to be imported and exported; other proteins act as receptors
What are receptors?
enable the cell to receive information about changes in its environment and reopens to them in appropriate ways
What happens when a cell grows? (in relation to its membrane)
its membrane also grows; it can deform without tearing allowing the cell to move or change shape
What happens when the cell is damaged?
it is self-healing
What does it mean when we say a lipid bilayer is flexible and fluid?
small lipids shift and move particles around which allows for the movement of proteins
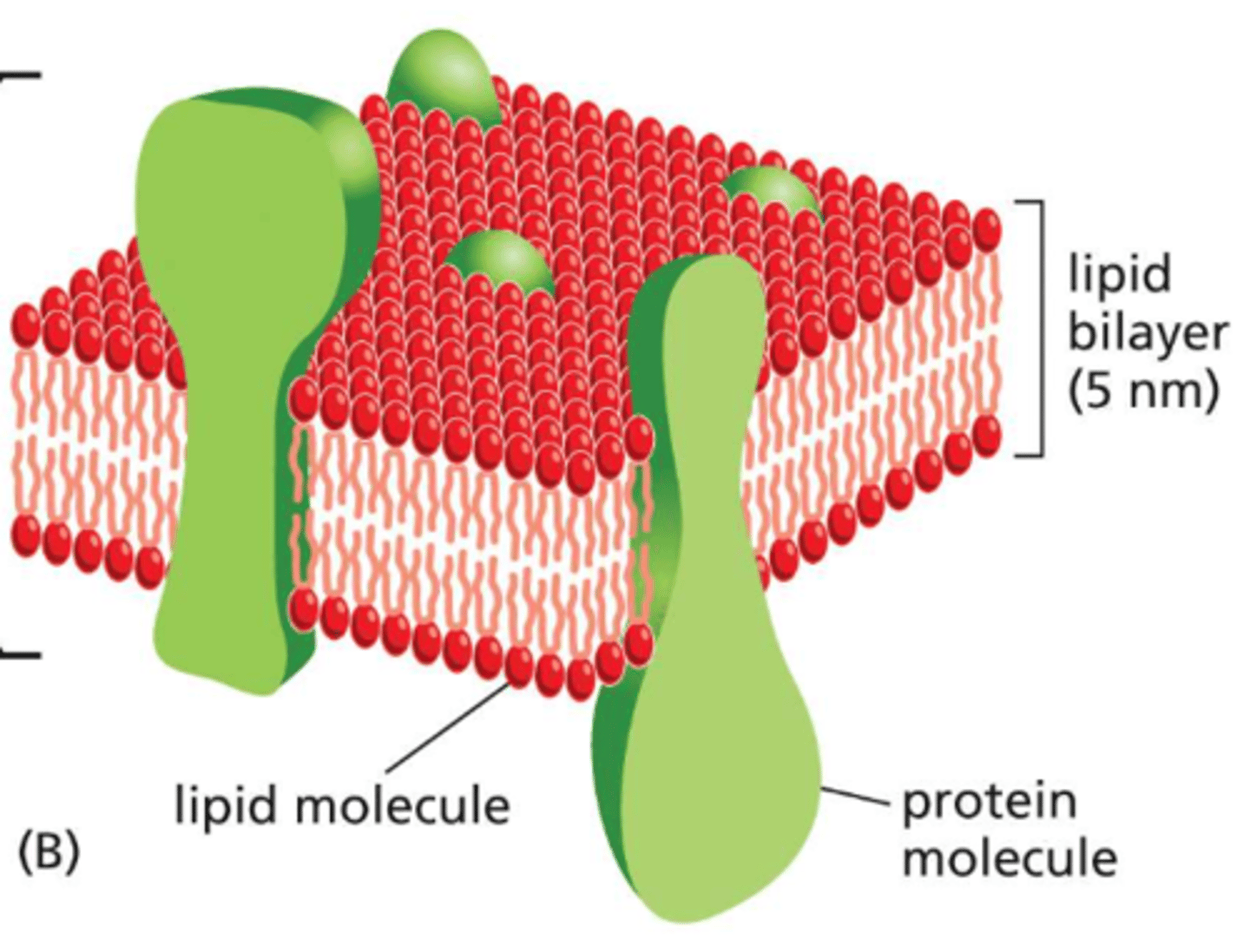
What are the functions of the lipid bilayer?
serves as a permeability barrier to most water-soluble molecules and hold proteins that carry out the other functions of the membrane and give different membranes their individual characteristics
How did scientists discover the structure of the lipid bilayer?
1925; they extracted lipids from plasma membranes of RBC and then they were spread out in a film on the surface of a trough filled with water. They used a movable barrier to push the floating lipids together until they formed a continuous sheet only one molecule thick. Then they measured the surface area of this monolayer which they found that it occupied twice the area of the original, intact cells.
So, they concluded that in an intact cell membrane, lipid molecules must double up to form a bilayer
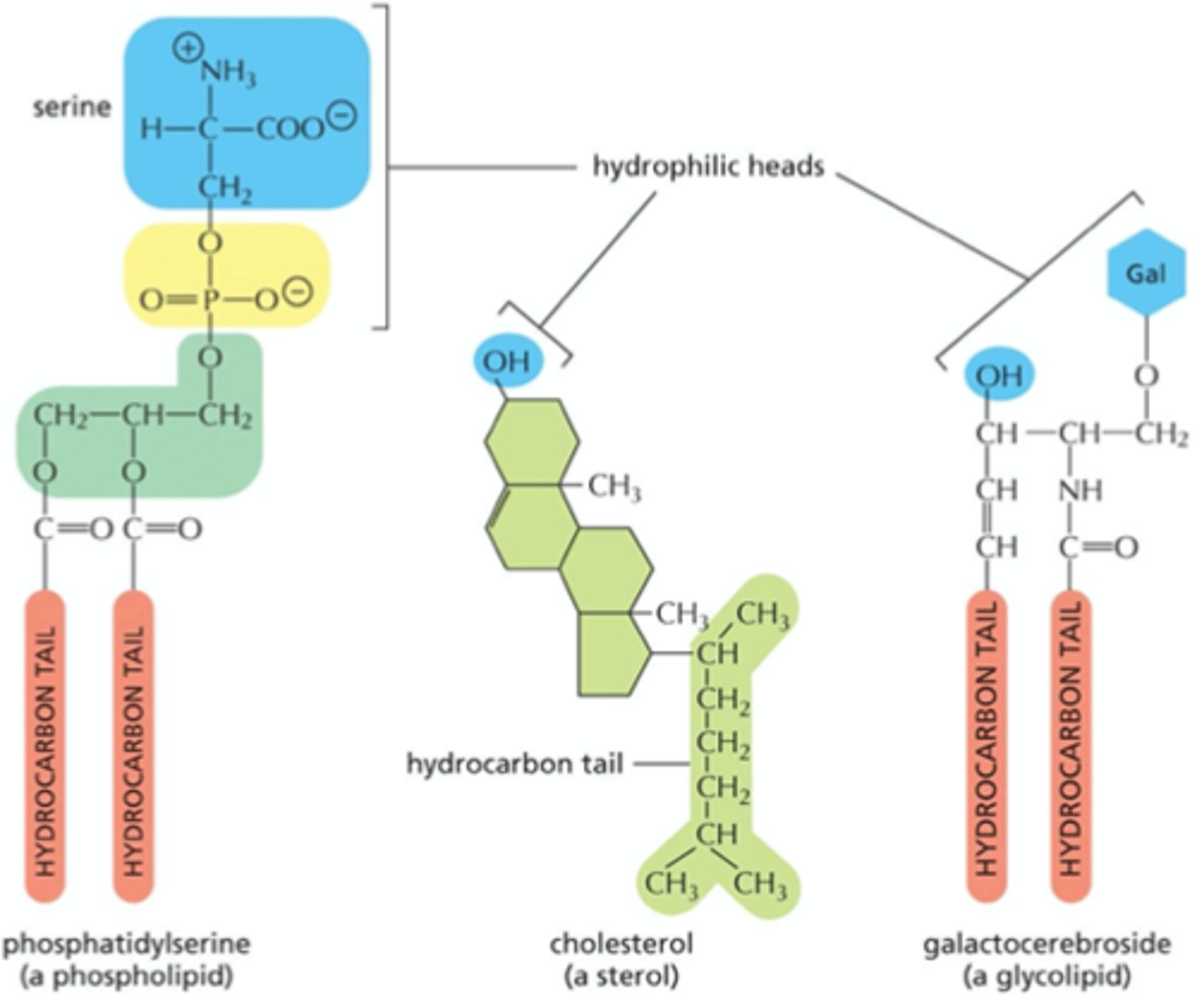
What does amphipathic mean?
having both hydrophilic and hydrophobic regions
What are the characteristics of the hydrophobic tail?
"water-fearing" -- non-polar; inside of the membrane to protect the tails from water, cant form Hydrogen bonds with water
What are the characteristics of the hydrophilic head?
"water-loving" -- polar; can for Hydrogen bonds with water
What are the most abundant lipids in cell membranes?
phospholipids
What is the general structure of a phospholipid?
phosphate-containing, hydrophilic head linked to a pair of hydrophobic, hydrocarbon tails
Membrane lipids are __________ and _________
phospholipids and amphipathic
What is the most abundant / common phospholipid?
phosphatidylcholine (choline attached to a phosphate group as its hydrophilic head)
What are two other membrane lipid that is also amphipathic?
cholesterol -- found in animal cell membranes
glycolipids -- have sugars as part of their hydrophilic head
What do the amphipathic membrane lipids encourage?
this drive lipid molecules to assemble into bilayers in an aqueous environment
List 3 examples of membrane lipids
- phosphatidylerine (a phospholipid)
- cholesterol (a sterol)
- galacerebroside (a glycolipid)
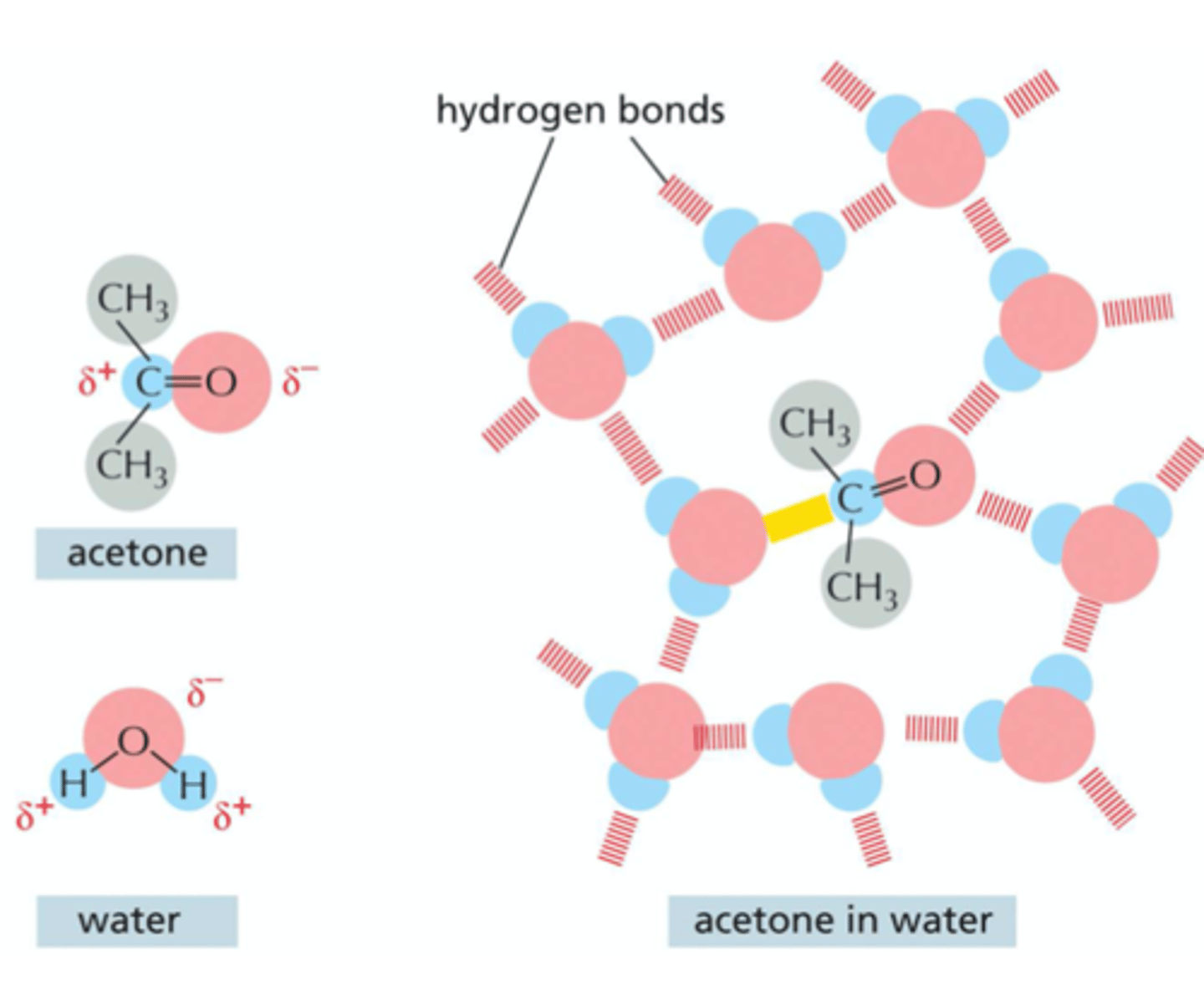
REVIEW: Why do hydrophilic molecules dissolve readily in water?
they contain either charged groups or uncharged polar groups that can form electrostatic attractions or hydrogen bonds with water molecules
What happens when a hydrophilic molecule attracts a water molecule? (such as acetone and water)
both acetone and water are polar molecules so acetone readily dissolves in water (polar atoms are shown in red and blue). Hydrogen bonds (red) and an electrostatic attraction (yellow) form between acetone and the surround water molecules. Non-polar groups are shown in gray
Why are hydrophobic molecules insoluble in water?
all -- or almost all -- of their atoms are uncharged and non-polar therefore they cant form favorable interactions with water molecules. instead they force adjacent water molecules to reorganize into a cavelike structure around them
Why does a cage-like structure require more free energy?
it is more highly ordered than the rest of water
How can this energy cost be lowered?
hydrophobic molecules can cluster together which limits their contacts with the surrounding water molecules. So this results in purely hydrophobic molecules coalesce into large fat droplets when dispersed in water (fat molecules are entirely hydrophobic)
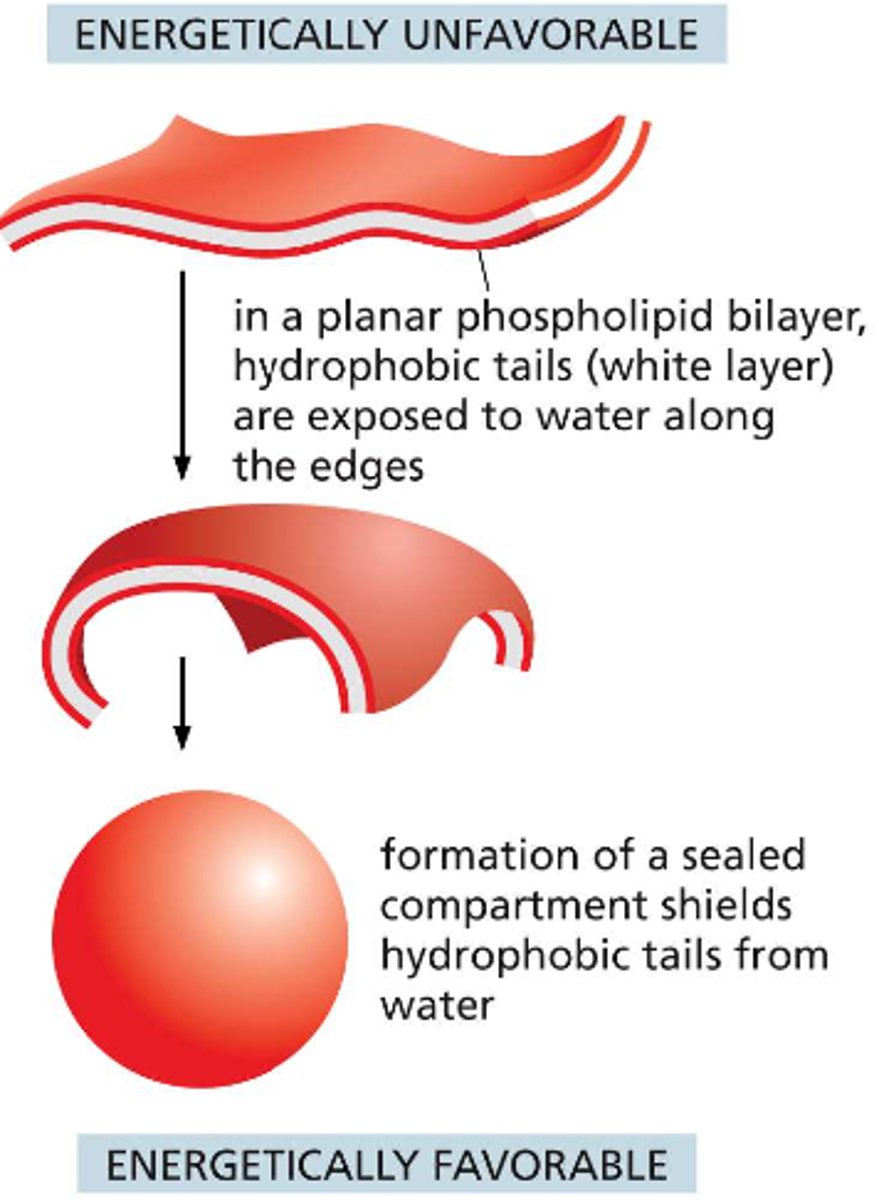
What happens to the lipid bilayer when a tear occurs?
this will create a free edge that is exposed to water, this is energetically unfavorable so the molecules of the bilayer will spontaneously rearrange to eliminate the free edge
if the tear is small: spontaneous rearrangement will exclude water molecules
if the tear is large: the sheet may begin to fold in on itself and break up into separate closed vesicles
How can an amphipathic sheet avoid having any free edges?
bend and seal to form a boundary around a closed space -- assemble into self-sealing containers that define a closed compartment (from vesicles and organelles to entire cells)
What does it mean when we say that the lipid bilayer is a flexible two-dimensional fluid
molecules within the bilayer are able to move about and change places with one another within the plane of the membrane; important for membrane function and integrity
flexibility is important for membrane function and it sets a lower limit of about 25 nm to the vesicle diameter that a cell membrane cane form
Pure phospholipids form closed, spherical vesicles called what?
liposomes
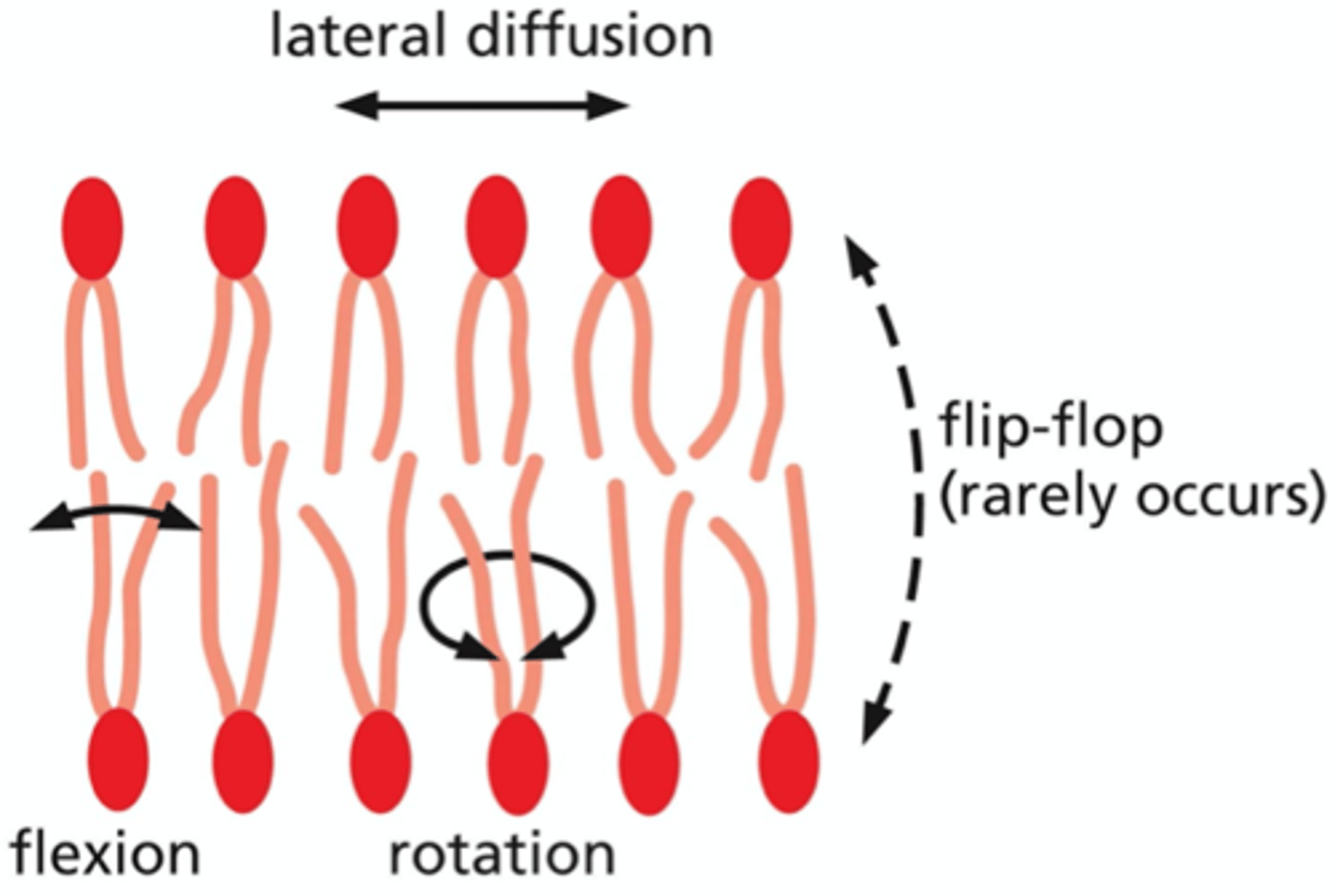
What are the four types of movement that occur within the lipid bilayer?
1. flip-flop -- very rare, requires proteins that facilitate this movement; occurs less than once a month
2. later diffusion -- random thermal motions; exchange with neighbors within the same monolayer, rapid
3. flexion -- flex / bend hydrocarbon tails
4. rotation -- rotate hydrocarbon tails about their long axis, reach speeds of 500 revolutions/second
What does it mean when we say the cell membrane has fluidity?
the ease with which its lipid molecules move within the plane of the bilayer
lipid molecules do not move spontaneously from one monolayer to the other
What does the fluidity of the cell membrane at a specific temperature depend on?
on its phospholipid composition and on the nature of the hydrocarbon tails: the closer and more regular the packing of the tails, the more viscous and less fluid the bilayer will be
What two major properties of hydrocarbon tails affect how tightly they pack in the bilayer?
their length and the number of double bonds they contain
a shorter chain length reduces the tendency of the hydrocarbon tails to interact with one another and therefore increases the fluidity of the bilayer
What is the length of the hydrocarbon tails of membrane phospholipids?
14-24 carbon atoms (18-20 atoms being the most common)
Describe the differences between the two hydrocarbon tails on a phospholipid?
one of these hydrocarbon tails contains only single bonds between its adjacent carbon atoms, whereas the other tail includes one or more double bonds
What does it mean when a hydrocarbon tail is unsaturated?
the chain that has the double bond(s) does not contain the maximum # of hydrogen atoms that could be attached to its carbon backbone
creates a small kink in the tail which makes it more difficult for the tails to pack against one another -- for this reason, lipid bilayers that contain a large proportion of unsaturated hydrocarbon tails are more fluid than those with lower proportions
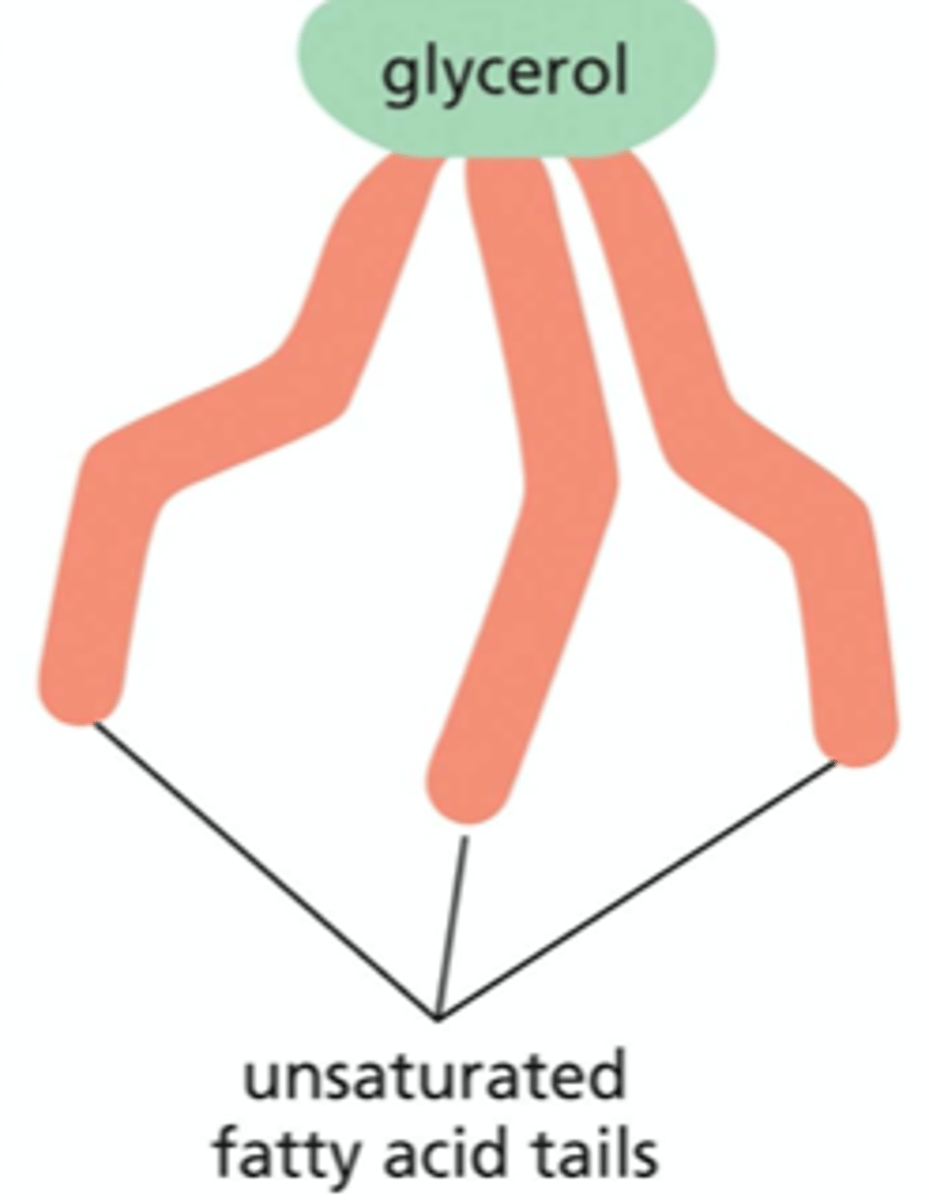
What does it mean when a hydrocarbon tail is saturated?
the chain with no double bonds has its full complement of hydrogen atoms

How do bacterial and yeast cells adapt to varying temperatures in relational to its lipid bilayer?
both the lengths and the degree of saturated of the hydrocarbon tails are adjusted constantly to maintain a membrane with a relatively consistent fluidity
at higher temps -- the cell makes membrane lipids with tails that are longer and that container fewer double bonds
How is membrane fluidity modulated in animal cells?
by the inclusion of the sterol: cholesterol
cholesterol is present in esp. large amounts in the plasma membrane, makes up ~20% of the lipids in the membrane by weight
with its short, rigid steroid ring structure, it can fill the spaces between neighboring phospholipids left by the kinks in their unsaturated hydrocarbon tails -- tends to stiffen the bilayer, making it less flexible AND less permeable
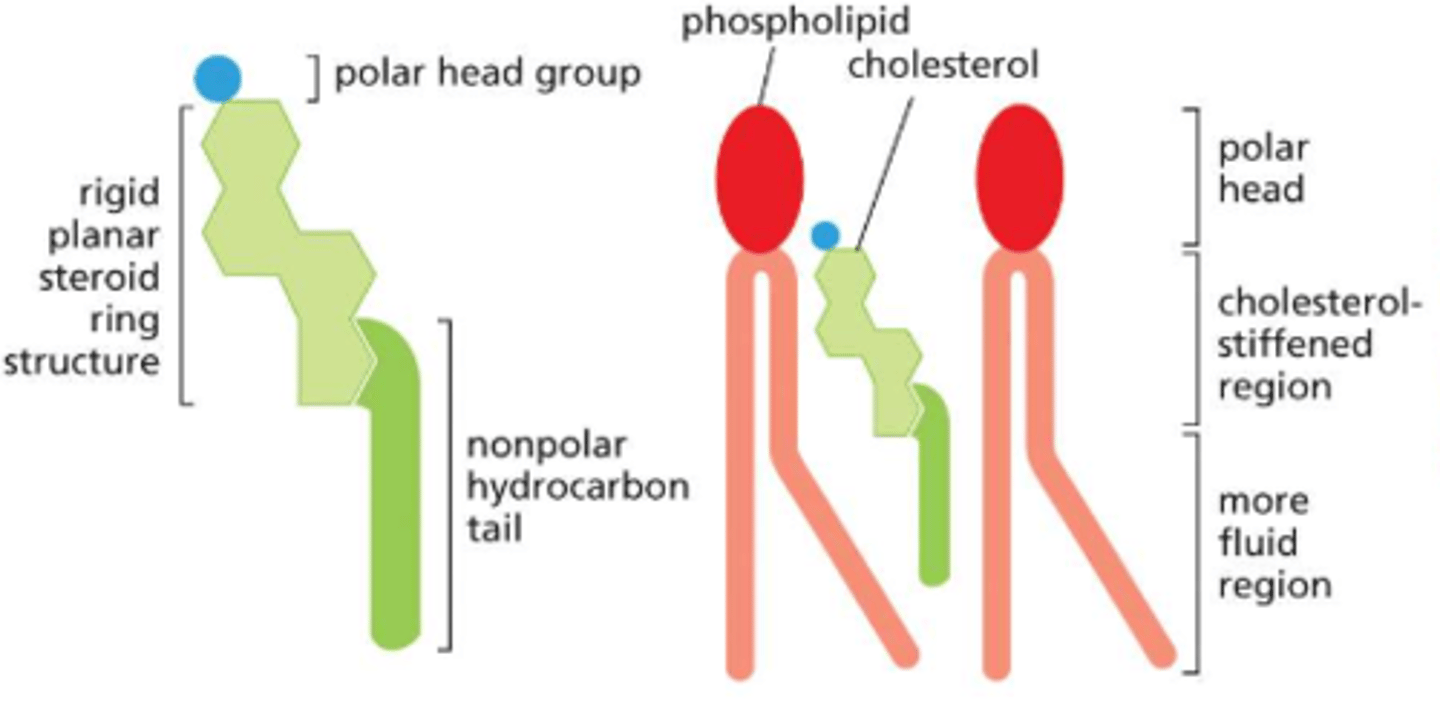
What are all the functions of fluidity within a cell?
enables membrane proteins to diffuse rapidly in the plane of bilayer and to interact with one another (ex: cell signaling)
permits membrane lipids and proteins to diffuse from sites where they are inserted into the bilayer after their synthesis to other regions of the cell
ensures that membrane molecules are distributed evenly between daughter cells with a cell divides
under appropriate conditions, it allows membranes to fuse with one another and mix their molecules
Membrane assembly begins where?
endoplasmic reticulum
new phospholipids are manufactured by enzymes bound to the cytosolic surface of the ER. using free fatty acids as substrates, these enzymes deposit the newly made phospholipids exclusively in the cytosolic half of the bilayer
this is distributed randomly throughout both sides of the bilayer, promotes symmetric growth
How does the cell membrane manage to grow evenly despite the unbalanced add-on of newly made phospholipids?
phospholipid transfers are catalyzed by a scramblase -- a type of transport protein that removed randomly selected phospholipids from one half of the bilayer and inserts them in the other half. this results in an even distribution between each monolayer of the ER
some of this newly synthesized membrane will remain in the ER and the rest will be used to supply fresh membrane to other compartments of the cell (including the golgi apparatus and the plasma membrane)
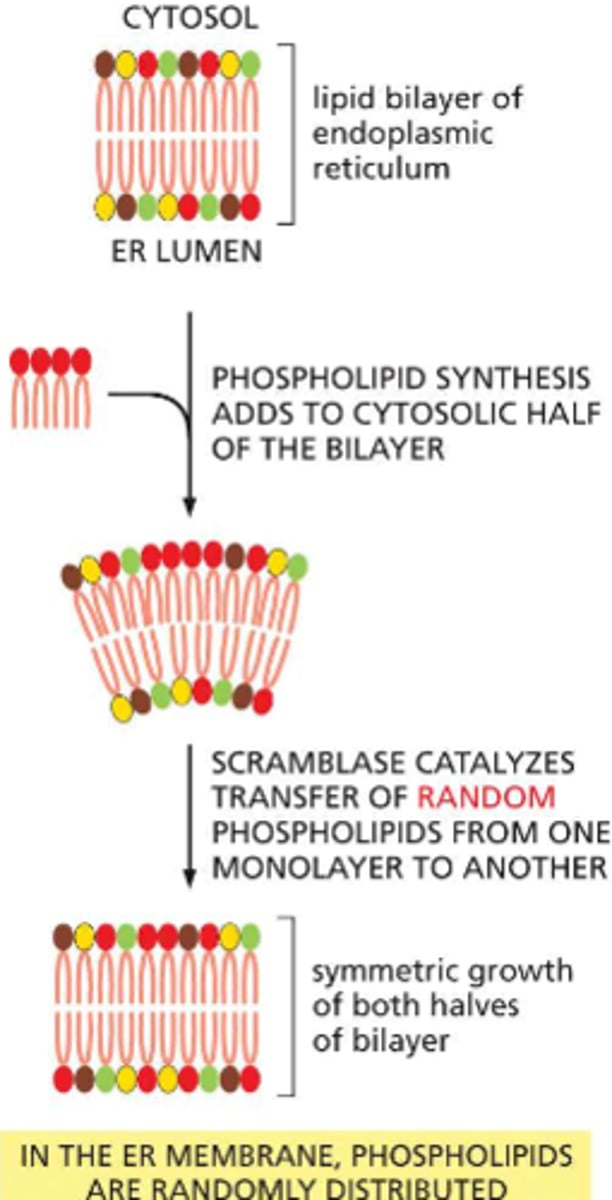
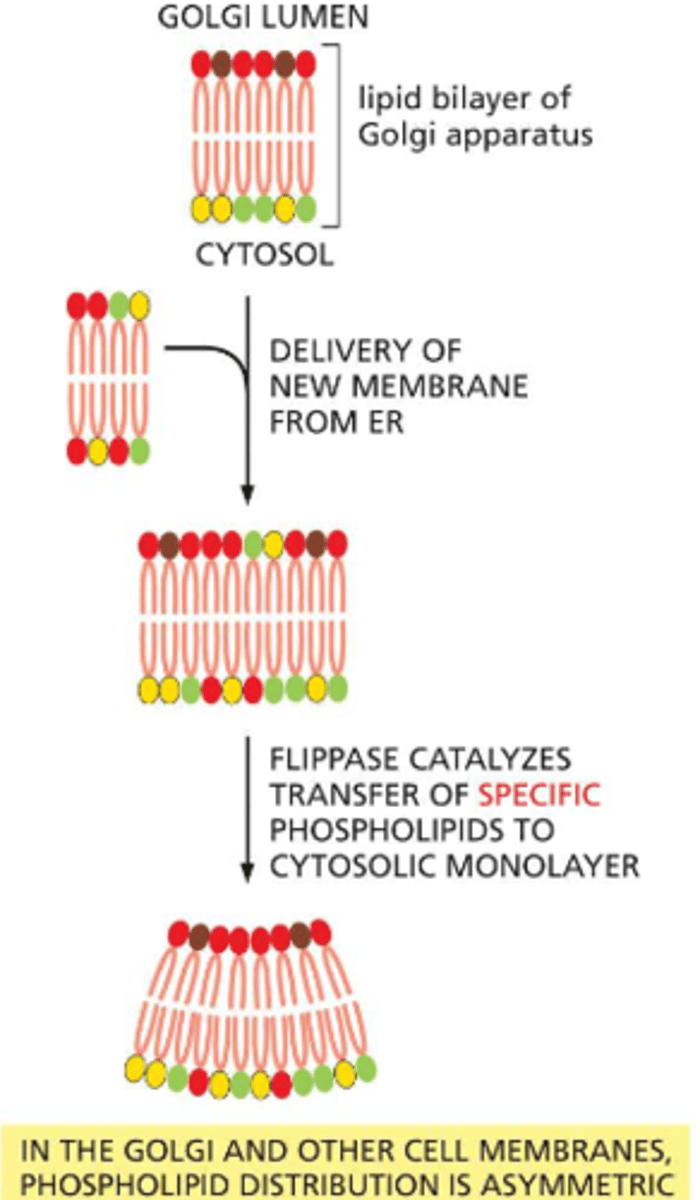
If membranes emerge from the ER with an evenly assorted set of phospholipids, why are most cells asymmetric: the two halves of the bilayer often include noticeably different sets of phospholipids?
the golgi membrane contains flippases -- these move random phospholipids from one half the bilayer to the other, this removed specific phospholipids from the side of the bilayer facing the exterior space and flip them into the monolayer that faces the cytosol
this may help drive subsequent vesicle budding
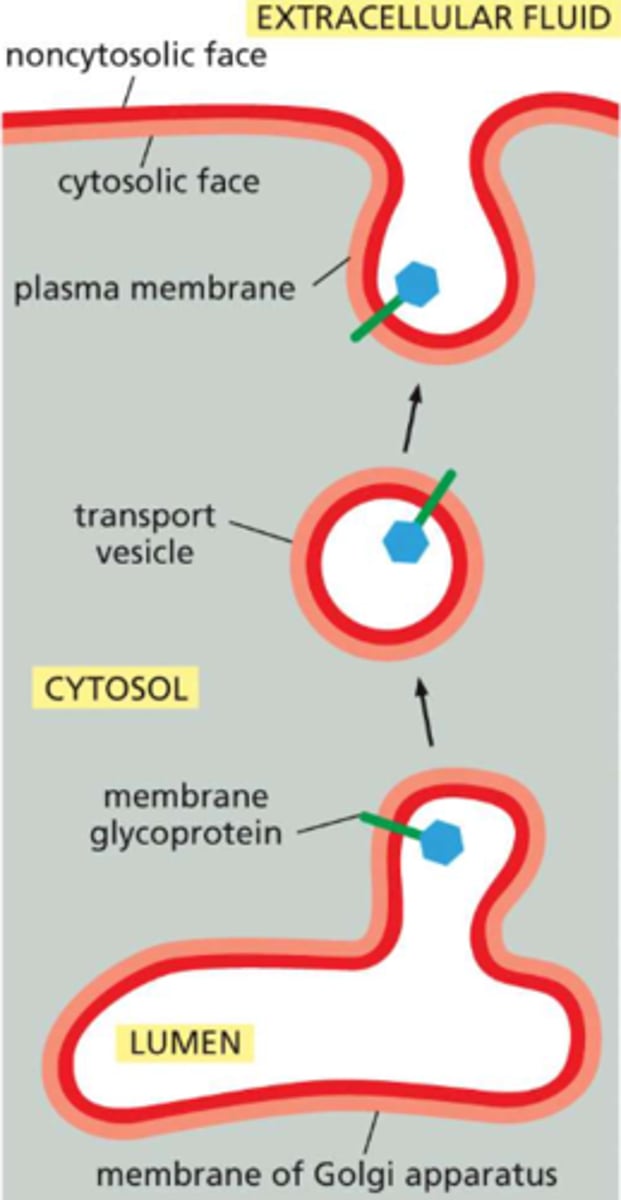
All cell membranes have distinct "inside" and "outside" faces
the cytosolic monolayer always faces the cytosol
the non-cytosolic monolayer is exposed to either the cell exterior (in the case of the plasma membrane) or the interior space (lumen) of an organelle
this applies to phospholipids that make up the membrane AND any proteins within the plane
Which lipid shows the most dramatically lopsided distribution in a cell?
glycolipids -- located mainly in the plasma membrane and only in the non-cytosolic half of the bilayer
the sugar groups face the cell exterior, where they form part of a continuous coat of carbohydrate that surrounds and protects the animal cell -- acquire these sugars in the golgi. there are no flippases that transfer glycolipids to the cytosolic side so when it is finally delivered to the plasma membrane, it displays sugars to the exterior of the cell
Other lipids show different types of asymmetric distributions which relate to specific functions
components that need to be housed on a certain side of the bilayer
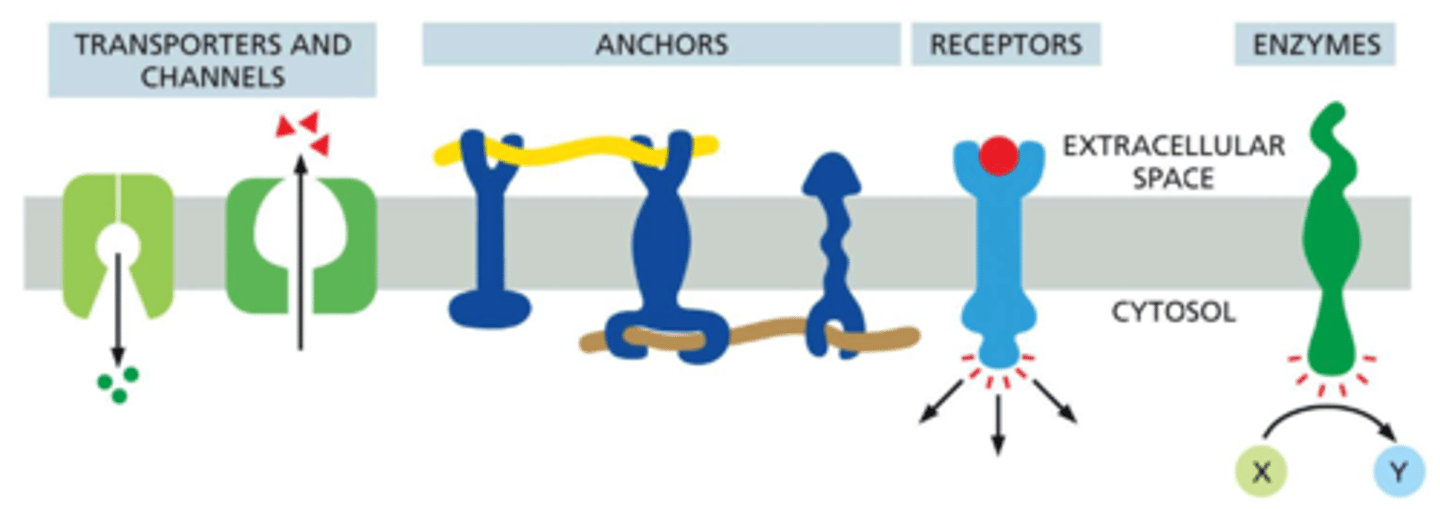
What are the functions of plasma membrane proteins?
transport molecules -- Na+ pump: actively pumps Na+ out of cells and K+ in
transport ions -- K+ leak channel: allows K+ ions to leave cells, thereby influencing cell excitability
act as anchors -- ingrains: link intracellular matrix filaments to extracellular matrix proteins
detect signals / receptors -- platelet-derived growth factor (PDGF) receptor: binds extracellular PDGF and (as a consequence) generates intracellular signals that direct the cell to grow and divide
catalyze reactions / enzymes -- adenylyl cyclase: catalyzes the production of the small intracellular signaling molecule cyclic AMP in response to extracellular signals
In animals, what percentage of proteins constitute the mass of most plasma membranes, the remainder being lipid plus the small amounts of carbs found on some of the lipids (glycolipids) and many proteins (glycoproteins)
50%
lipid molecules are much smaller than proteins but a cell membrane typically contains about 50 times the number of lipid molecules compared to protein molecules
What are the four ways membrane proteins can interact with a cell membrane?
1. transmembrane -- proteins that extend through the bilayer, with part of their mass on either side; amphipathic with hydrophobic regions on the interior and hydrophilic tails exposed to the aqueous environment on either side of the membrane. Can have just one a-helices, multiple a-helices, or B-sheets/barrels
2. Monolayer-associated -- located almost entirely in the cytosol and are associated with the cytosolic half of the lipid bilayer by an amphipathic a-helix exposed on the surface of the protein
3. Lipid-linked -- proteins that lie entirely outside the bilayer, on one side or the other, attached to the membrane by one or more covalently attached lipid groups
4. protein-attached proteins that are bound indirectly to one face of the membrane or the other, held in place only by their interactions with other membrane proteins
What does it mean to be an integral membrane protein?
transmembrane, monolayer-associated, or lipid linked -- can be removed only by disrupting the bilayer with detergents
What does it mean to be a peripheral membrane protein?
protein-attached -- can be released from the membrane by more gently extraction procedures that interfere with protein-protein interactions but leave the lipid bilayer intact
Peroxidase
a. is found at high concentrations in cytoplasm
b. strengthens the cell wall of plants
c. strengthens the cell wall of plants and is used as a antimicrobial agent
d. can be used by cells as an antimicrobial agent
c. strengthens the cell wall of plants and is used as a antimicrobial agent
Fixation
a. increases the permeability of the cell to dyes and stains
b. increases the permeability and alters the structure of the cell wall
c. alters the structure of the cell wall
d. helps the cells stick to the slides
e. increases the permeability and helps the cells stick to the slide
e. increases the permeability and helps the cells stick to the slide
The artificial substrate of peroxidase
a. chloronaphthol
b. water
c. nitrophenyl phosphate
d. hydrogen ions
e. hydrogen peroxide
a. chloronaphthol
__________ is used to locate high concentrations of electrons by producing a red color.
a. methylene blue
b. peroxidase
c. janus green
d. tetrazolium
e. iodine
d. tetrazolium
_______ is used to locate starch.
a. iodine
b. janus green
c. peroxidase
d. methylene blue
e. tetrazolium
a. iodine
_____ is the chemical we use as a fixative.
a. water
b. acetone
c. ethanol
d. methanol
e. hydrochloric acid
c. ethanol
Methylene blue stains the _________; eosin stains the __________.
a. nucleus; cytoplasm
b. cytoplasm; nucleus
c. cytoplasm; cytoplasm
d. nucleus; nucleus
a. nucleus; cytoplasm
Bird mature erythrocytes lack ______.
a. hemoglobin
b. mitochondria
c. nuclei and mitochondria
d. nuclei
b. mitochondria
Mammalian mature erythrocytes lack ______.
a. mitochondria
b. nuclei
c. hemoglobin
d. nuclei and mitochondria
d. nuclei and mitochondria
The enzyme used in the kinetics lab this week is
a. alkaline phosphatase
b. nitrophenyl phosphate
c. potassium hydroxide
d. nitophenyl
e. acid phosphatase
e. acid phosphatase
The substrate used in the kinetics lab is
a. alkaline phosphatase
b. nitrophenyl phosphate
c. potassium hydroxide
d. nitophenyl
e. acid phosphatase
b. nitrophenyl phosphate
Product generated in kinetics lab
a. nitrophenol
b. acid phosphate
c. peroxide
d. nitrophenyl phosphate
e. potassium hydroxide
a. nitrophenol
Which graph starts off curved and then levels off after a specific x value?
a. lineweaver burk
b. standard curve and lineweaver burk
c. michaelis menten
d. standard curve and michaelis menten
e. standard curve
c. michaelis menten
Which of the following will have an effect on the reaction rate of an enzyme?
a. amount of enzyme
b. temp, ph, and amounts of enzyme and substrate
c. amount of substrate
d. ph
e. temp and ph
f. temp
b. temp, ph, and amounts of enzyme and substrate
This chemical disrupts the plasma membrane of the wheat germ
a. dawn dishwashing detergent
b. SDS
c. NP-40
d. potassium hydroxide
e. salt
c. NP-40
Purpose of potassium hydroxide added to the reactants
a. denature the enzyme and produce color
b. raise the temp of solution by interacting with the water in test tube
c. serve as substrate
d. denature the enzyme to stop the reaction
e. produce the color
a. denature the enzyme and produce color
A __________ is a protein or glycoprotein that is not produced by the immune system that will bind to carb chains on the outside of a cell.
a. cell receptor protein
b. antibody
c. lectin
d. transmembrane protein
e. immunoglobin
c. lectin
In the presence of buffer and hydrogen peroxide, the enzyme peroxidase converts ___________ from a colorless soluble substrate to an insoluble purple product, allowing us to localize the protein of interest in todays lab.
a. tris buffer
b. bromothymol blue
c. agglutinin
d. chloronaphthol
e. concanacalin A
d. chloronaphthol
Lectin will only react with the correct __________________.
a. carb chain which is part of a glycoprotein or glycolipid
b. amino acid sequence found on the outer surface of the cell
c. phospholipid on the inner surface of the plasma membrane
d. phospholipid on the outer surface of the plasma membrane
a. carb chain which is part of a glycoprotein or glycolipid
The chemical used to fix our cheek cells is__________ which causes the cells to stick to the slides.
a. ethanol
b. acetic acid
c. methanol
d. water
a. ethanol
Agglutination occurs because _______ have more than one site than can participate in binding at a single time.
a. neither red blood cells nor lectins
b. red blood cells
c. lectins
d. red blood cells and lectins
d. red blood cells and lectins
By flooding the reaction with a high concentration of sugar, we are
a. occupying all of the sites on the lectin so it can no longer bind to the recpetor
b. occupying all of the sites on the red blood cells and cheek cells so they can no longer bind the lectin
c. occupying all of the sites on the lectins, red d cells, and cheek cells so they can no longer bind
a. occupying all of the sites on the lectin so it can no longer bind to the recpetor
What two areas of the corn kernel show peroxidase activity and why?
Outer ring and embryo
- protect corn kernel from outside materials
- helps in synthesis of plant cell wall
- strengthens cell wall
Where is peroxidase found in the onion cell and why?
cell wall
- protects substance from other materials and helps in the synthesis and strengthening of the cell wall.
What molecule does iodine react with and why?
Starch, without the starch the embryo would not have enough energy to grow.
What region of the chicken erythrocyte contains the highest concentrations of LDH activity and does this make sense?
Cytoplasm, because LDH is involved in fermentation in the cytoplasm.
What does a lack of purple color and lack of clumping tell us which sugar the lectin binds?
Lack of Purple/Clumping shows which sugar conA was bound to, no purple or clumping means the ConA did not bind to the cell membranes it was bound to the sugar, which means mannose was bound to the ConA.
How much clumping would occur if a high concentration of fructose was added to the red blood cell suspension?
A lot of clumping, because the ConA does not bind to fructose therefore it binds to the cell membranes, which will clump the cells together.
What are the solutions used to detect peroxidase?
What are the solutions used to detect lactate dehydrogenase activity?
What are the solutions that detect respiration?
What does methylene blue & toluidine stain?
nuclei
What does eosin stain?
cytoplasm
What does janus green satin?
mitochondria
How can an enzyme be detected using cytochemistry?
by a specific substrate reaction resulting in a visible marker
What is Vmax?
the maximum rate of reaction -- when the enzyme is saturated with substrate
What is Km?
Substrate concentration at 1/2 Vmax
Difference between lectins and antibodies
Lectins specifically attach to carbohydrates
Antibodies develop against proteins
Lectin
A protein that binds a carbohydrate
- causes cells to clump or stick together
Hemagglutination
clumping of red blood cells
How can you tell if someone is A positive?
If there is clumping in the A and Rh
What if there is no clumping at all?
O negative
) Saturation can be defined as
A) denaturation of an enzyme.
B) the inability to increase reaction velocity beyond a finite upper limit.
C) inhibition of enzyme function by blocking the active site.
D) the substrate concentration at which velocity reaches one-half maximum velocity.
E) a characteristic of all uncatalyzed reactions.
B) the inability to increase reaction velocity beyond a finite upper limit.