Chemistry Exam 2
1/19
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
Lindlar’s catalyst does not work when you start with an alkene
determining whether syn or anti dihydroxylation
Syn products (cis-diols) are formed with OsO4 or KMnO4 , while anti products (trans-diols) arise from peroxyacids (RCO3H) followed by acid hydrolysis (H3O+)
O-O oxidizing agents go…
Anti (add the OHs on opposite sides)
Metal-O oxidizing agents (KMnO₄ and OsO4) for dihydroxylations go…
syn (add the OHs on the same side)
Examples of O-O oxidizing agents
O2, O3, H2O2, peroxyacids such as m-CPBA
Examples of Metal-O oxidizing agents
NaCr2O7, CrO3, PCC, KMnO4, OsO4, Ag2O
In dihydroxylation, KMnO₄ and OsO4 will go…
syn
In dihydroxylation, peroxyacids (RCO3H) will go
anti
KMnO4 in NaOH/H2O with heat is great for synthesizing…
ketones, acids, and CO2
this is called oxidative cleavage of alkenes
any singular hydrogen will be turned into an alcohol
if one side of the alkene has two hydrogens on it, it results in CO2
KMnO4 in NaOH/H2O (has water) when the reaction is cold will get you…
Syn dihydroxylation (just like OsO4)
remember plus enantiomer
A STRONG oxidizing agent in water (both have to be in the same step for the reagents) will lead to oxidative cleavage. you also need heat.
strong oxidizing agents include…
OsO4, KMnO4, Ag2O
Cleaving alkynes always yields…
carboxylic acids and CO2 EVEN when you use O3
A terminal alkyne is what would make CO2
Oxidations of alcohols can only go…
on primary or secondary alcohols (because tertiary has no H to remove)
primary alcohols will yield a carboxylic acid
secondary alcohols will yield a ketone
The reagents you need to oxidize an alcohol are…
NaCr2O7 or CrO3 with aq H2SO4
it will yield ketones and acids
if you want an aldehyde instead of an acid when oxidizing a primary alcohol, go with milder conditions such as…
PCC in CH2Cl2
Tollen’s reagent (Ag2O / NH4OH) can only oxidize…
pi bonds… not alcohols. So NR in terms of oxidation of an alcohol
Swern Oxidation is just like…
PCC, it is mild and can yield aldehydes
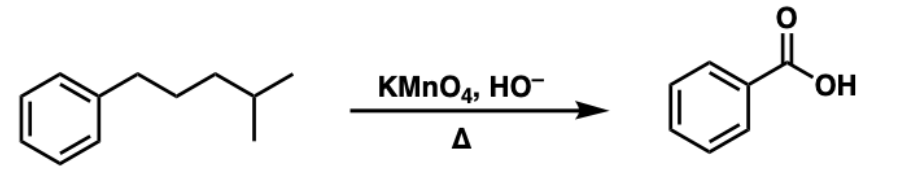
when you have a strong oxidizer then acid on an aromatic compound, you will always make…
benzoic acid with the side chain NO MATTER what the side chain is (it gets cleaved down to benzoic acid)
A good Nu will attack a neutral C=O, but a poor Nu can’t so you must…
protonate the O
(because of resonance, the positive charge can be on the carbon. all of this is in terms of the Nu wanting to attack a positive (or partially positive) carbon because that means it is a good electrophile)
Reduction of aldehydes and ketones requires…
a strong reducing agent (H:) which comes from NaBH4 or LAH