Organic Chemistry Reactions and Mechanisms
1/30
Earn XP
Description and Tags
Flashcards covering key organic chemistry terms, reactions, and mechanisms from the lecture notes.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
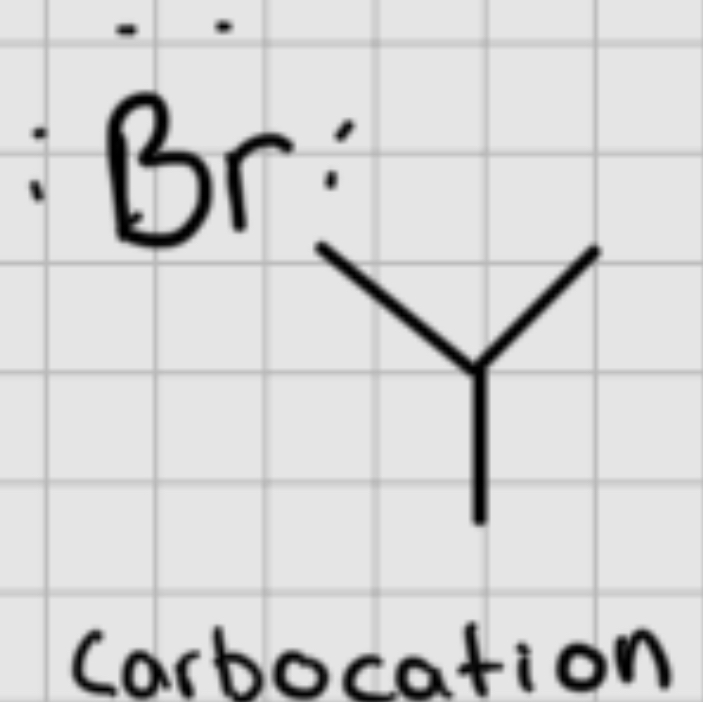
Carbocation
A positively charged carbon atom that is an intermediate in chemical reactions.
Syn addition
A type of addition reaction where the new substituents are added to the same side of the double bond.
Trans addition
A type of addition reaction where the new substituents are added to opposite sides of the double bond.
Hydroboration-Oxidation
A two-step reaction process where alkenes are converted to alcohols, using BH₃ and H₂O₂.
Ketone
A functional group characterized by a carbonyl (C=O) group bonded to two carbon atoms.
Aldehyde
A functional group consisting of a carbonyl (C=O) group bonded to at least one hydrogen atom.
Lindlar Catalyst
A catalyst used in hydrogenation reactions to selectively reduce alkynes to cis-alkenes.
NaNH₂
Sodium amide, a strong base often used in organic synthesis for deprotonation and as a nucleophile.
H₂O
Water, often used as a solvent or reactant in chemical reactions.
H₂
Hydrogen gas, commonly used as a reducing agent in organic reactions.

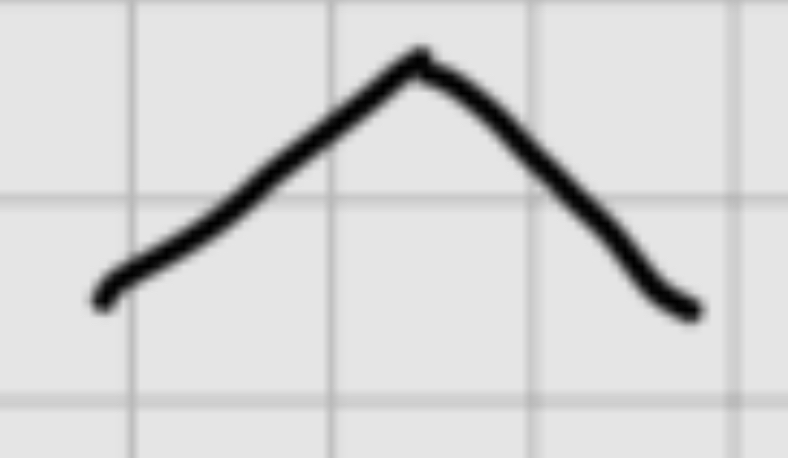
When adding :
H+
——
H20


When adding :
H2
—————
Pt/ Pd/ Ni
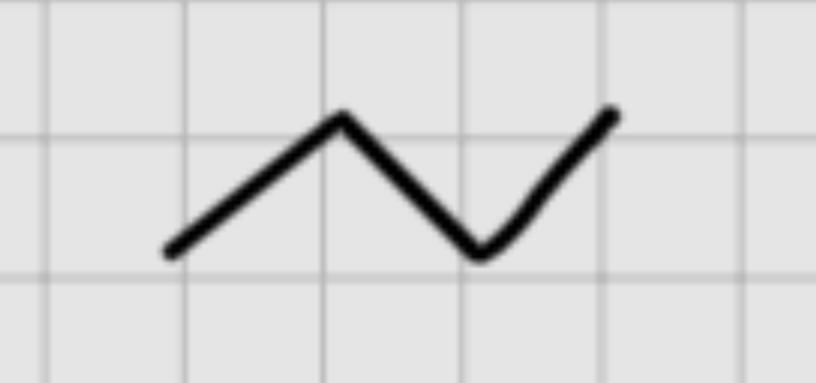

When adding :
1. NaNH2
——————
2. CH3-Br


When adding :
H2
———-
Lindlar
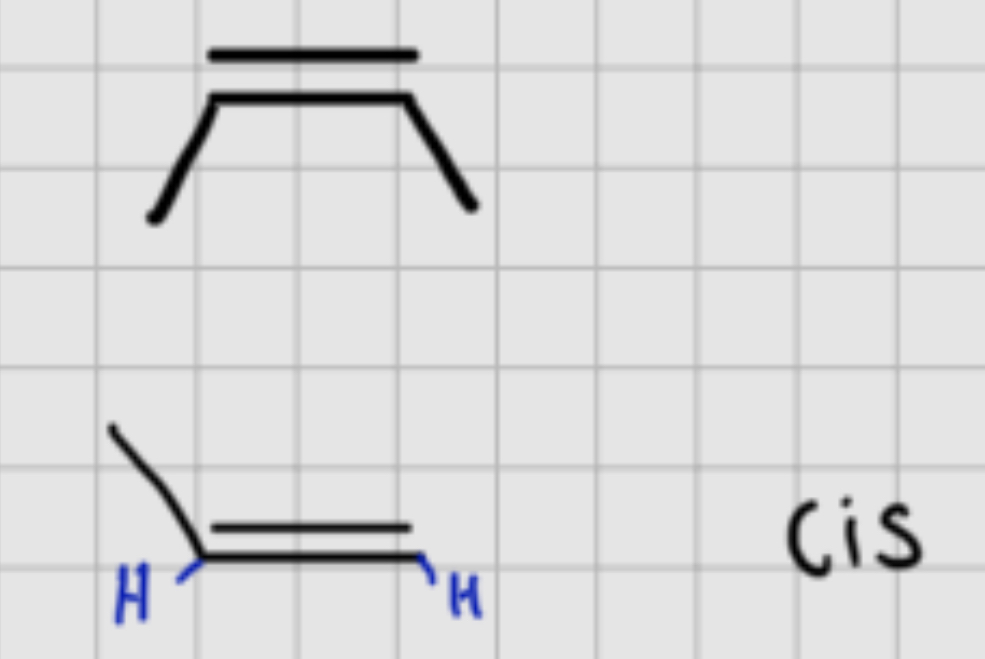

When adding :
H2M
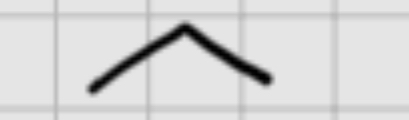

When adding:
1.(sia)2BH
———————
2.H2O2,NaOH
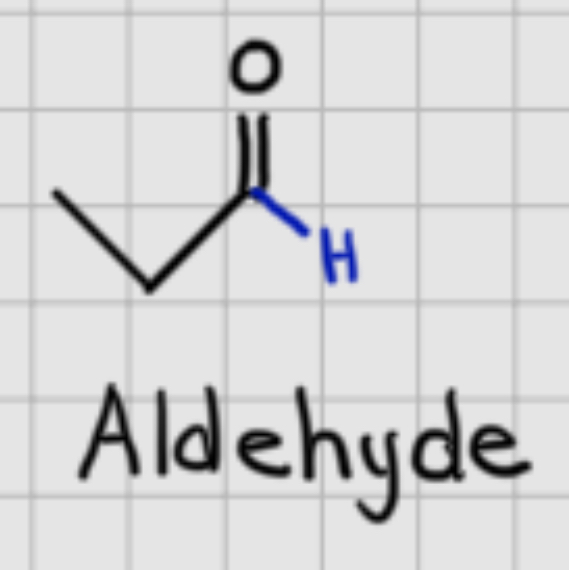

When adding:
2Na\omicron (not oxygen but exponent zero)
———
2NH3
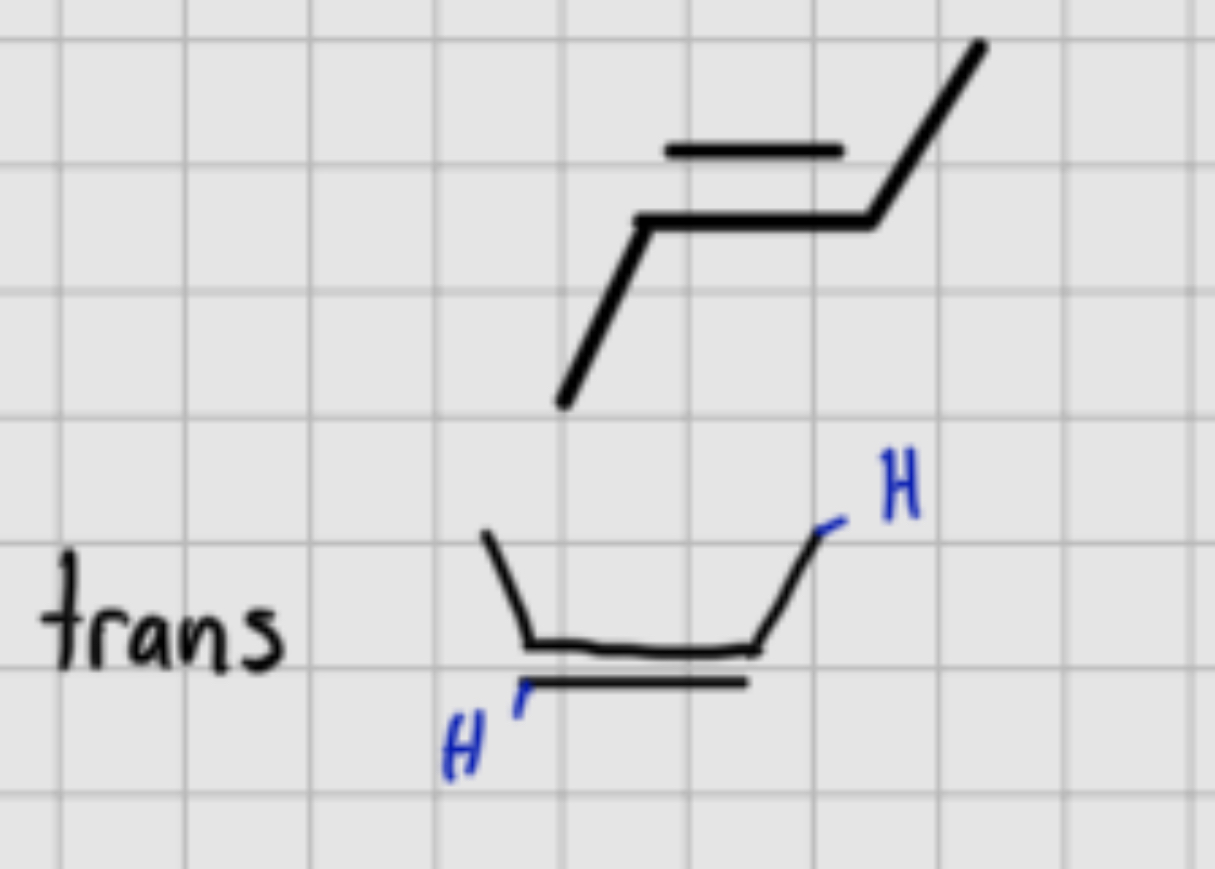

When adding:
1.Br2
————-
2. 3NaNH3
3. H2O
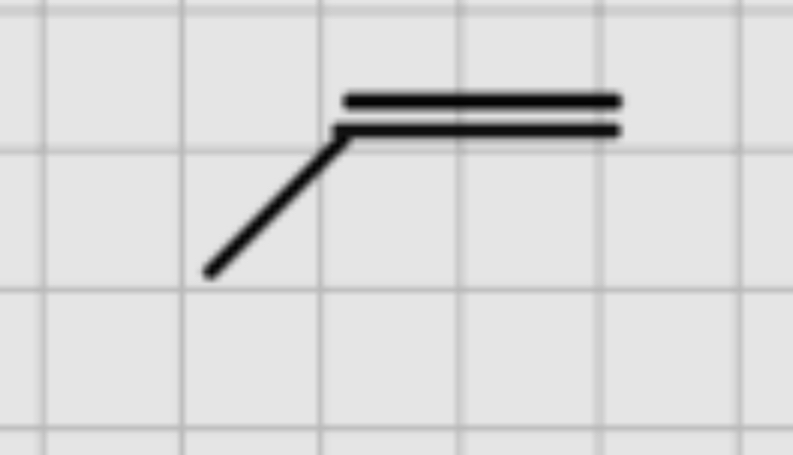

When adding:
1.BH3………….H20
————- OR ———
2. H2O2………H2SO4
NaOH
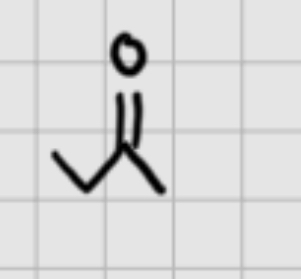
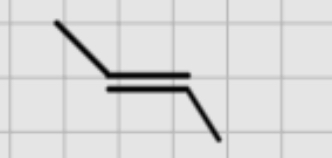
Now we want a alkyne . We have this what do we add.
1.Br2
—————
2. NaNH2


Now we want a alkyne. We have this what do we add.
1.NaNH2
————
2. CH3-Br

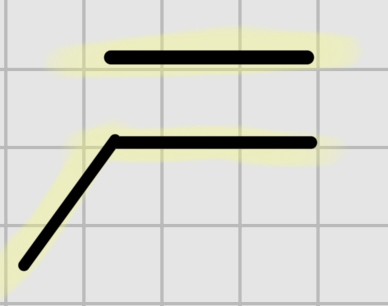
We add:
Br2
It’s trans addition.
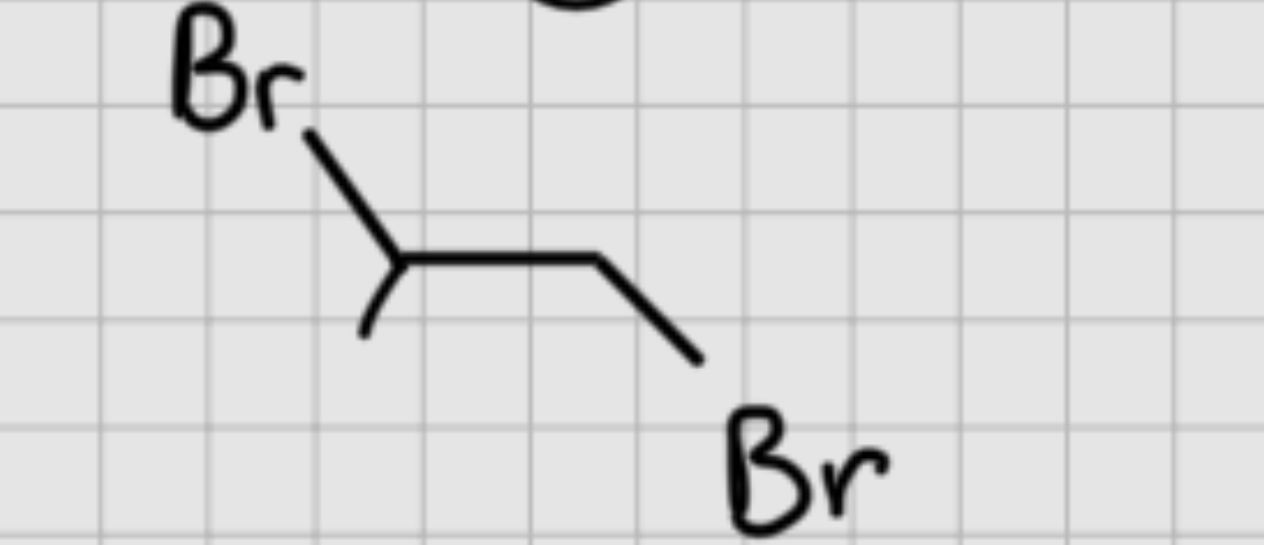
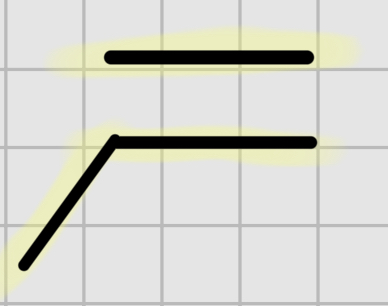
We add:
Br2
——
H20
It’s trans addition.
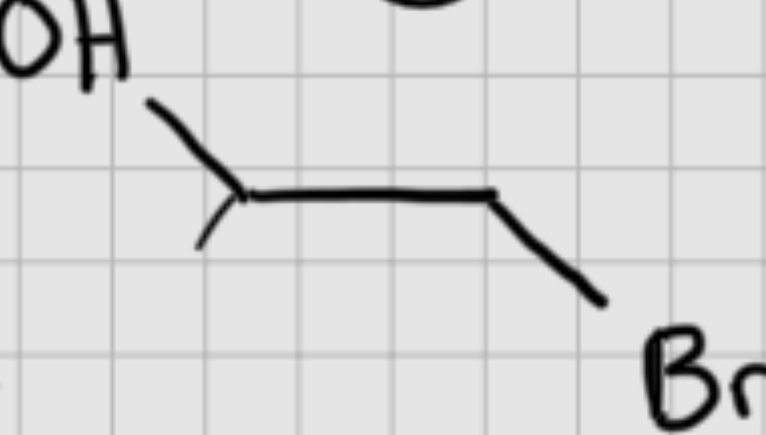
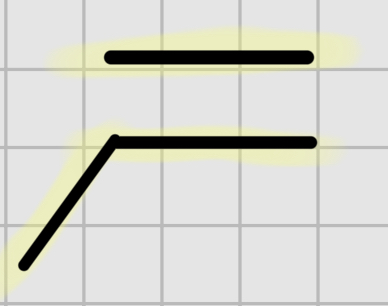
We add :
BH3
It’s syn addition.
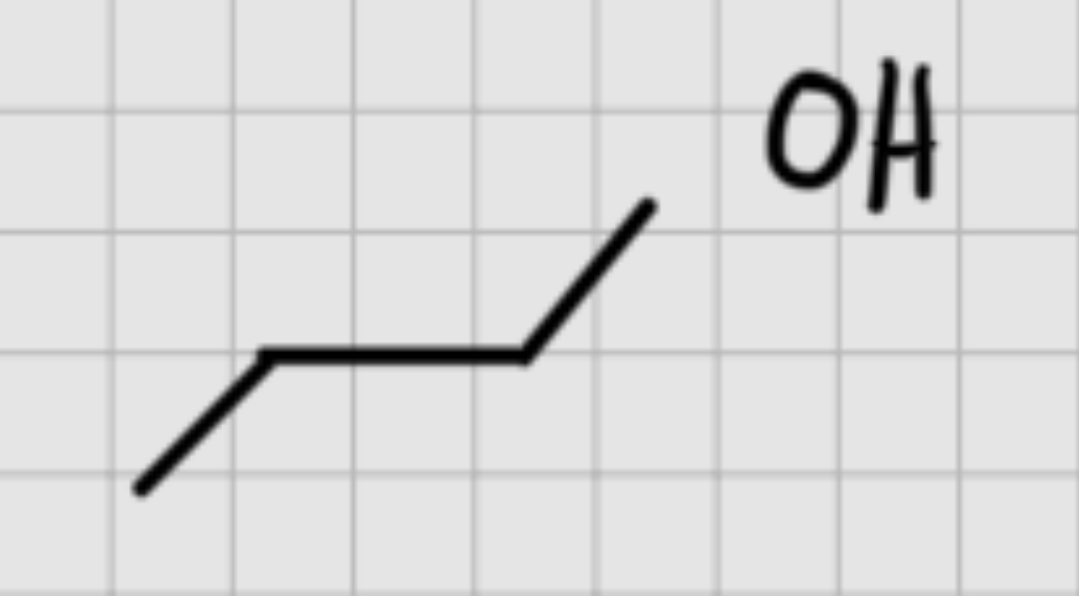
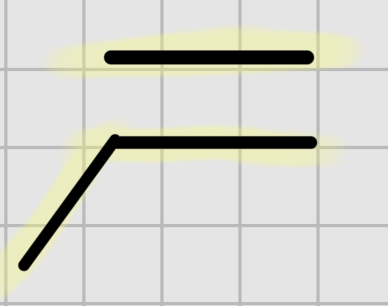
We add:
OSO4
It’s syn addition.
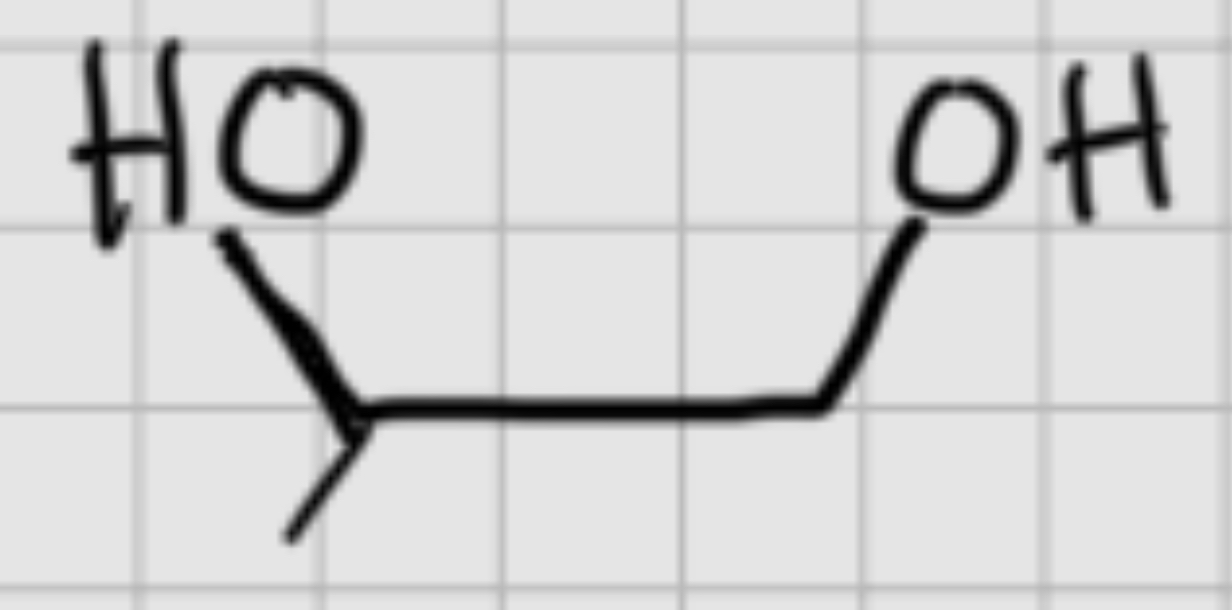
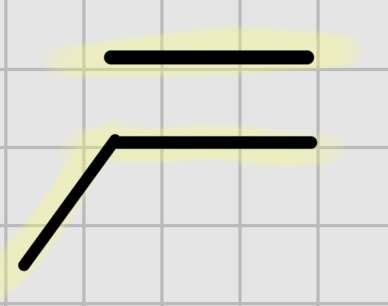
We add:
O3.
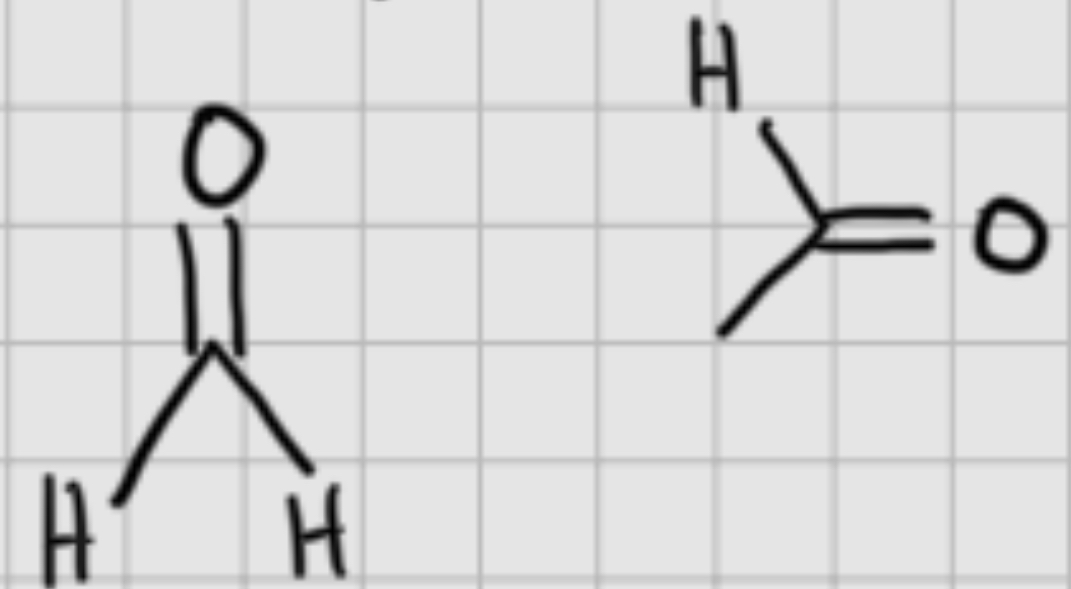
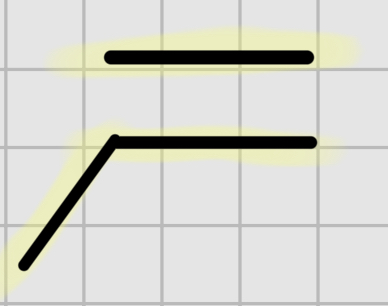
We add:
H+,(cat)
—————
H20
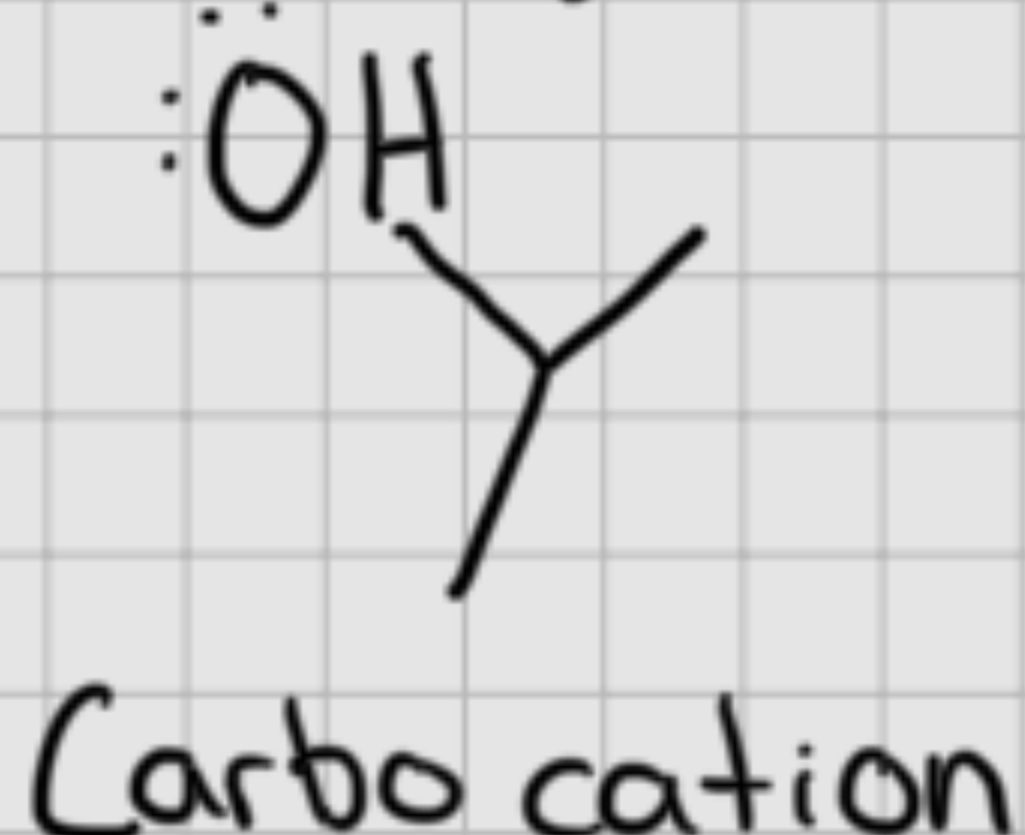
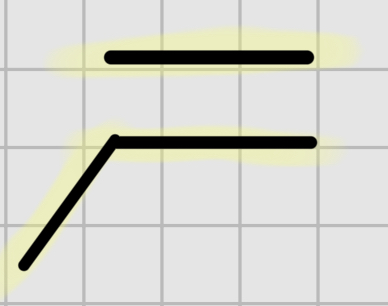
We add
1.Hg, H20
—
2. NaBH
It’s trans addition.
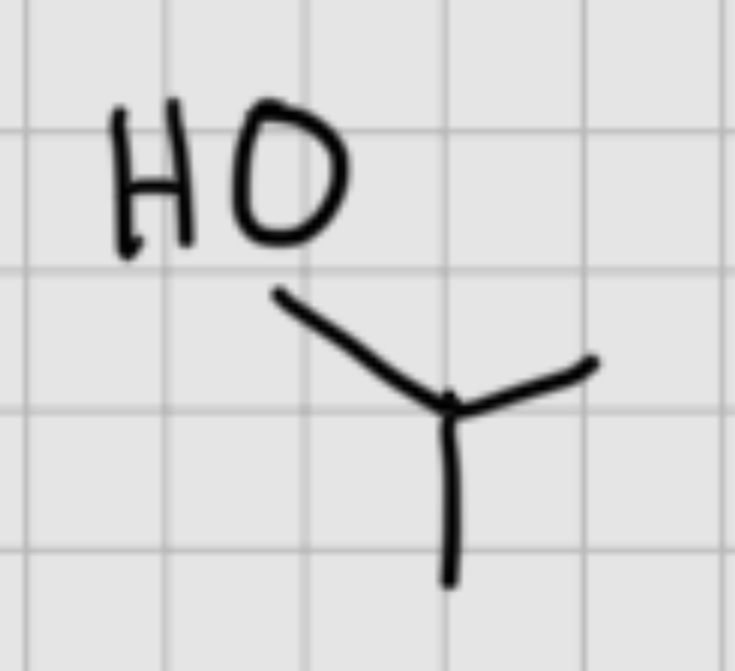
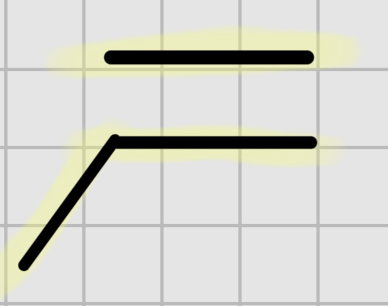
We add
H
—
Br
We add
H2
——
Pd