Acids and Alkailis
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
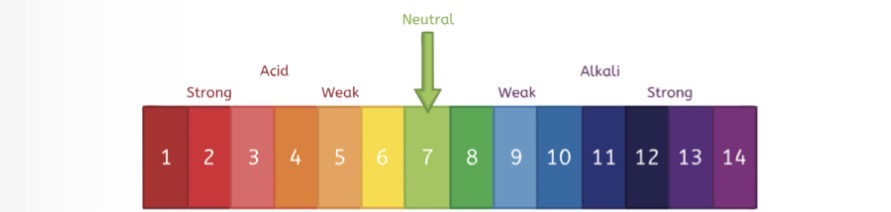
What will a concentrated acid have more of
A concentrated acid will have more acid particles per litre than a dilute acid
What are some common lab acids
Sulfiroc acid H2SO4
Hydronic acid HLC
Nitric acid HNO3
What is an indicator
An indicator is a solution that changes colour to determine the PH of a solution
What are some common indicators
Universal indicator, it turns red in acidic solutions and blue in alkaline solutions
Blue litmus paper turns red in acids
Red litmus paper turns blue in alkaline solutions
What is a well known Alkali
Sodium hydroxide NaOH is a well known alkali
What is neutralisation
Neutralisation involves adding an alkali to an acid to produce a neutral solution
Name tow acids
lemon water, vinegar
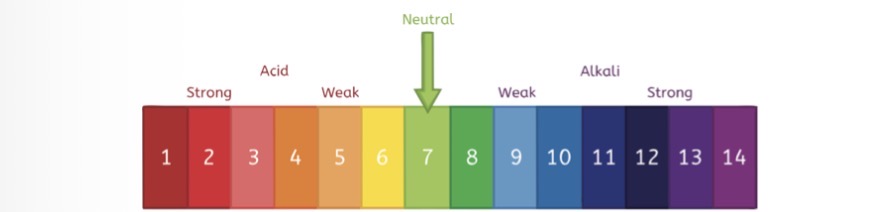
Name three alkali
Toothpaste
milk
oven cleaner
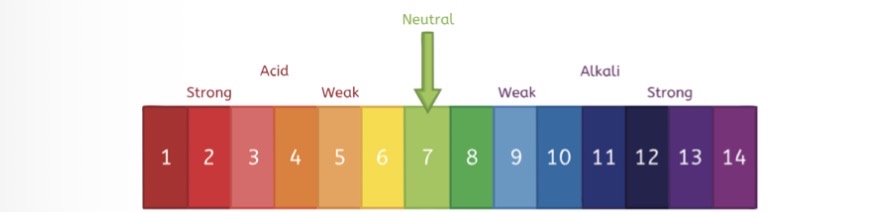
Name one neutral solution
water
What does hydronic acid make
Hydronic acid makes chloride salts
What does sulfuric acid make
sulfuric acid makes sulfate salts
What does nitric acid make
Nitric acid makes nitrate salts
How do you make copper sulfate crystals
Add powdered copper oxide to sulfuric acid
Filter the mixture to collect the un-reacted copper oxide and collect the copper sulfate solution
Place the copper sulphate solution into an evaporating dish and heat gently
The water will evaporate and copper sulfate crystals will form in the evaporating dish
Why are neutralisation reactions useful
Neutralisation reactions are useful because they can be used to neutralise soil or lakes