Exam 2
1/44
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
45 Terms
Enzyme
An enzyme is a biological catalyst that speeds up a chemical reaction by reducing the activation energy
Substrates
The reactants of an enzyme‐catalyzed reaction
Active site
Specific location of an enzyme that the substrate binds to via weak hydrogen bonds
T/F - Enzymes are consumed by their chemical reaction and cannot be used again
False - they are never consumed and can be recycled again. Thus VERY LITTLE enzyme is required to catalyze a reaction
Holoenzyme vs. Apoenzyme
Apoenzyme —→ the INACTIVE protein component of an enzyme that requires a non-protein component (cofactor) to function
Holoenzyme —→ the complete, catalytically ACTIVE form of an enzyme formed when the apoenzyme binds with its specific helper (cofactor or coenzyme)
Holoenzyme = Apoenzyme + Cofactor
Cofactor vs. Coenzyme
Enzymes need a helper in order to be activated and gain ability to catalyze reactions. The helper is either cofactor (inorganic) or coenzyme (organic)
Cofactor —→ Inorganic non-protein helper (Mg+ ion)
Coenzyme —> Organic non-protein helper (NAD+)
What are the 3 main factors affecting enzyme activity?
Temperature
pH
Amount of substrate
T/F - Increasing temperature will decrease the rate of enzyme activity
Depends
Increasing temperature BEYOND enzyme optimal temperature level will DECREASE the activity and cause denaturation, eventually rendering enzyme dysfunctional
Increasing temperature UP to enzyme optimal temperature will INCREASE enzyme activity
T/F - Increasing pH will decrease the rate of enzyme activity
Depends
All enzymes also have an optimal pH at which the enzyme can maintain its three−dimensional structure and have maximal activity. At a pH value too far ABOVE or BELOW the optimal pH, the enzyme will become denatured and will no longer be active
T/F - Increasing substrate concentration will decrease the rate of enzyme activity
False
Unlike pH or temperature, increasing substrate level will never DECREASE enzyme activity. It will increase only up to its SATURATION POINT (# enzyme = # substrate, i.e. every enzyme is bound to substrate so excess substrate needs to wait until enzyme is free)
Enzyme inhibitor (and describe specific vs. non-specific inhibitor)
Any factor that interferes with enzyme function is called an enzyme inhibitor. Temperature and pH are nonspecific inhibitors because they act by denaturing all proteins. Some inhibitors are more specific and prevent an enzyme from functioning by specifically binding to them.
Reversible vs. Irreversible inhibitors
Specific inhibitors can be either reversible or irreversible.
A reversible inhibitor can compete with a substrate for the active site of an enzyme. While the inhibitor is bound to the active site, the substrate cannot bind. The inhibition can be reversed if the substrate concentration is increased to a level at which it can out−compete the inhibitor for the active site on the enzyme.
Irreversible enzyme inhibitors bind to an enzyme in such a way that the enzyme is changed chemically and catalytic activity is permanently lost. Many poisons and toxins act by irreversibly inhibiting the action of important enzymes
The active site of an enzyme is the region that binds to the ____________________.
substrate
Succinate dehydrogenase normally catalyses the conversion of succinate to fumarate. If malic acid is added to this reaction, fumarate is no longer produced.
Identify the enzyme, product, substrate, and inhibitor in this reaction.
Enzyme —→ succinate dehydrogenase
Substrate —→ succinate
Product —→ fumarate
Inhibitor —→ malic acid
Most enzymes lose their function if they lose their structure. Most enzymes are also proteins. What type of structure do enzymes normally have?
Tertiary structure
A chemical reaction will proceed twice as fast for every 10°C rise in temperature. However, a reaction catalyzed by an enzyme will speed up only to a certain temperature at which point the reaction will cease. How do you explain this observation?
Temperature increases activity only up until the enzyme’s optimal temperature, beyond that will decrease it via denaturation
A catalyst is a substance that
a. increases the rate of a chemical reaction but is itself unchanged by the reaction
b. is toxic to most cells.
c. is composed mostly of lipids.
d. does not usually participate in any chemical reactions.
a. increases the rate of a chemical reaction but is itself unchanged by the reaction
A change in environmental pH can inhibit the proper functioning of an enzyme by
a. changing the shape of its active site.
b. altering the enzyme`s primary structure.
c. altering the specificity of the enzyme.
d. a, b, and c. e. a and c
e) a and c
What is a reducing sugar? What test is used to identify reducing sugar?
Reducing sugar —→ Carbohydrates with free ketone or aldehyde group that can donate electrons/hydrogen atoms
Benedict’s test:
Blue —→ no reducing sugar
Green to yellow —→ low to medium concentration
Orange to red to dark red —→ high to very high concentration
What is diffusion? Does it require energy?
Diffusion —→ movement of solutes DOWN their concentration gradient (go from high to low)
Does NOT require energy
What types of molecules move easily through plasma membrane? What doesn’t?
Small and non-polar —→ moves easily (hydrocarbons, CO2, O2)
Large and polar (ions) —→ does NOT move easily
Does water move easily through plasma membrane?
It moves, but NOT easily (remember, water is polar).
It needs facilitated diffusion via aquaporin membrane proteins that facilitate FAST movement (osmosis)
Passive transport vs. Facilitated diffusion vs. Active transport
Passive —→ diffusion of solutes across membrane with NO energy required
Facilitated —→ a TYPE of passive transport that requires aid of proteins (i.e. osmosis of water via aquaporins)
Active transport —→ transport that REQUIRES energy. active transport is NOT diffusion
T/F - Higher temperature increases rate of diffusion
True
Higher temp —→ higher kinetic energy —→ faster movement and collision of molecules
Lab: Higher temp caused dye to spread more quickly vs cold
What increases diffusion - higher or lower surface area to volume ratio?
Higher SA to V ratio
Higher SA to V ratio means there is less space in the center required for diffusion to reach the center efficiently
Scenario:
4 beakers of water solution, each solution is 1 mole of glucose
Each beaker contains 1 bag, with different levels of glucose solute concentration
Can glucose solute diffuse out of the bag?
Label each solution as hypertonic, hypotonic, or isotonic
If the experiment ran for 60 minutes, label each bag from most to least water volume level INSIDE the bag
No, glucose can’t move in/out. Only water moves to where there is higher solute concentration
Label:
Hypertonic —→ If solute bag has less glucose concentration than solution, the SOLUTION is HYPERTONIC to the bag because it has MORE solute level
Hypotonic —-→ if solute bag has more glucose concentration than solution, the SOLUTION is HYPOTONIC to the bag because it has LESS solute level
Isotonic —→ both bag and solution have equal level of solute
After 60 min:
Hypertonic solution —→ bag would LOSE water. if 2 solutions are hypertonic, then the bag with LESS solute would lose MORE water
Hypotonic solution —→ bag would GAIN water. if 2 solutions are hypotonic, then the bag with MORE solute would gain MORE water
Isotonic —→ bag experiences no net water gain or loss
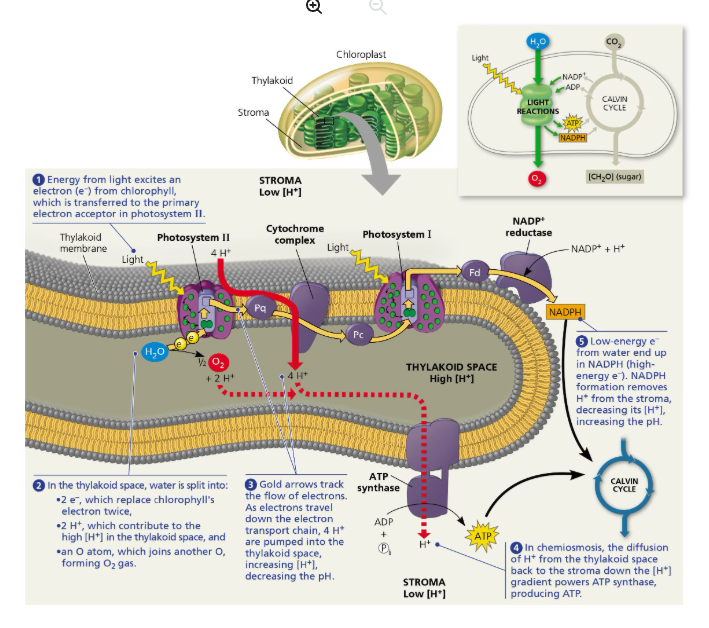
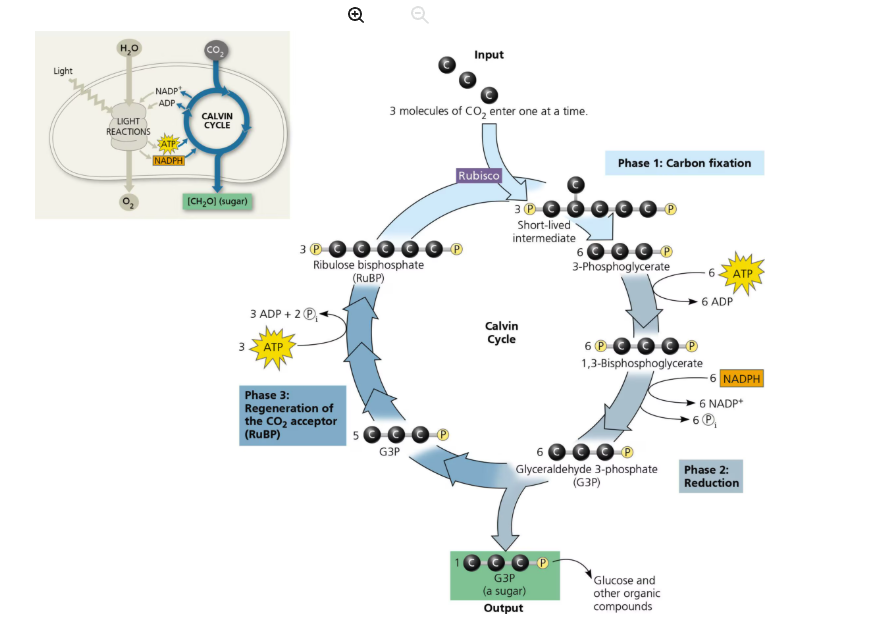
What do Light Reaction and Calvin cycle require and produce?
Light reaction:
Requires: Light Energy, Water, ADP, NADP+, Phosphate
Produces: ATP, NADPH, O2
Calvin cycle:
Requires: CO2, ATP, NADPH
Produces: Glucose/G3P (sugar), ADP, NADP+ (Note: ADP and NADP+ are returned to light reactions for reuse)
Photosynthesis paper chromatography:
What are the 2 factors affecting pigment movement on the paper?
is cellulose paper polar or nonpolar? is solvent usually polar or nonpolar?
Given various molecules with different distances traveled up the chromatography paper, describe the origin, stationary phase, mobile phase, retention factor/Rf
How to tell what the unknown compound is made of?
1) solubility in the solvent and 2) degree of attraction to the paper affect pigment movement
Cellulose paper is polar, solvent is usually then nonpolar
Origin —→ the starting line, usually drawn in pencil near bottom of paper. serves as the zero point for all measurements (why pencil? b/c graphite is nonpolar and insoluble so it doesn’t dissolve into dye and mess up results)
Stationary phase: the paper is polar and has water molecules attached. polar (hydrophilic) pigments have high affinity for paper therefore move less and stick to paper
Mobile phase: the solvent is nonpolar and is the vehicle that moves up the paper. nonpolar (hydrophobic) pigments dissolve in the solvent better (i.e. do not stick to the paper as much) and therefore move up the paper along with the solvent
Retention factor: ratio of distance traveled by substance divided by distance of solvent. cannot exceed 1. higher Rf means it traveled more distance therefore has higher affinity for mobile phase (more soluble in solvent and less attracted to paper)
NONPOLAR —→ higher affinity to mobile phase
POLAR —→ higher affinity to stationary phase
Color of a pigment comes from wavelengths of visible light that is _______
absorbed
reflected
Reflected
Describe the action spectrum in the context of photosynthesis. What is measured? What is expected? What is the significance?
Action spectrum —→ describes how effective different wavelengths of light are in initiating photosynthesis
Measured —→ oxygen production (oxygen is product of photosynthesis). wavelength that produces more floating discs means more photosynthesis
Expected: highest number of floating discs in red and blue light wavelengths
Significance —→ not all light is equally useful for photosynthesis
Describe the absorption spectrum in the context of photosynthesis. What is measured? What is expected? What is the significance?
Absorption spectrum —→ measures how much light can be absorbed by a pigment via spectrophotometer
Measured —→ absorption numeric value
Expected —→ peaks in red and blue wavelengths, dips in green
Significance —→ graph of absorption spectrum should closely match action spectrum because most absorbed wavelengths should be ones with most floating discs (i.e. most photosynthesis reactions)
In response to shortened daylight and cool temperatures during the fall, tree leaves display a beautiful array of colors from red to orange to gold to yellow. Explain this phenomenon using your knowledge of photosynthesis and photosynthetic pigments.
Trees stop producing chlorophyll when there is less and less daylight as well as during colder temperatures. As chlorophyll (green pigment) breaks down, the green color fades and other pigments that were present start to reveal (carotene for yellow/orange, for example).
Cellular respiration is an _________ reaction
Exergonic
Endogonic
Exergonic
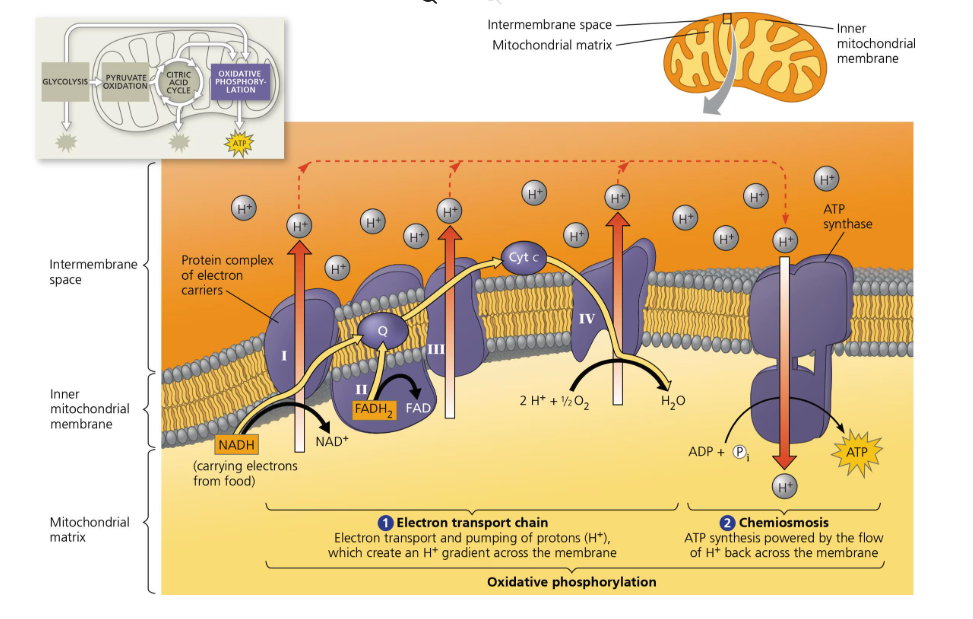
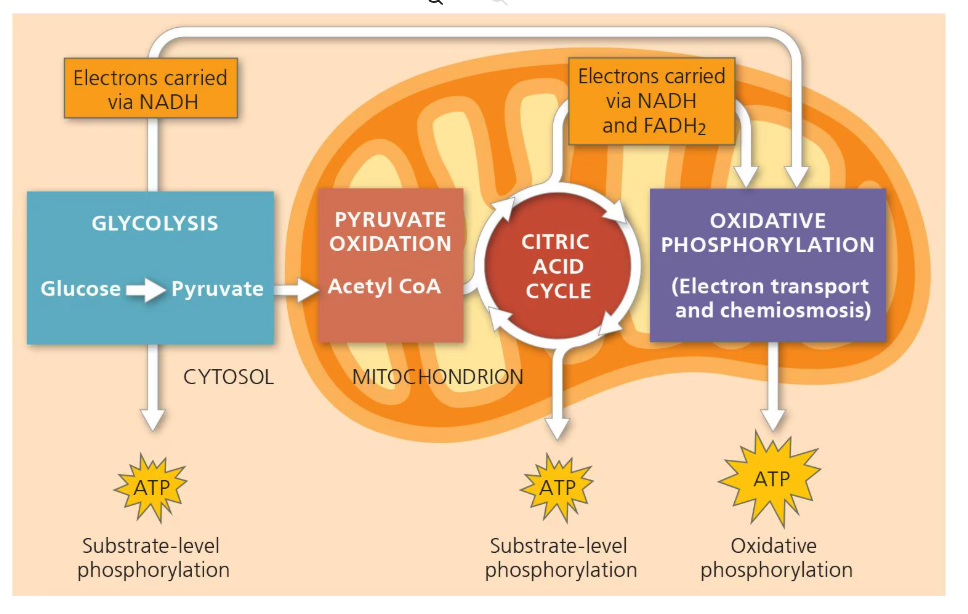
Describe the stages of aerobic cellular respiration and state the ATP yield for each (assuming one glucose molecule per cellular respiration)
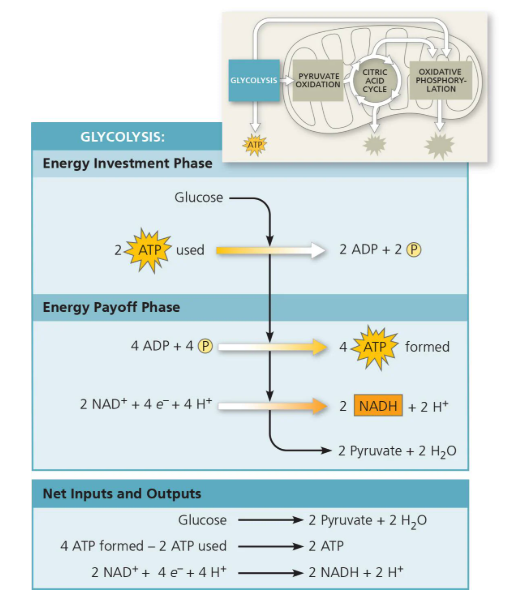
Glycolysis —→ 2 ATP (+ 2 pyruvate molecules)
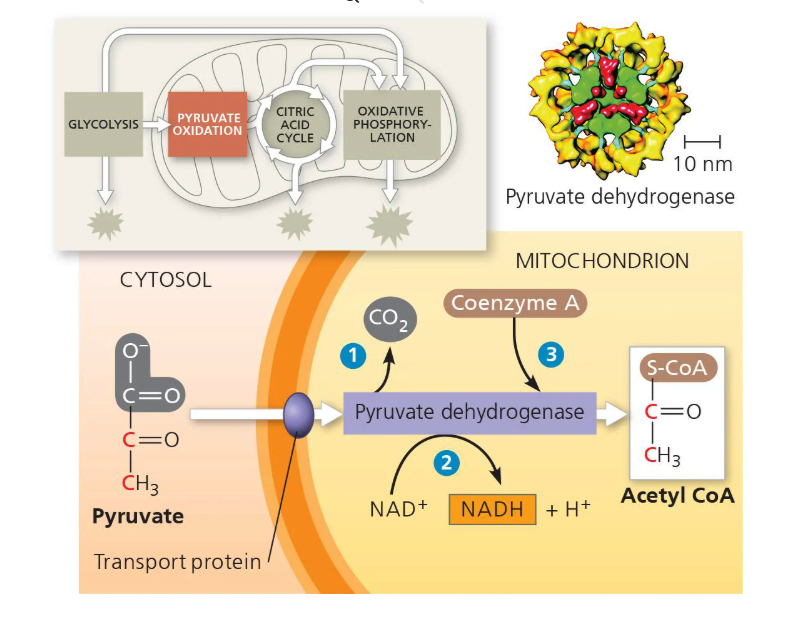
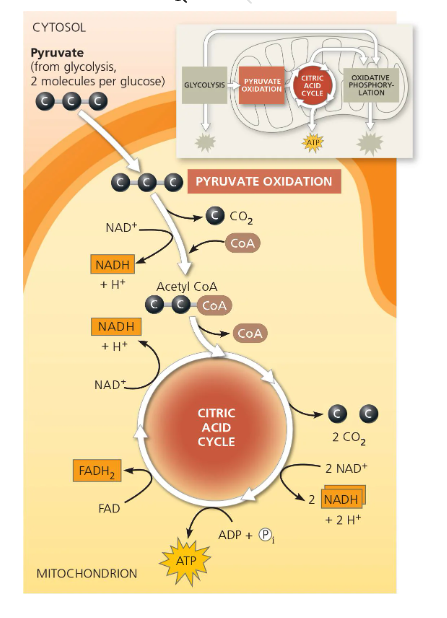
Intermediate reaction/Pyruvate Oxidation cycle —→ 0 (i.e. 2 pyruvate are turned into 2 Acetyl CoA)
Kreb’s Cycle/Citric Acid Cycle —→ 2 ATP total (+ 6 NADH, 2 FADH2, and most of CO2)
Oxidative Phosphorylation via electron transport chain —→ 32 to 34 ATP total (10 NADH x 3 ATP plus 2 FADH2 × 2 ATP). Why is not exact? Because due to differences in efficiency and non-integer ratios
Lab scenario: What is the effect of sugar on fermentation?
Expected:
Higher concentration of SIMPLE sugars in fruit juice should lead to more carbon dioxide production
if sugar is too acidic (high pH) then it can INHIBIT fermentation
if sugar is too complex, it takes longer for yeast enzymes to ferment and produce carbon dioxide
Significance:
not all sugars are created equal (simple vs complex)
volume of carbon dioxide is proportional to substrate (sugar), more fuel input leads to higher output
environmental inhibition: factors like pH can increase fermentation enzymatic activity up to an optimal level
Note: besides carbon dioxide, the smell of ethanol (alcohol) can also indicate fermentation activity
Lab scenario: Describe the effect of anti-oxidants on apple slices
Aerobic respiration is a redox reaction where oxygen is the __________
In living cells, oxidation can often lead to accumulation of_______, charged particles that cause damage to cells. _______ are substances that prevent buildup of these.
Aerobic respiration is a redox reaction where oxygen is the oxidizing agent.
In living cells, oxidation can often lead to accumulation of free radicals, charged particles that cause damage to cells. Anti-oxidants are substances that prevent buildup of these.
Expected:
Higher pH (lemon juice) should inhibit oxidization because it reacts with the oxygen before the apple can.
Significance:
pH can be used to control enzymatic reactions
Lab scenario: Describe the effect of temperature on fermentation
Expected:
Higher temperature increases carbon dioxide production (i.e. more fermentation and enzymatic activity) up to a certain point, after which is plateaus and then drops if in extreme heat
Cold = no activity
With respect to cellular respiration, what kind of organism is yeast?
Why can’t aerobes survive using fermentation for energy production?
What does gas displacement mean in context of fermentation and/or respiration?
What is the literal meaning of glycolysis?
If human muscles undergo fermentation and produce lactic acid during exercise, why don’t they produce alcohol?
With respect to cellular respiration, what kind of organism is yeast? —→ unicellular, heterotroph, energy via fermentation not respiration
Why can’t aerobes survive using fermentation for energy production? —→ fermentation produces way too little energy/ATP
What does gas displacement mean in context of fermentation and/or respiration? —→ gas displacement means carbon dioxide production (i.e. output of fermentation)
What is the literal meaning of glycolysis? —→ cutting/splitting of glucose (remember, glycolysis cuts 1 glucose into 2 pyruvate molecules)
If human muscles undergo fermentation and produce lactic acid during exercise, why don’t they produce alcohol? —→ we don’t have the same enzymes that cause alcohol production
(Diagram of Photosynthesis Light Reaction)
(Diagram of Photosynthesis Calvin Cycle)
(Diagram of Cellular Respiration Overview)
(Diagram of Cellular Respiration - Glycolysis Phase)
(Diagram of Cellular Respiration - Pyruvate Oxidation Cycle/Intermediate Phase)
(Diagram of Cellular Respiration - Citric Acid/Kreb’s Cycle)
(Diagram of Cellular Respiration - Oxidative Phosphorylation)