Biochemistry- Unit 2
0.0(0)
Studied by 7 peopleCard Sorting
1/48
Earn XP
Description and Tags
Last updated 10:29 PM on 9/18/23
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
49 Terms
1
New cards
monomer
The building blocks of macromolecules
2
New cards
universal solvent
a substance (water) that dissolves more solutes than any other substance because WATER IS POLAR
3
New cards
carbohydrate
"Compound containing carbon, hydrogen, and oxygen in the approximate ratio of C:2H:O (e.g., sugars, starches, and cellulose)"

4
New cards
protein
Class of nutrients made up of amino acids. They are needed to build and repair body structures, and to regulate processes in the body

5
New cards
lipid
Organic molecules made of carbon, hydrogen, and oxygen and store food energy until needed (Fats)

6
New cards
nucleic acid
A biological macromolecule (DNA or RNA) composed of the elements C, H, N, O, and P that carries genetic information.
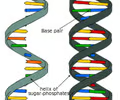
7
New cards
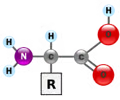
amino acid
Monomer of protein. There are only 20.
8
New cards
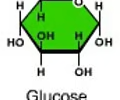
monosaccharide
A simple sugar that is the basic subunit of a carbohydrate
9
New cards
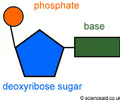
nucleotide
Monomer of nucleic acids made up of a 5-carbon sugar, a phosphate group, and a nitrogenous base
10
New cards
fatty acids
Building Blocks of Lipids
11
New cards
macromolecule
A very large molecule (as of a protein, nucleic acid, or carbohydrate) built up from smaller chemical structures
12
New cards
enzyme
A protein that makes a reaction happen QUICKER; decreases activation energy of a reaction.
13
New cards
glucose
A simple sugar that is an important source of energy.
14
New cards
Substrate
A substance on which an enzyme acts on during a chemical reaction.
15
New cards
High specific heat
A property of water. Water can absorb lots of heat before changing temperature
16
New cards
Carbon and Hydrogen
Elements in ALL living organisms. Considered organic
17
New cards
Catalyst/Enzyme
A substance that speeds up the rate of a chemical reaction.
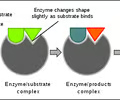
18
New cards
organic
any compound that contains carbon
19
New cards
denature
A change in the shape of a protein (such as an enzyme) that can be caused by changes in temperature or pH (among other things).
20
New cards
Activation energy
The minimum amount of energy needed to start a chemical reaction
21
New cards
active site
Region of an enzyme into which a particular substrate fits.
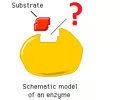
22
New cards
Polar
Describes a molecule with opposite charges on opposite ends
23
New cards
Adhesion
Tendency of water to stick to other substances
24
New cards
Cohesion
Tendency of molecules of the same kind to stick to one another
25
New cards
Hydrophillic
Attracted to water molecules, "water loving"
26
New cards
Hydrophobic
Repelled by water molecules, "water fearing" Example: oil/wax
27
New cards
Solvent
A substance that dissolves another.
28
New cards
Negative
In a water molecule, Oxygen has a partially \___ charge.
29
New cards
Positive
In a water molecule, Hydrogens have a partially \___ charge.
30
New cards
H₂O
The formula for water
31
New cards
pH Scale
Ranges from 0 to 14. Measures the amount of H+ in solutions.

32
New cards
Acid
any solution that is 0-6.9 on the pH Scale. Measures more H+ than OH-
33
New cards
Base
any solution that is 7.1-14 on the pH scale. Measures more OH- than H+
34
New cards
Neutral
solution that is 7 on the pH scale - water is an example. Equal H+ to OH-

35
New cards
Hydrgoen bonding
bonding between two water molecules. One negative oxygen bonding to another water molecule's positive hydrogen
36
New cards
Covalent bonding
a bond formed when atoms share one or more pairs of electrons
37
New cards
Ionic bonding
Chemical bonding that results from the electrical attraction between positive and negative ions
38
New cards
Reactant
A chemical substance that is present at the start of a chemical reaction
39
New cards
Product
A substance made in a chemical reaction
40
New cards
compound
a pure substance that is formed when 2 or more elements combine
41
New cards
ion
an atom that has a negative or positive charge because it has gained/lost an electron
42
New cards
6 main elements?
Carbon, Hydrogen, Nitrogen, Oxygen, Phosphorus, Sulfur
43
New cards
Water is?
Polar
44
New cards
Polar molecules
Water has a positive side and a negative side
45
New cards
Forms Hydrogen bonds
Attraction between the positive end of 1 water molecule & the negative
46
New cards
Expands when frozen
Frozen water is less dense than liquid water
47
New cards
Capillary Action
Water moves against gravity due to cohesion/adhesion
48
New cards
Surface tension
Water makes a film on the surface due to cohesion/adhesion
49
New cards
if temperature rates are too low?
reaction rates are slow