Physics Definitions Unit 3
1/55
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
56 Terms
Angular Velocity ⍵ (circular motion)
Rate of change of angle with time
Radian (circular motion)
angle when arc length is equal to radius
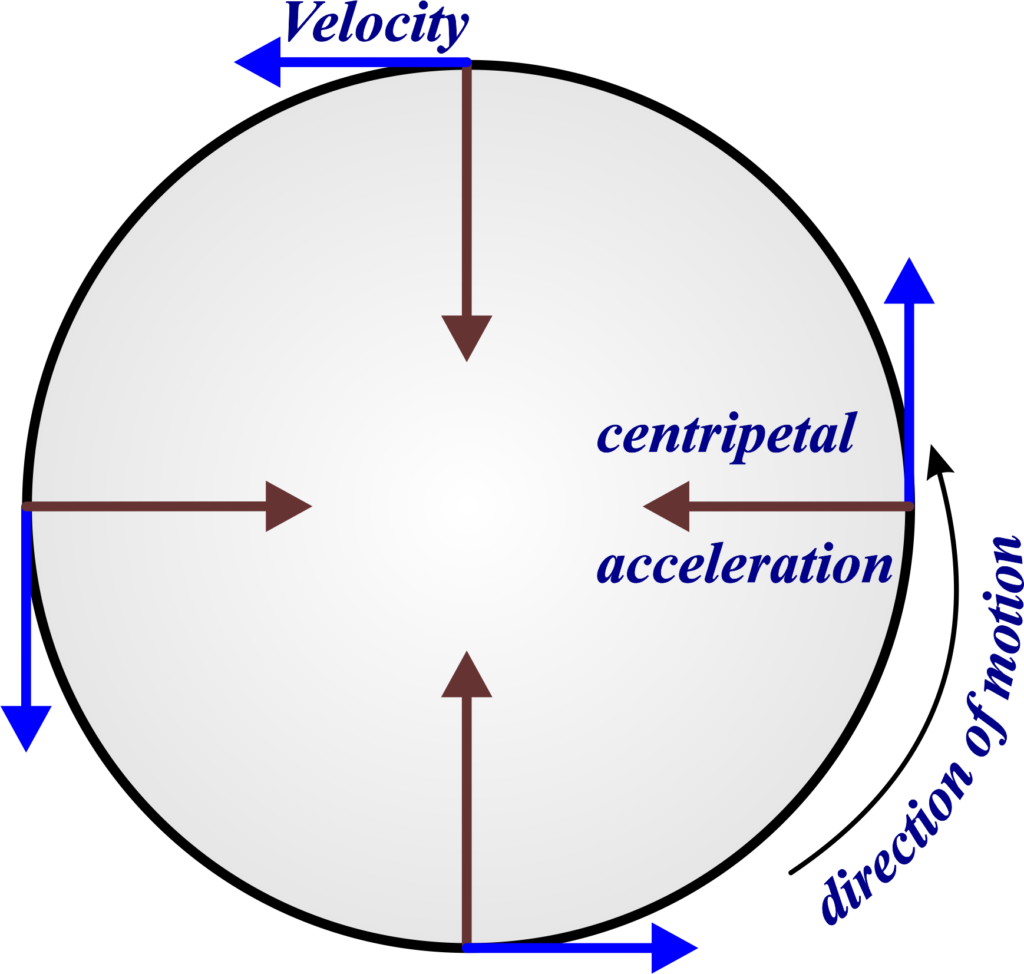
Why does and object moving at a constant speed in a circle have acceleration? (circular motion)
velocity is a vector - direction changes but not magnitude
acceleration = rate of change of velocity
Where is greatest tension in a vertical circle? (circular motion)
the bottom
Where is least tension in a vertical circle? (circular motion)
the top
Simple Harmonic Motion (SHM)
The acceleration of the body is directly proportional to its distance from a fixed point and is always directed towards that point
Period (SHM)
The time for one complete oscillation or cycle
Frequency (SHM)
number of cycles completed per second
amplitude (SHM)
maximum displacement from equilibrium position
Phase constant ε (SHM)
angular displacement when displacement (x) = 0
Damping (SHM)
the process whereby energy is taken from an oscillating system and the oscillations decrease as a result
Types of damping (3) (SHM)
Light, Heavy, Critical
Time taken for system to return to equilibrium in critical damping? (SHM)
usually ¼ T
Free Oscillations (SHM)
Oscillating body has no force applied and vibrates at its natural frequency
(displacement applied then released)
Forced oscillations (SHM)
System vibrating due to periodic application of external driving force
Resonance (SHM)
driving force of a oscillating system with a frequency that matches the natural frequency, resulting in a large amplitude
Ideal gas assumptions (kinetic theory) (4)
collisions of gas molecules are perfectly elastic
intermolecular forces are negligible, except during collision
gas molecules move rapidly and randomly
volume of molecules is negligible compared to volume of gas
Avogadros constant (kinetic theory)
number of particles per mole
Radioactive Atom (radioactivity)
atom with an unstable nucleus
alpha particle (radioactivity)
2 protons and 2 neutrons
helium nucleus
beta particle (radioactivity)
electron
how fast is beta radiation? (radioactivity)
just less than the speed of light
gamma radiation (radioactivity)
high energy, high frequency electromagnetic wave
Half-life (radioactivity)
time taken for the number of radioactive nuclei to half
3 methods of distinguishing between alpha, beta and gamma radiation? (radioactivity)
differing penetration, deflection by electric field, deflection by magnetic field
results for differing penetration (radioactivity)
alpha - decrease with paper absorber
beta - further decrease with 3mm aluminium
gamma - further decrease with 3cm lead or remaining radiation above background level after aluminium
results for deflection by electric field (radioactivity)
alpha - attracted by negative plate
beta - attracted by positive plate
gamma - no change
results for deflection by magnetic field (radioactivity)
alpha - deflected
beta - deflected more in opposite direction
gamma - no change
ion (radioactivity)
an atom which has lost or gained one or more electrons
Activity (radioactivity)
number of radioactive disintegrations per second
rate of decay
Becquerel (Bq) (radioactivity)
one disintegration per second
what does the decay constant determine? (λ) (radioactivity)
rate of decay
Internal energy (thermal physics)
sum of the random KE and PE of the molecules
potential energy of gases (thermal physics)
0 because negligible intermolecular forces
Absolute zero (thermal physics)
the temperature when internal energy is a minimum
heat (thermal physics)
thermal energy moving from an area of high temperature to an area of low temperature
ΔU in ΔU=Q - W (thermal physics)
change in internal energy
Q in ΔU=Q - W (thermal physics)
Heat added to system
W in ΔU=Q - W (thermal physics)
work done by the gas
condition for bodies to be in thermal equilibrium (thermal physics)
they are the same temperature
what happens when a gas does work (thermal physics)
expands
what happens when work is done on a gas (thermal physics)
contracts
clockwise cycle on a graph gives (Q and W) (thermal physics)
positive Q and W
anticlockwise cycle on a graph gives (Q and W) (thermal physics)
negative Q and W
specific heat capacity (thermal physics)
the amount of thermal energy needed to raise the temperature of 1kg of material by 1K
Binding Energy (nuclear energy)
work done on a nucleus to completely separate it into its neutrons and protons
Mass defect (nuclear energy)
mass lost when energy released
U- unified atomic mass unit
1/12 mass of carbon 12 atom
n (thermal physics/kinetic theory)
number of moles
N (thermal physics/kinetic theory)
number of molecules
U (kinetic theory)
translational KE of a monotomic gas
NA (kinetic theory)
avogadros
Centripetal force (circular motion)
resultant force for circular motion towards the centre
M (Kinetic theory)
total mass
mr (Kinetic theory)
molar mass
m (Kinetic theory)
mass of a molecule