Chemistry - using reasources
1/81
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
82 Terms
examples of finite resources
- fossil fuels (coal, oil, gas)
- metal ores (copper, iron)
- nuclear fuels (uranium)
- minerals (limestone)
Examples of renewable resources
- wheat
- leather
- lumber
- corn
- drinking water
- paper
- wool
What is potable water?
water that is safe to drink
How is potable water made?
1. first the water passes through filters to remove large, solid particles, like dirt, leaves and small debris
2. Chemicals like aluminium sulfate are added to the water to cause smaller particles to flocculate and sink to the bottom
3. The water is filtered again, and chlorine is added to kill pathogens
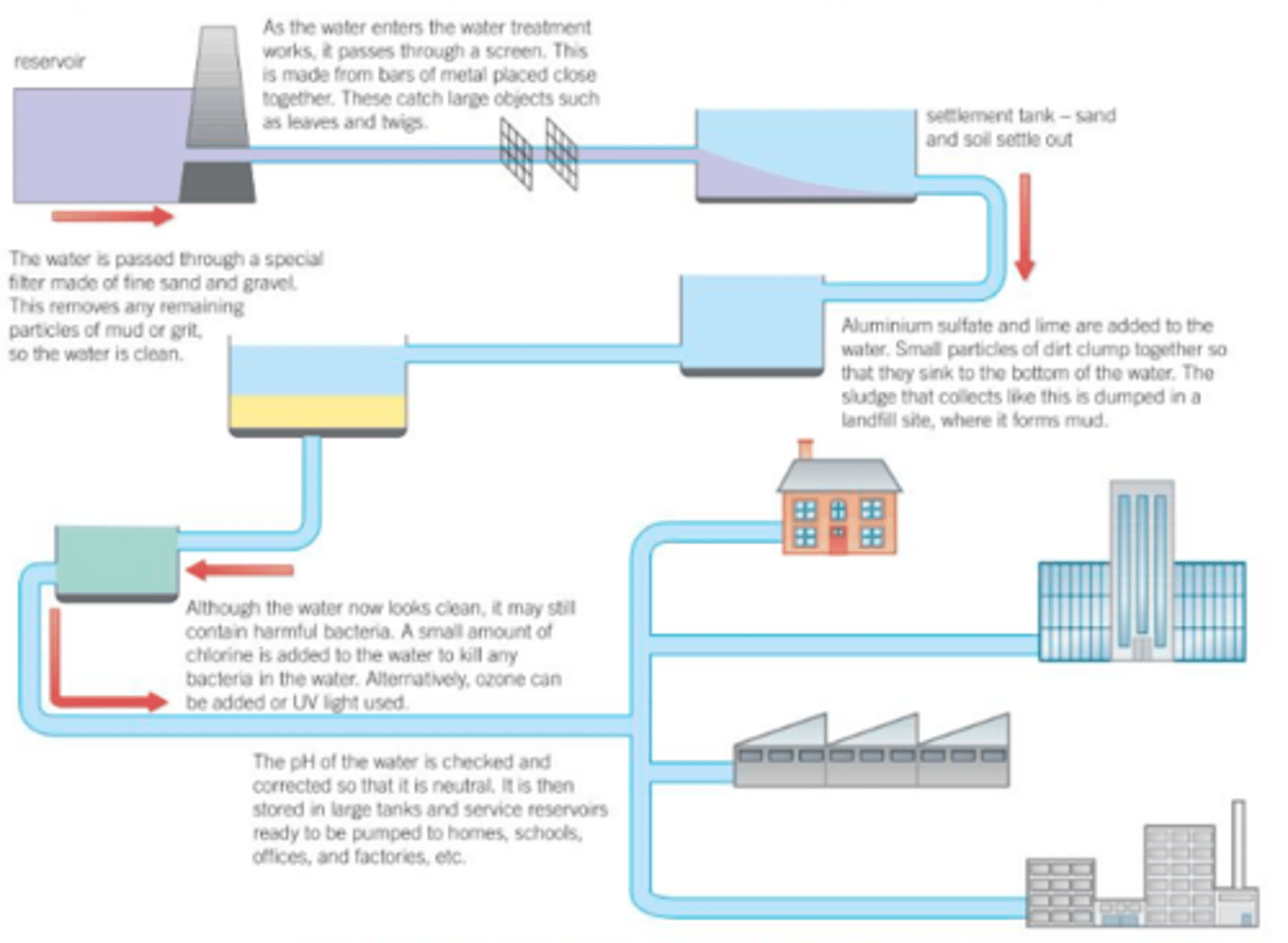
How is potable water made in countries with little rainfall?
desalination
What is desalination?
Removing salt from seawater
How can water be desalinated?
through distillation or reverse osmosis
How does reverse osmosis work?
Pressure is applied to the water to force it through a semi-permeable membrane. The membrane allows water molecules through, whilst the contaminants are left behind
What is flash distillation?
Flash distillation is when, under reduced pressures, water boils below 100 degrees to save energy costs.
How does distillation work?
1. water is heated until it evaporates and turns into steam.
2. Salts and impurities don't evaporate, so they are left behind.
3. The steam is cooled and condensed back into liquid water.
4. The collected water is pure.
What are the disadvantages of distillation?
lots of energy, expensive
What are the advantages of reverse osmosis?
- it removes 99%+ of contaminants
- it uses less energy than distillation
What are the disadvantages of reverse osmosis?
- can be expensive
- memeberanes need to be replacd
- essentail minerals like calcium and magnesium are removed
How is waste water treated?
1. Sewage is screened by passing through a mesh to remove large solids, like sticks, plastic, and rags
2. the water is then left to settle in large sedimentation tanks.
- Solid particles sink to the bottom, forming sludge
- The liquid left on top is called effluent
3. air is pumped through the effluent to provide oxygen for aerobic bacteria
- The bacteria break the organic matter down
- this water goes back into the waterways
4. The sludge is kept in a sealed tank to remove the oxygen for anaerobic bacteria
- the bacteria produce methane and safe solid matter
- The sludge can be used as fertilisers for farming
What is an ore?
rock containing metal compounds
What are low-grade ores?
Ores that only contain small amounts of the metal.
How can metals be extracted from low-grade ores?
bioleaching and phytomining
How are metals more reactive than carbon extracted?
by electrolysis
How are metals less reactive than carbon extracted?
reduction with carbon in a blast furnace
What are the advantages of electrolysis?
Produces pure metals
What are the disadvantages of electrolysis?
- high energy consumption
- high cost
- electrodess need to be replaced frequently
What is phytomining?
1. Plants are grown on soil that contains low-grade ores
2. The plants absorb the metal ions through their roots and concentrate them in their leaves and stems.
3. The plants are harvested and burned.
4. The ash contains the metal compounds.
5. The metal compounds in the ash are dissolved in an acid to create a solution called leachate
6. The metal is then extracted from the ash through electrolysis or displacement reactions using a more reactive metal (e.g., scrap iron)
What are the advantages of phytoming?
- Useful for low-grade ores
- Causes less damage to the environment than mining
- cost effective
- helps clean up contaminated land
What are the disadvantages of phytoming?
- Slow process
- Depends on plant growth and on the seasons
- Large amounts of land are required to grow plants
- limited to specific areas
How can copper be extracted from copper-rich ores?
- reacting with sulfuric acid
- smelting
What is leachate?
an acidic, metal-rich liquid solution produced during bioleaching, where bacteria break down low-grade metal ores
What is smelting?
- process used to extract metals from their ores by heating them to high temperatures in a furnace with carbon
What is bioleaching?
1. Bacteria are added to a low-grade ore.
2. The bacteria feed on the ores and produce an acidic solution containing metal ions, known as the leachate
3. The metal is then extracted from the solution (e.g. by electrolysis or displacement)
What are the advantages of bioleaching?
- Works with low-grade ores
- Less energy is needed than traditional mining
- Causes less environmental damage
What are the disadvantages of bioleaching?
- results in a lower yield
- Slow process
- The acidic solution can be harmful to the environment if it leaks
What is the LCA?
A Life cycle assessment is used to assess the environmental impact a product has over it's entire lifespan
What are the stages of the LCA?
1. Raw materials extraction
- Getting the materials from the Earth (e.g. mining, drilling).
- Uses energy and can damage habitats.
2. Manufacturing and packaging
- Turning raw materials into a product.
- Requires energy and produces waste and pollution.
3. Use of the product
- Some products need energy to work (e.g. electrical appliances).
- May produce pollution during use.
4. Disposal
What happens at the end of its life:
- Landfill
- Recycling
- burning
Why are LCAs useful?
- Help compare products (e.g. paper vs plastic bags)
- Help companies reduce environmental damage
What are the limitations of LCAs?
- Not always reliable (data can be incomplete)
- Different assumptions can give different results
How is gold found?
as a pure metal because it is very unreactive
What is rusting?
The corrosion of iron due to the reaction of iron with air in the presence of water
What causes rusting?
iron reacting with oxygen and water
Formula for rusting
iron + oxygen + water -> hydrated iron oxide
What are the conditions for rusting?
water and oxygen
What type of reaction is rusting?
redox reaction, Iron is oxidised (loses electrons) when it reacts with oxygen and water
What are the two methods of preventing rust?
Barrier methods (stop air and water from reaching iron) and sacrificial protection
What are the barrier methods to stop rusting?
- Paint
- Oil/grease
- Plastic coating
Why does salt water make rusting faster?
it acts as an electrolyte, greatly increasing the conductivity of water and allowing electrons to move more easily between the iron and oxygen
What is galvanising?
- coating iron or steel with a protective zinc layer to prevent rusting
- zinc is more reactive than iron, so it reacts with the moisture instead of the iron
- if the surface is scratched, corrosion is still prevented as it is a sacrificial protection
What is sacrificial protection?
How does alloying avoid rusting?
- Some metals are alloyed to make them more resistant to corrosion.
- For example, stainless steel is an alloy of iron, chromium, and nickel, which is highly resistant to rust.
How does paint avoid rusting?
- by acting as a protective barrier that seals metal surfaces from moisture and oxygen
How do oil and grease avoid rusting?
by creating a hydrophobic, physical barrier that seals metal surfaces from oxygen and moisture
How do plastic coating avoid rusting?
by creating barrier between the metal surface and the environment, which blocks oxygen and moisture from reaching the metal
What is steel?
- An alloy made from iron and carbon
- it is used in construction, automotive industries, and tools.
What is stainless steel?
- An alloy of iron, carbon, and chromium.
- It is resistant to rusting and corrosion, making it ideal for use in kitchen utensils, medical equipment, and structures exposed to moisture.
What is bronze?
- An alloy of copper and tin.
- It is used in making coins, sculptures, and musical instruments because it is durable and has a pleasing appearance.
What is brass?
- An alloy of copper and zinc.
- It is used for making items that need to resist corrosion, such as in plumbing fittings and electrical connectors.
What is high-carbon steel?
a strong, hard, and brittle alloy of iron containing higher carbon content
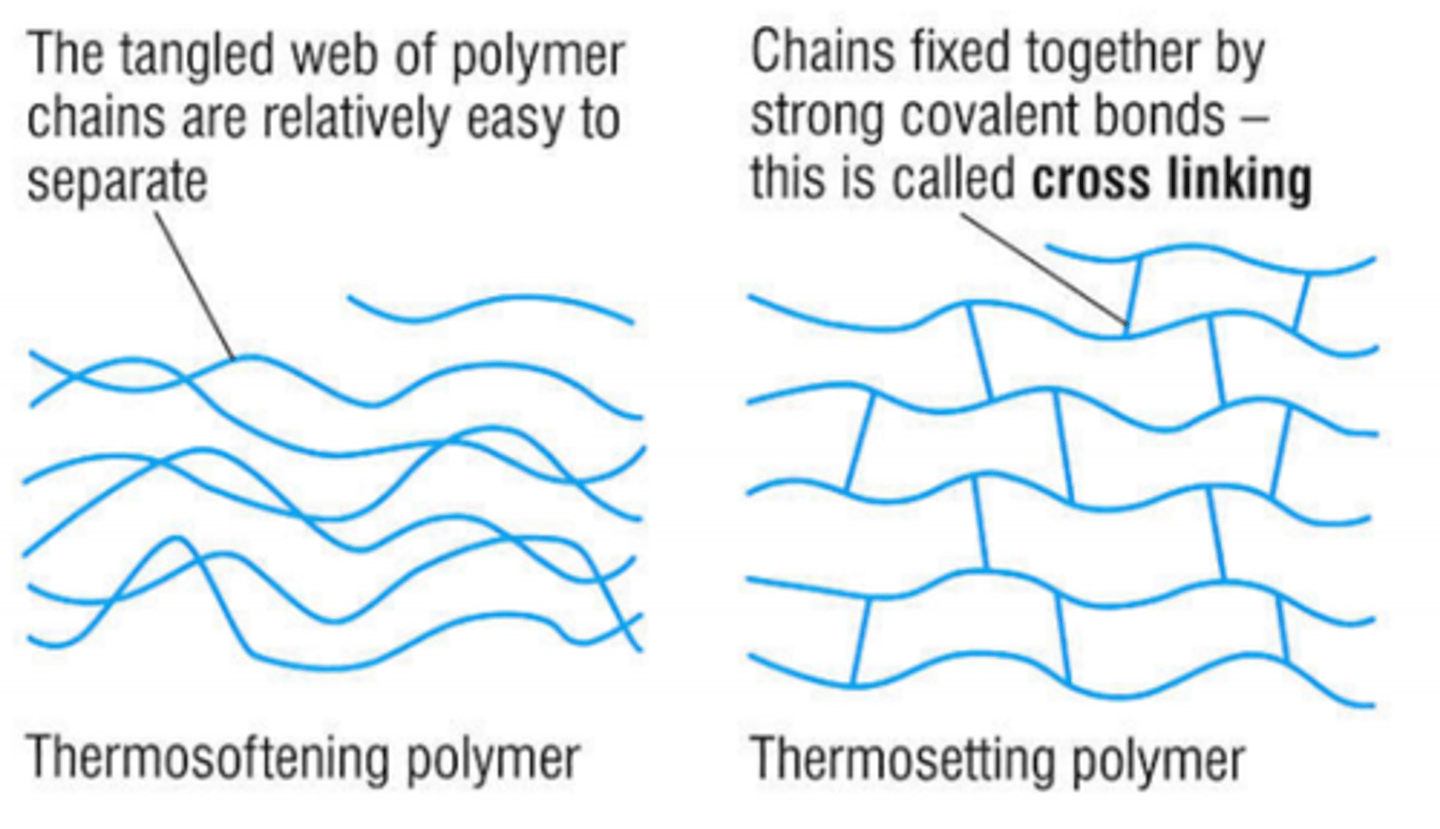
What are thermoforming polymers?
Plastics that can be heated and reshaped many times.
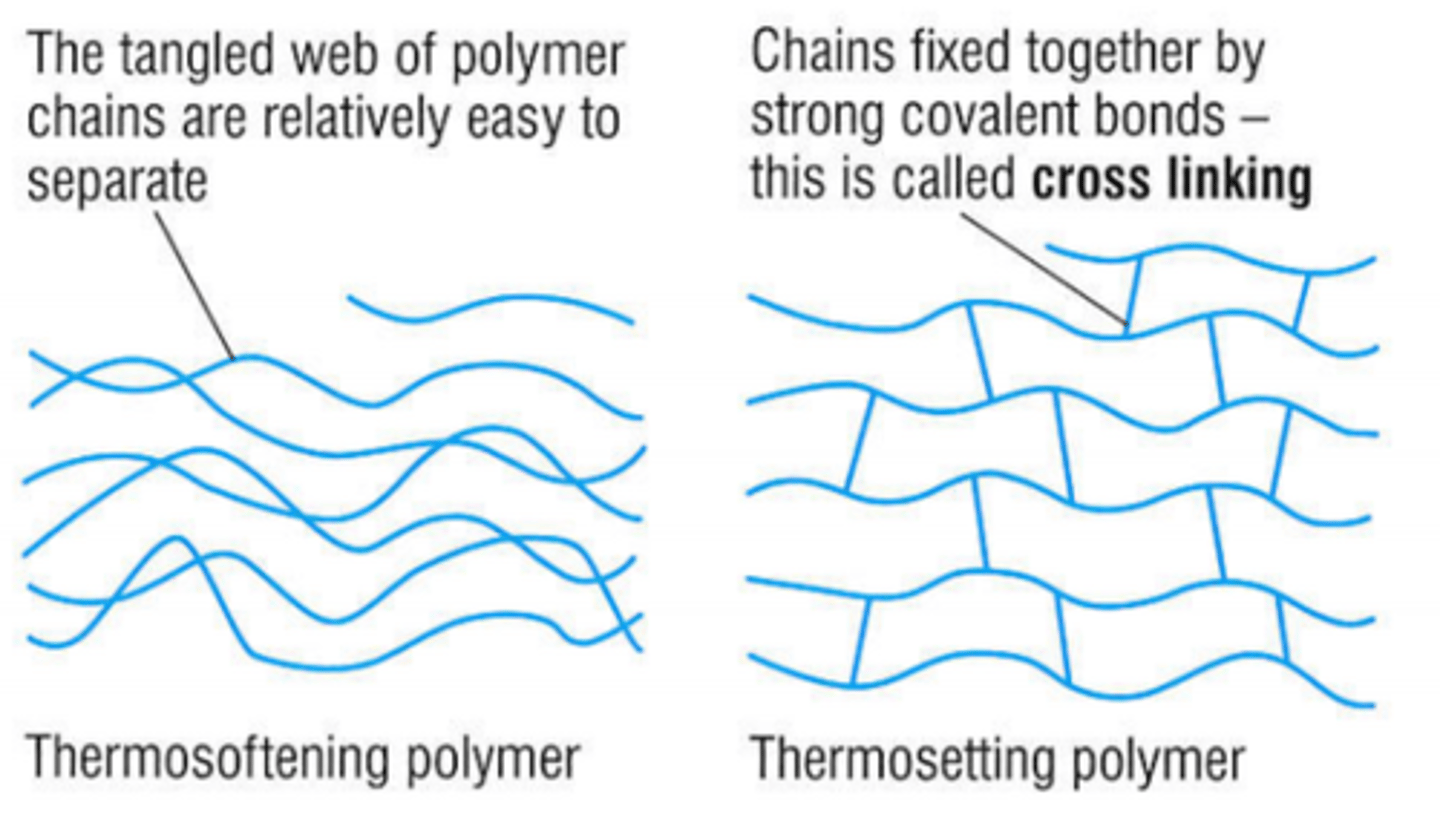
Why can thermoforming plastics be reshaped?
- The polymer chains are held together by weak intermolecular forces.
- When heated, these forces are easily overcome, so the plastic softens.
What are the properties of thermoforming plastics?
- Melt when heated
- Flexible
- Can be recycled and remoulded
What are thermoforming plastics used for?
- Plastic bottles
- Carrier bags
What are thermosetting polymers?
Plastics that do not melt when heated after they are set.

Why can't thermosetting polymers melt?
- The polymer chains are linked by strong covalent bonds.
- These bonds do not break easily, so the shape is fixed.

What are the properties of thermosetting plastics?
- Do not melt (they may burn instead)
- Rigid and strong
- Cannot be reshaped or recycled easily
- heat-resistant
- durable
- chemically resistant
- excellent electrical insulators
What are thermosetting plastics used for?
- Electrical plugs
- Saucepans handles
Is poly(ethene) a thermoforming or thermosetting polymer?
thermoforming
What are the two types of polyethene?
- Low density poly(ethene) (LDPE)
- high density poly(ethene) (HDPE)
What is low-density polyethene?
a flexible, durable, and transparent thermoplastic which is used for packaging, films, and bottles
What is the structure of LDPE?
- Polymer chains are branched.
- This means they cannot pack closely together.

What are the properties of LDPE?
- Less dense
- Soft and flexible
- Lower melting point
What is LDPE used for?
- Plastic bags
- Squeezable bottles
What is the structure of HDPE?
- Polymer chains are straight (unbranched).
- They can pack closely together.

What is high-density polyethene?
a strong, rigid, and durable thermoplastic usually used in milk bottles and water pipes
WHat are the properties of HDPE?
- More dense
- Strong and rigid
- Higher melting point
What is HDPE used for?
- Milk bottles
- Water pipes
What is glass?
Glass is a transparent, brittle material made by melting sand (silicon dioxide) and cooling it quickly so it doesn't form crystals.
What are the properties of glass?
- Transparent (you can see through it)
- Hard but brittle (breaks easily)
- Waterproof
- Can be shaped when hot
Uses of glass
- Windows
- Bottles and jars
- Screens (phones, TVs)
What are ceramics?
Ceramics are materials made from clay or other minerals, shaped and then heated in a kiln at high temperatures.
What are the properties of ceramics?
- Very hard and strong
- Brittle
- Heat-resistant
- Good insulators
Examples of ceramics
glass, pottery, porcelain, bricks
uses of ceramics
- Bricks and tiles
- Plates and cups
- Toilets and sinks
What are composite materials?
Composite materials consist of two materials with different properties. Made to produce a material with improved properties
What are composites made from?
- Reinforcement (gives strength, e.g. fibres)
- Matrix (holds it together, e.g. resin)
What is reinforced concrete?
Concrete set around steel bars to combine strength and flexibility with hardness