Spectroscopy and Electromagnetic Spectra
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
Briefly state what the three main types of spectroscopy are
electronic spectroscopy
how electrons move between orbitals
vibrational spectroscopy
how molecules vibrate
rotational spectroscopy
how molecules rotate
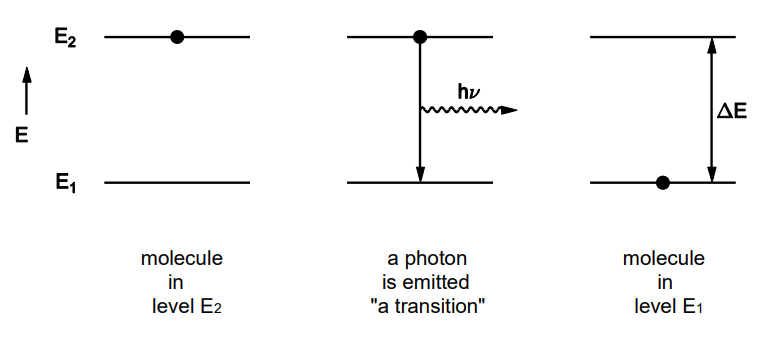
What are transitions
Transition are when a molecule changes from one energy level / state to another
because energy levels are quantized, a transition involves the intake or release of a discrete amount of energy
Describe energetics of transitioning
When a transition occurs, the energy released is Delta E = hv
So the light emitted will have frequency v = ΔE/h
for a transition to occur, the energy provided must be ΔE
the energy of a photon is hv
How can we describe electromagnetic radiation
Can be describe as a wave of oscillating electric and magnetic fields.
Give equation for frequency and wave number
Quantification of properties
Speed of Light (in a vacuum), c, ms^-1
Wave length (length of one wave), λ, m
Frequency (Number of waves per second), v, s^-1 = Hz
Wave number (Number of waves in 1 cm) v ~ = cm^-1
Describe what a photon is
Electron magnetic radiation is also describe as a stream of particles called photons
Each carries a specific amount of energy (a quantum of energy)
An intense beam of light will contain many photons
Give equation for One photon and One mole of photons
Quantification of properties
Speed of Light (in a vacuum), c, ms^-1
Planck Constant, h
Avogadro constant, NA
Boltzmann constant, k

Describe how wave particle duality applies to photons
At the atomic level, radiation or matter can appear to be waves or particles depending on how we observe them
So wave and photon descriptions are used interchangeably
High Frequency and High Wave Number = High Energy
Energy given in cm-1 units
Low wave length = Low Energy
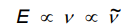
Describe what is meant by the electromagnetic spectrum
The range over which electromagnetic radiation is typically observed and used
Different regions of the spectrum interact with matter in different ways
Different types of spectroscopy
Draw out the wave spectrum detailing type of spec/analysis
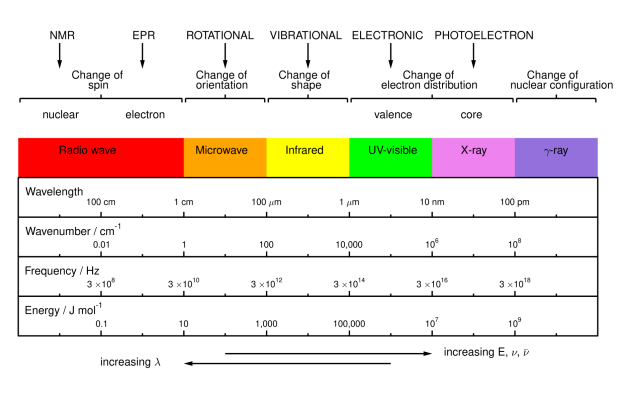
What does a spectrum refer too
A spectrum refers to a plot of absorption, emission, or scattering versus the wavelength, wavenumber, or frequency
Shows the specific energies that a molecules can take in or release
Draw out a basic absorption spectrometer set up
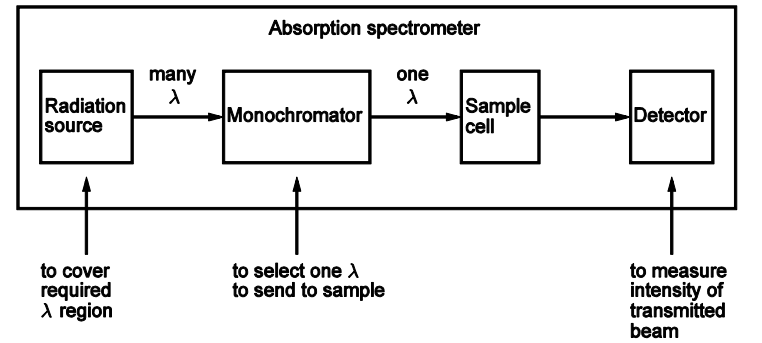
Describe what is measured in an absorption spectrometer
to record a spectrum
scan the monochromator across a range of λ
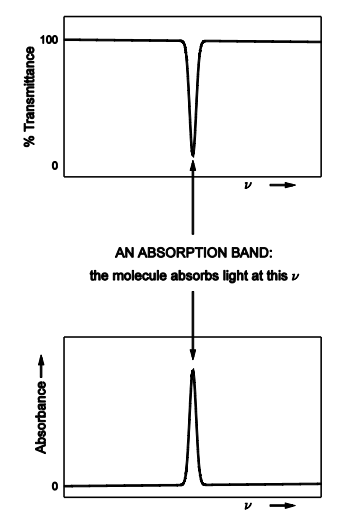
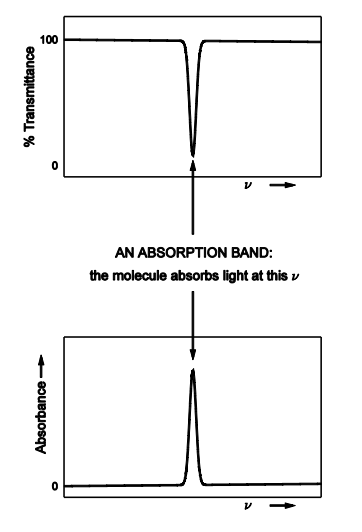
regions where a molecule absorbs / emits / scatters are typically called
LINES if they are narrow
BANDS if they are broad
PEAKS generally
outside these regions is - the BASELINE
the appearance of the spectrum depends on various spectrometer settings that can often be set by the user, including
the RESOLUTION , smallest peak separation that can be distinguished (in λ, ν ~ , or v) e.g. determined by the monochromator slit width
Draw out a quantitative absorption set up
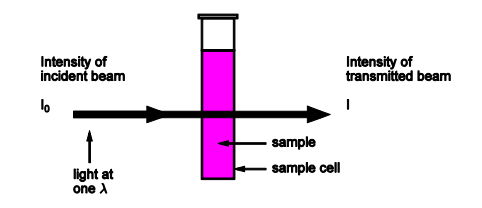
Give equations for transmittance %transmittance and absorbance

Draw out an absorption band for % transmittance and absorbance
Draw out an emission spectrum set up and an emission band
Why do we need selection rules for transition states
A molecule has many energy states, so many transitions are possible between them
Quantum theory tells us which transition can actually occur, and give us, selection rules stating whether transition state are allowed (strong band) or disallowed ( no strong band)
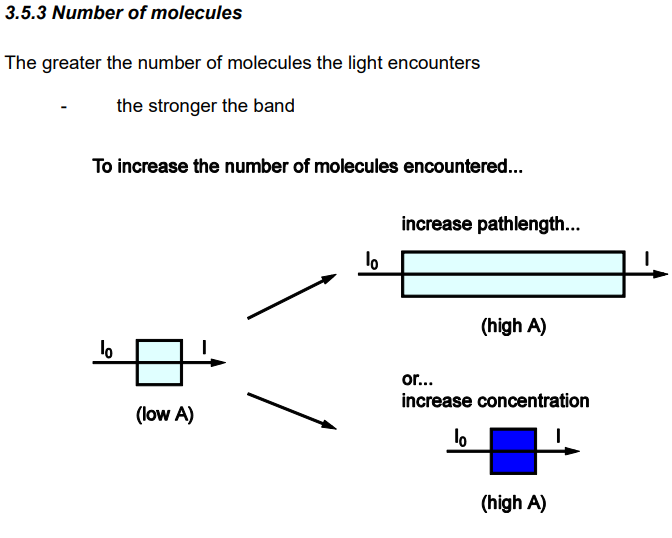
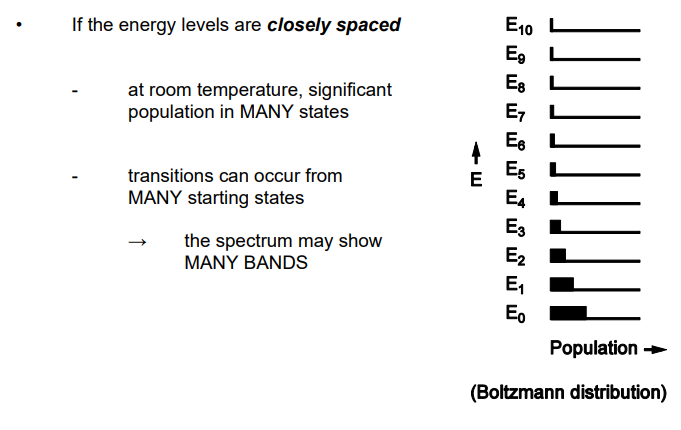
How does number of molecules within energy levels effect transition
The greater the number of molecules in the level from which the transition starts the stronger the band arising from this transition
Describe widely spaced energy levels

Describe closley spaced energy levels
How do we increase the number of molecules encountered by a spectrometer