3.1D Properties of Oxides
1/12
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
What is a Bronsted-Lowry acid?
a substance that donates protons, H⁺
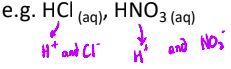
What is a Bronsted-Lowry base?
a substance that accepts protons, H⁺
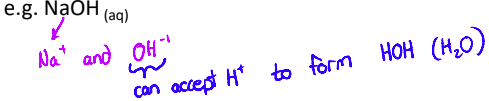
What is another way to describe acids and bases? How do Lewis acids and bases relate?
their ability to accept or donate a pair of electrons.
A Lewis acid can accept an electron pair
e.g. H⁺, Fe²⁺, Al³⁺
A Lewis base can donate an electron pair
Cl⁻, OH⁻ , NH₃
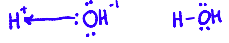
Many metal oxides are what? They react with _____ to form hydroxides by doing what?
Lewis bases
water
donating an electron pair to hydrogen in water
What are the general equations for the reactions between alkali and alkaline earth metals and water?
Reactions of alkali metal oxides with water have the general equation
M₂O(s) + H₂O(l) → 2 MOH (aq)
Reactions of group 2 metal oxides with water have the general equation
MO(s) + H₂O(l) → M(OH)₂ (aq)
Non-metallic oxides are what? They react with water to form _____ _____ by doing what?
Lewis acids
other acids
accepting an electron pair from oxygen in water
What are the general equations for the reactions between non-metallic oxides and water?
xₙOₙ(s) + H₂O(l) → HₙxOₙ(aq)
e.g.
CO₂(g) + H₂O(l) → H₂CO₃(aq)
SO₃(l) + H₂O(l) → H₂SO₄ (aq)
PO₄(s) + 6H₂O(l) → 4 H₃PO₄(aq)
What are Amphoteric substances? What is an example of an amphoteric substance?
substances that are able to behave both as a Lewis acid and a Lewis base.
Aluminum oxide
Aluminum acts as a Lewis base with ____ and a Lewis acid with ___
Aluminum oxide acts as a Lewis base when reacting with NaOH(aq)
Al₂O₃ (s) + 2 NaOH(aq) + 3 H₂O(l) → 2 Na[Al(OH)₄](aq)
Aluminum oxide acts as a Lewis acid when reacting with HCl (aq)
Al₂O₃ (s) + 6 HCl (aq) → 2 AlCl₃ + 3 H₂O(l)
What is the usual trend?
Going across a period, the oxides of the elements become less basic and more acidic.
Oxide | Na₂O | MgO | Al₂O₃ | SiO₂ | P₄O₁₀ | SO₃ |
|---|---|---|---|---|---|---|
Acid or Base? | Basic | Basic | Amph-oteric | Acidic | Acidic | Acidic |
What is pure water’s pH? Why is rainwater naturally acidic? What is the pH of the created product? What is the equation of this reaction? What is acid rain and what creates it?
7.0
Rainwater is naturally acidic due to the presence of dissolved CO2 which forms carbonic acid. It has a pH of 5.6
H₂O (l) + CO₂ (g) → H₂CO₃ (aq)
Oxides of sulfur and nitrogen are more acidic than oxides of carbon. When these gases dissolve in rainwater, the rain is more acidic than normal. This is known as acid rain, which has a pH of less than 5.6
What two ways can sulfur and nitrogen oxides be produced?
naturally by volcanic eruptions and decomposing vegetation
released as pollutants from industrial processes such as the burning of fossil fuels.
How are oceans affected by changes in acidity?
Oceans absorb a large proportion of the CO2 released into the atmosphere. As a result, carbonic acid is formed in the ocean. The increased acidity can affect biodiversity, including coral reefs and shellfish