Chapter 4: Carbon & the Molecular Diversity of Life
0.0(0)Studied by 0 people
Card Sorting
1/5
Earn XP
Last updated 6:46 AM on 3/14/26
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
6 Terms
1
New cards
How many bonds can carbon form?
4 bonds (single bond = +1, double bond = +2, triple bond = +3)
2
New cards
What does Carbon usually bond to?
Hydrogen
Oxygen
Nitrogen
Other Carbon
3
New cards
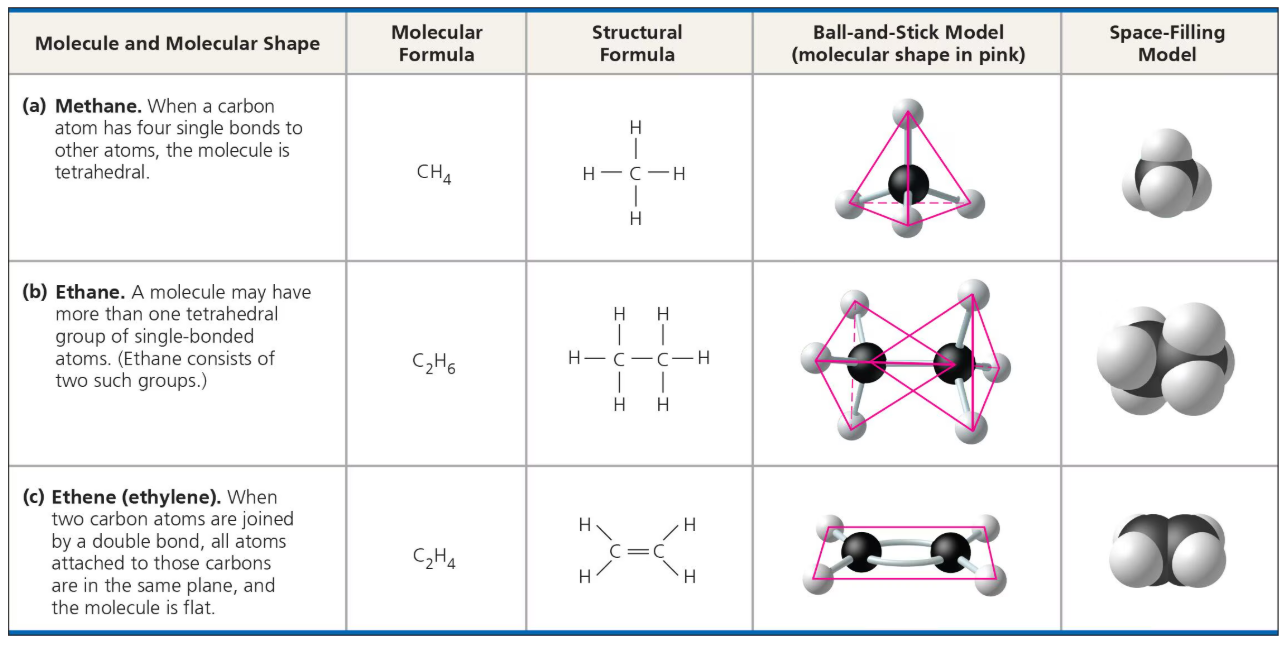
Carbon’s usual molecular geometry?
Tetrahedral
4
New cards
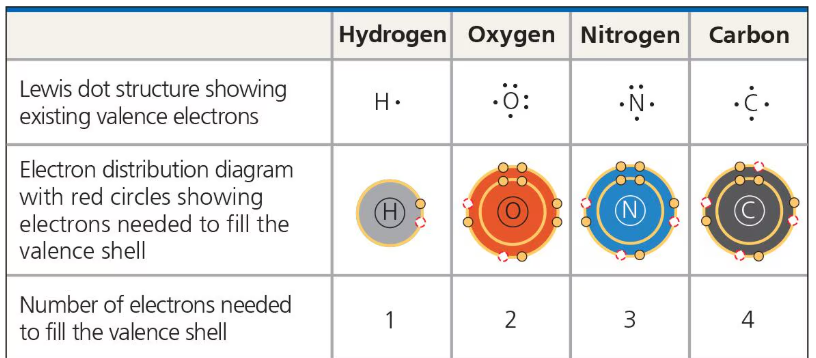
Valence
the number of covalent bonds an atom can form
Generally equal to the number of e- required to fill an atom’s outermost shell
5
New cards
Organic Chemistry
The study of compounds containing carbon
6
New cards
Basis of most organic molecules?
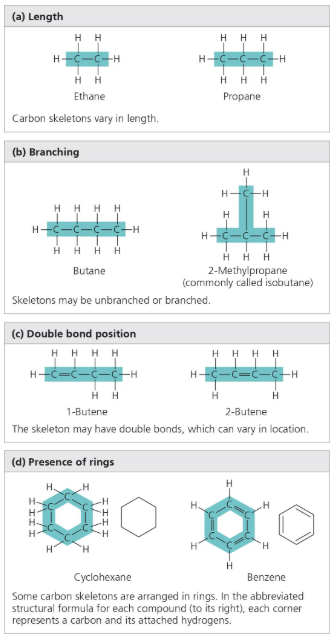
Carbon Chains
Vary in:
Length
Arrangement: straight, branched, and cyclic