chemistry C2 (excluding 2.6)
1/24
Earn XP
Description and Tags
development of the periodic table, electronic structures and the periodic table, group 1, group 7, trends in the periodic table.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
john newland`s contribution to the development of the periodic table in 1864
he noticed that when elements were placed in order of their atomic weights there were sometimes similarities between every 8th element.
produced a table showing his “law of octaves”
he did not leave gaps for undiscovered elements.

john dalton`s contribution to the development of the periodic table in 1808
he arranged the elements in order of their atomic weights


Dmitri mendleev`s contribution to the development of the periodic table in 1869
rearranged elements into order or increasing atomic weight so that a pattern in properties could be seen
left gaps for elements that hadnt been discovered
predicted the properties of these undiscovered elements, and when they were discovered they had similar properties to his predictions
because of this, his table was accepted by other scientists.

Niels bohr`s contribution to the development of the periodic table, 1913
explained that an elements position in the periodic table is determined by its electron configuration
discovered shells, and that elements in the same column have the same number of electrons in their outer shell
this provided a theoretical reason why elements in the same group share similar chemical properties.
james chadwicks contribution to the development of the periodic table, 1932
discovered the neutron.
this proved that isotopes are simply atoms of the same element with different numbers of neutrons.
his work finally allowed scientists to calculate accurate atomic weights, solidifying the arrangement of the periodic table today.
describe the modern periodic table
ordered in increasing atomic number
groups- similar chemical properties, same number of electrons in outer shell
periods- the same number of shells
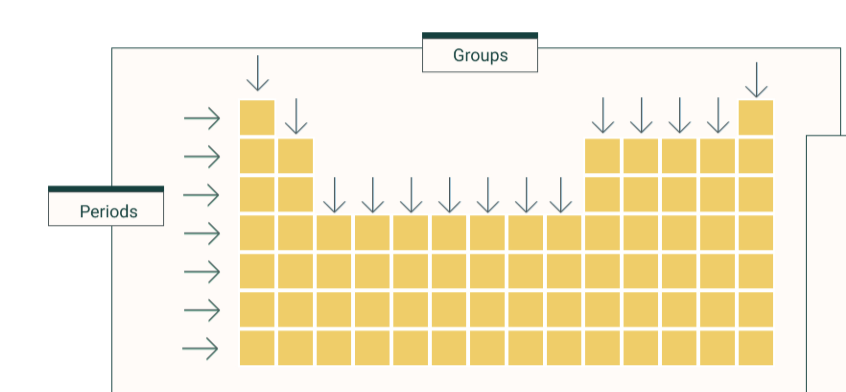
what is a group
column
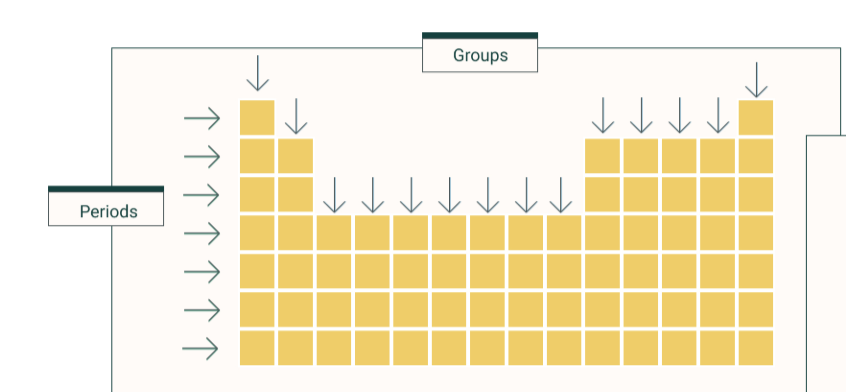
what is a period
row
what are group 0 elements
very unreactive non-metal gases, noble gases
what are group 1 elements
very reactive metals, alkali metals
what are group 2 elements
metals that form ions with a 2+ charge, alkaline earth metals
what are group 7 elements
very reactive non-metals, halogens
what are transition metals
typical metals
alkali metals become going down the group
alkali metals become more reactive going down the group. also the melting points decrease and they become softer.
explain the trend in group 1
atoms get larger down the group as they have more shells
single electron in the outer shell is attracted less strongly to the positive nucleus
electrostatic attraction with nucleus gets weaker
lose the single electron more easily
how does the trend in group 1 (alkali metals) apply to postassium
more inner shells shield outer electron in potassium, so it loses outer electrons more easily.
observations during the reaction of lithium in water
smoke+ fizzing
floats on water, then dissolves (low density)
observations during the reaction of sodium in water
fizzing+ smoke
turns into a ball
faster reaction that lithium
observations during the reaction of potassium in water
popping sound
smoke
lilac flame
floating on water
fastest reaction
physical properties of group 7 halogens
the melting point increases going down the group
diatomic molecules (contain 2 of the same atom)
the boiling points increase going down the group
the densities increase going down the group
appearance of the halogen flourine
pale yellow gas

appearance of the halogen chlorine
green gas

appearance of the halogen bromine
orange-brown liquid (easily vaporises into a gas)

appearance of the halogen iodine
shiny grey-black crystalline solid that sublimes into a purple vapour

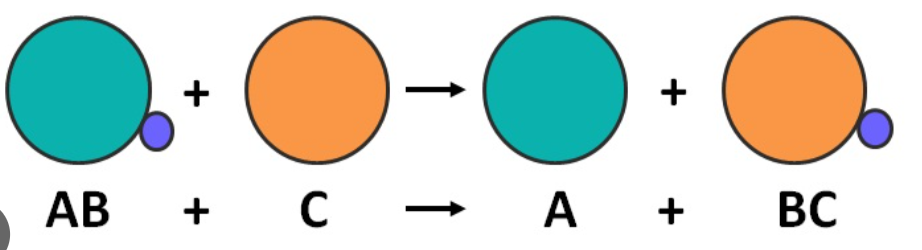
what is a displacement reaction
when a more reactive element displaces a less reactive element.