Chemistry IGCSE - Chemical Energetics
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
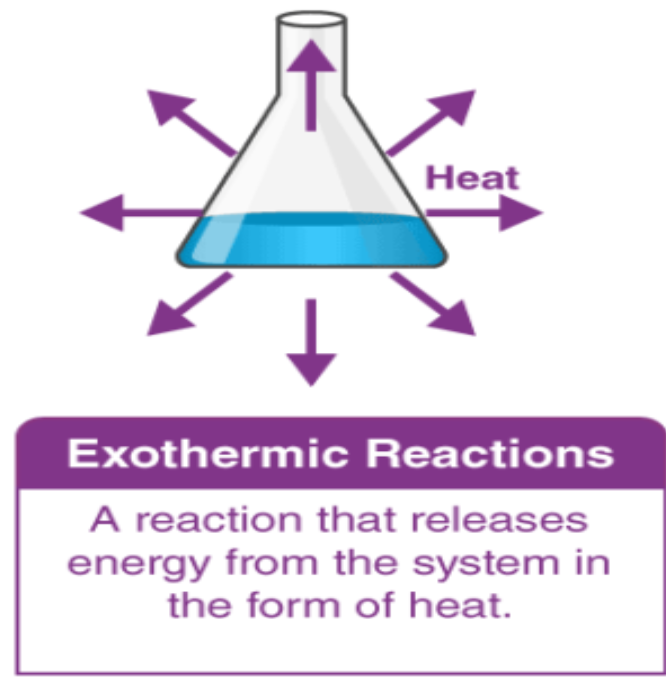
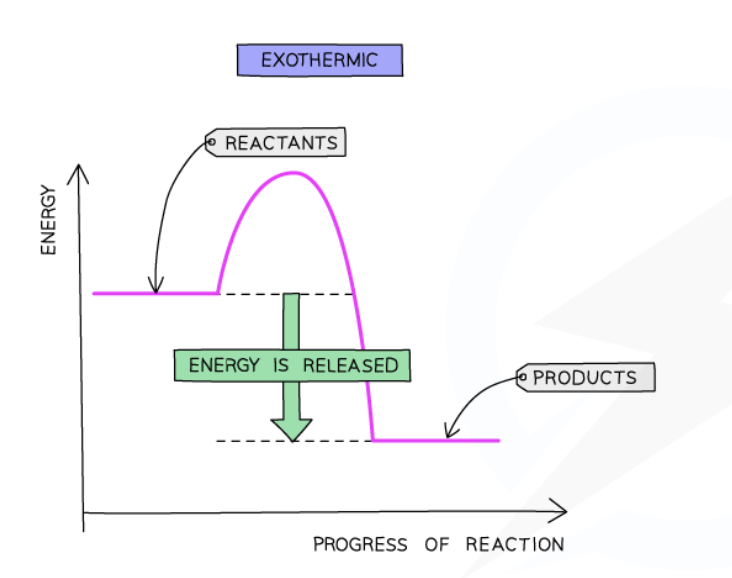
Exothermic reaction
transfers thermal energy to surroundings → increase in temperature of surroundings
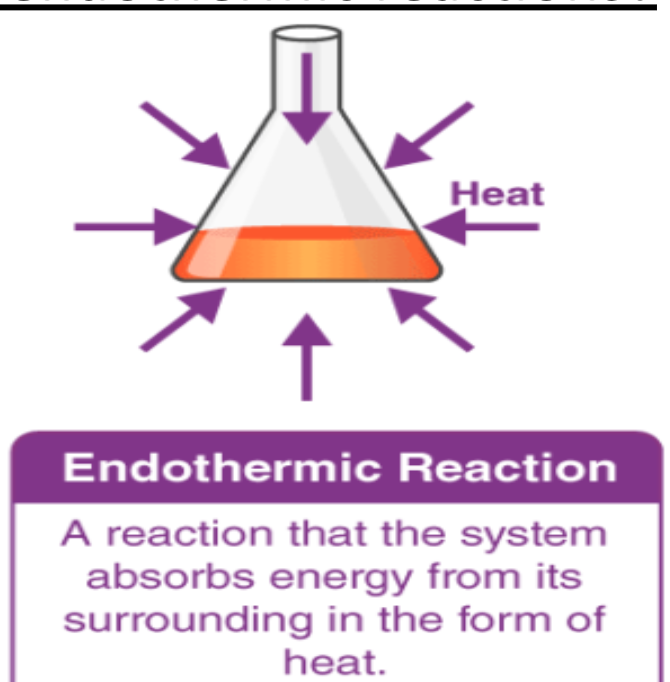
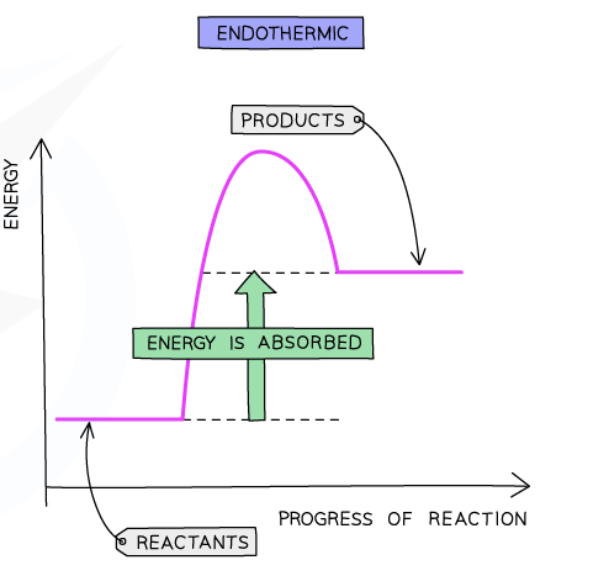
Endothermic reaction
takes in thermal energy from surroundings → decrease in temperature of surroundings
Exothermic bonds
Bond making is a exothermic process
Endothermic bonds
Bond breaking is a endothermic process
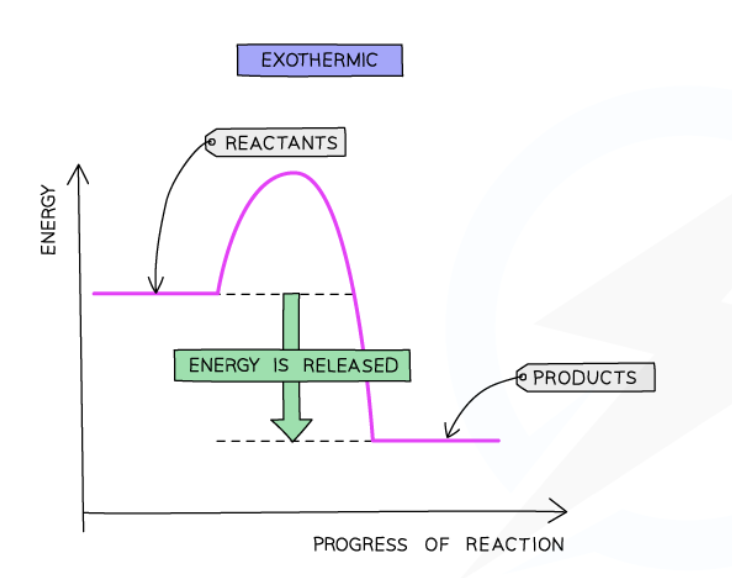
Exothermic reaction pathway diagram
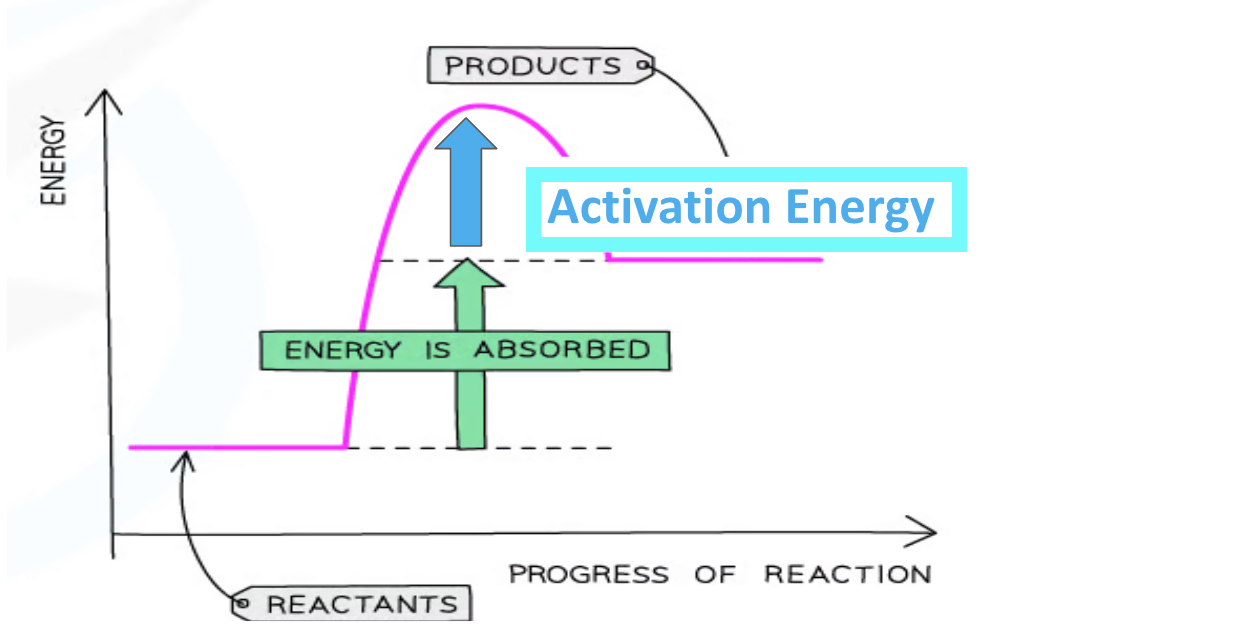
Endothermic reaction pathway diagram
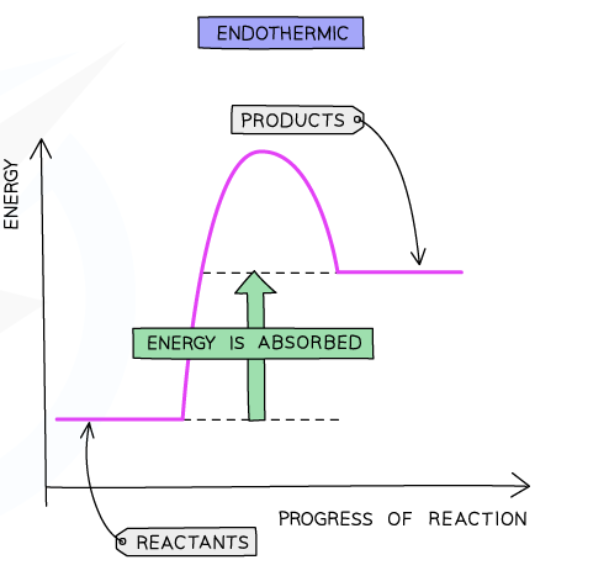
Important labels

ΔH
transfer of thermal energy during a reaction is called → enthalpy change
ΔH in exothermic reactions
Negative for exothermic reactions (green arrow)
ΔH in endothermic reactions
Positive in endothermic reactions (green arrow)
Activation energy (Ea)
minimum energy that colliding particles must have to react