Topic 2- molecular Biology
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
Why is water polar?
Due to unequal electron sharing between oxygen and hydrogen
Why is water a good solvent?
Because polarity allows dissolution of ionic substances
Why is water cohesive?
Hydrogen bonding between molecules
Why is water a good coolant?
Because of a high specific heat capacity
What are the examples of monosaccharides?
Glucose, fructose, and galactose
What are the examples of disaccharides?
Sucrose, lactose, and maltose
What are the examples of polysaccharides?
Starch, glycogen, and cellulose
What is the function of starch?
Energy storage in plants
What is the function of glycogen?
Energy storage in animals
What is the function of cellulose?
Structural support in plant cell walls
What are lipids made from?
Glycerol and three fatty acids
What’s the difference between saturated versus unsaturated fatty acids?
Saturated: no double bonds, solid
Unsaturated: double bonds, liquid
What is the function of lipids?
Long-term energy storage
Insulation
Membrane structure
Hormones
Why are lipids good for energy storage?
High energy per gram
Hydrophobic (so contain no water weight)
What are proteins made from?
Amino acids
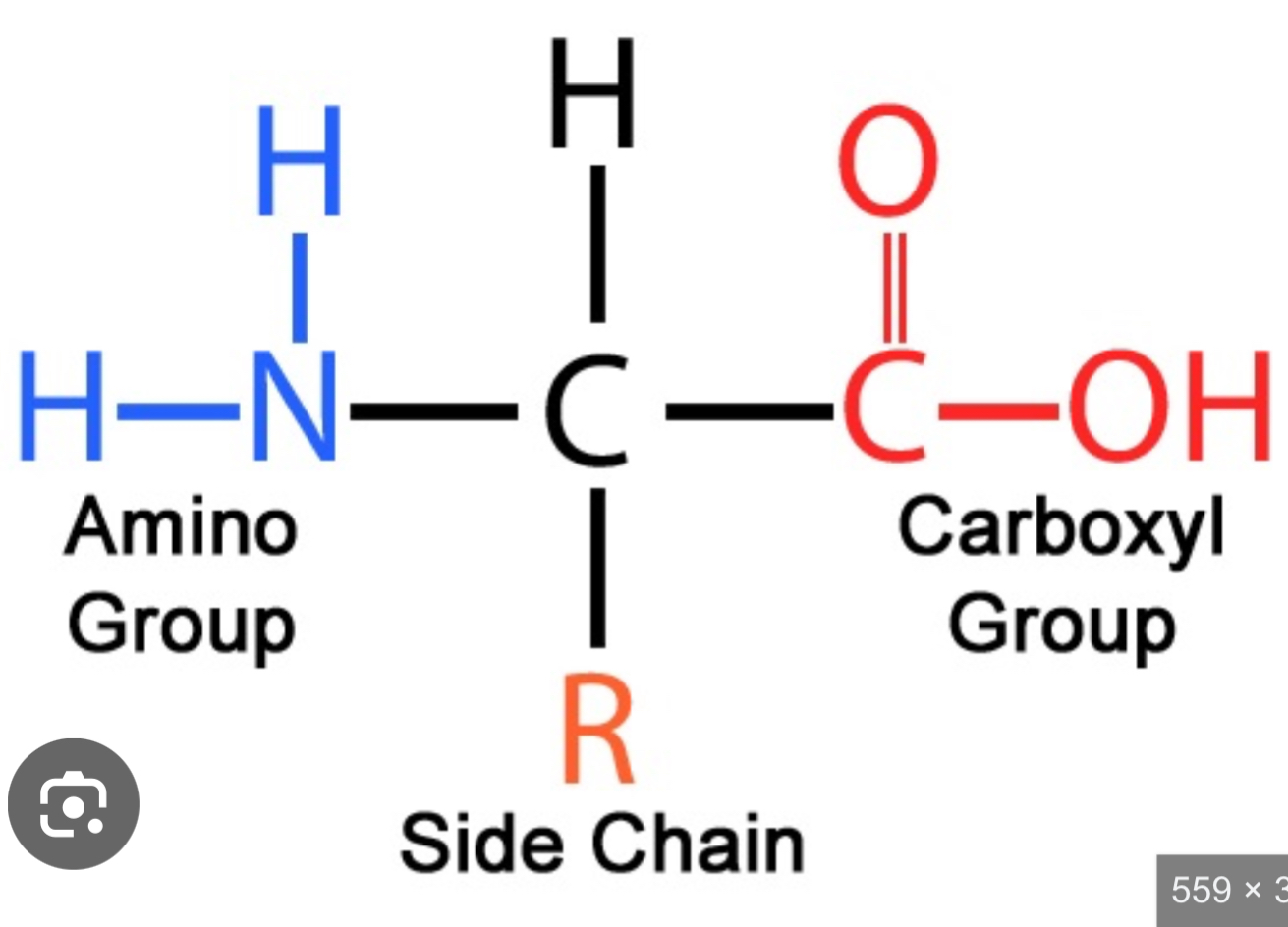
What is the structure of an amino acid?
amino group
Carboxyl group
Hydrogen
R group
Central carbon
What bonds form between amino acids?
Peptide bonds
What is the primary structure?
The sequence of amino acids
What are the secondary structures?
An alpha helix or a beta pleated sheet
What are tertiary structures?
A 3-D folding from our group interactions
What are Quaternary structures?
Multiple polypeptide’s joined together
What are the functions of proteins?
Enzymes
Structure
Transport
Hormones
Immunity
What are enzymes?
Biological catalysts made of proteins
What is an enzyme active site?
A region where a substrate binds
What is the lock and key model?
Where substrates fit exactly into the active site they are made for
What is the induced fit model?
Where an active site changes shape to fit the substrate
What is denaturation?
Change in enzyme shape resulting in a loss of function
What factors affect enzyme activity?
temperature
pH
Substrate concentration
Enzyme concentration
Why does temperature increase enzyme activity?
The higher the temperature, the more collisions between particles
Why does too high a temperature reduce enzyme activity?
Because it results in denaturation
What is a competitive inhibitor?
Substrates that compete for an active site
What is a non-competitive inhibitor?
A substrate that will bind elsewhere or changes she
What does DNA stand for?
Deoxyribonucleic acid
What does DNA nucleotides contain?
Phosphate
Deoxyribose sugar
And amino acid base
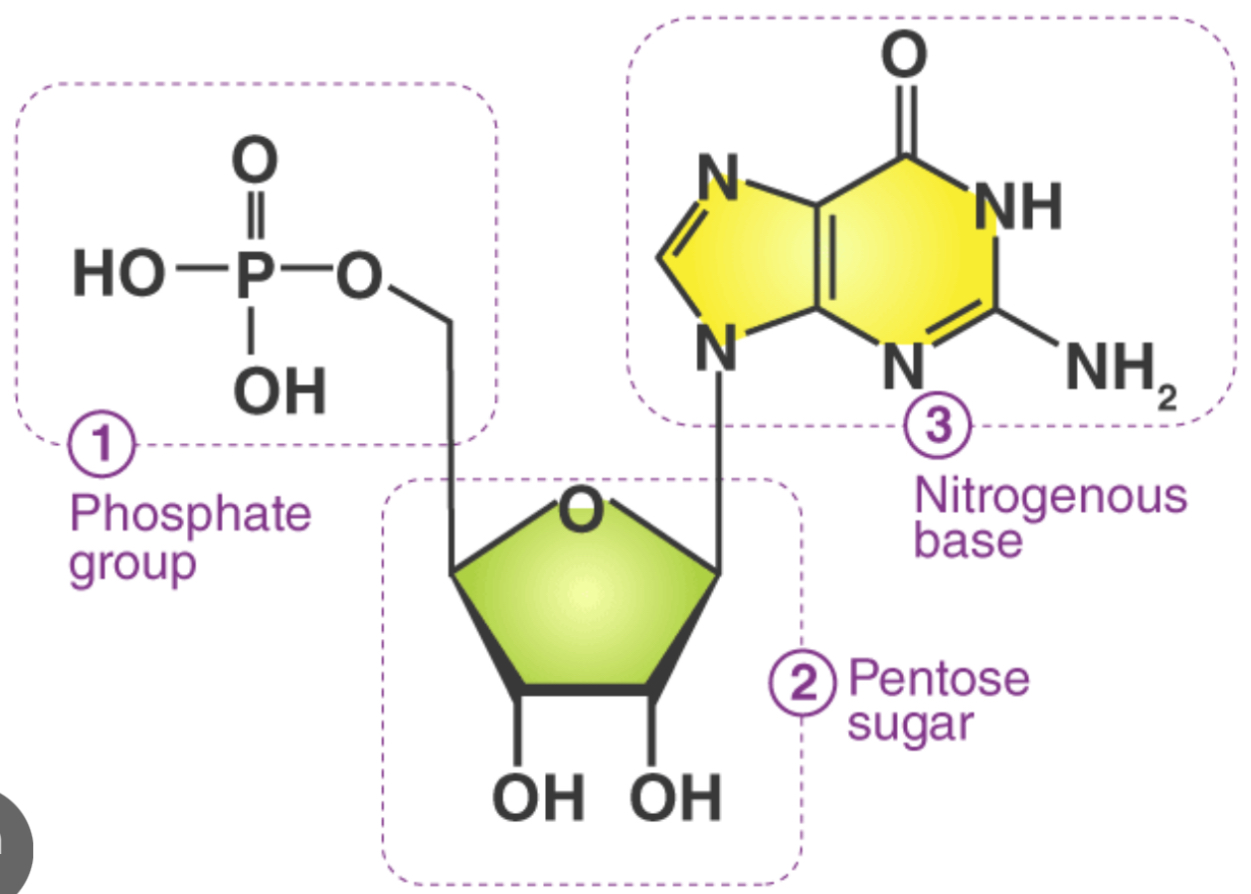
What are the DNA bases?
Adenine, Thymine, Cytosine, and Guanine
What are the DNA base pairings?
Adenine-thymine
Cytosine-guanine
What are the bonds between the DNA bases?
Hydrogen bonds
What is the shape of DNA?
A double helix structure
DNA replication is….
Semi conservative
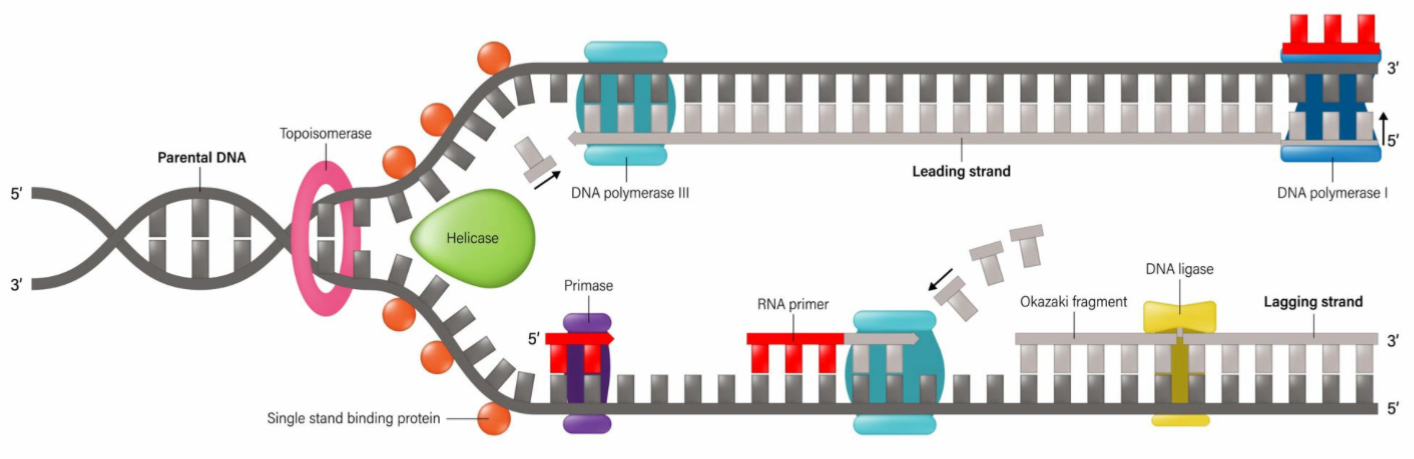
What is the function of helicase?
It unwinds or unzips the DNA’s double helix structure
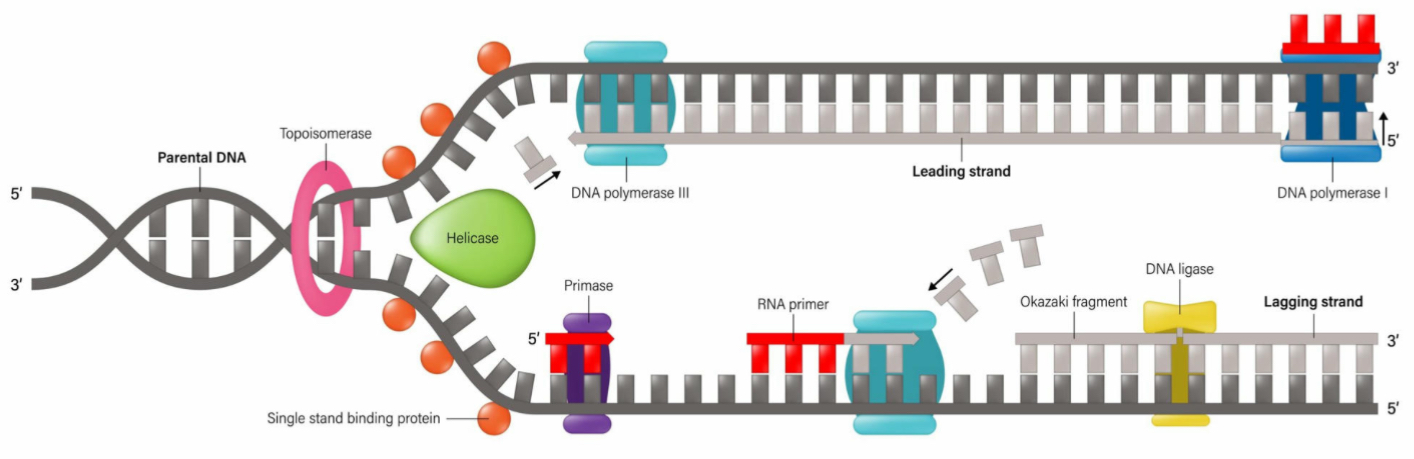
What is the leading strand?
The continuous synthesis
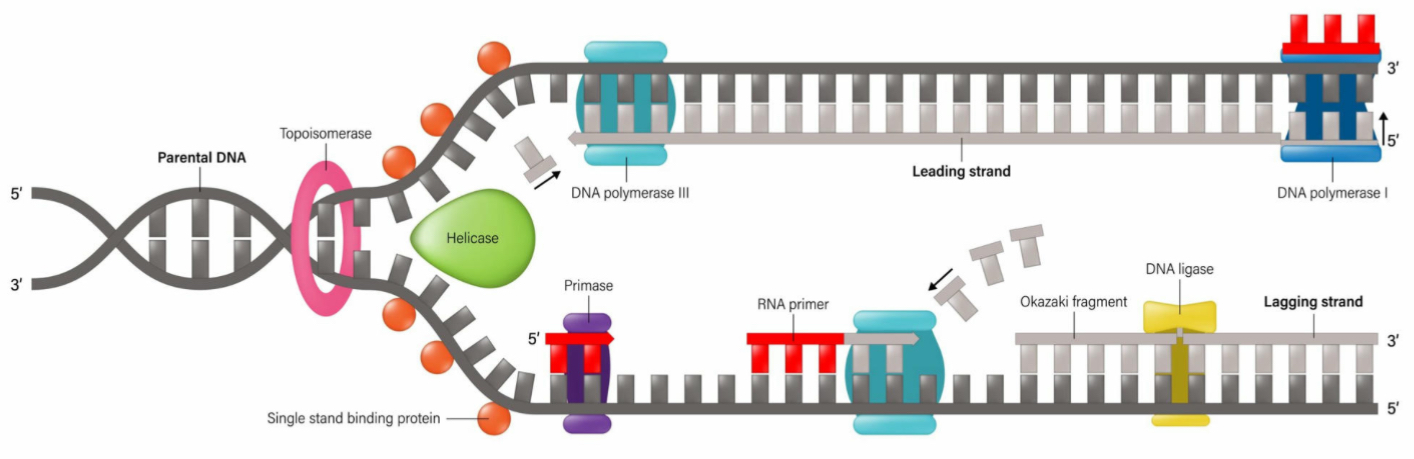
What is the lagging strand?
The strand made from Okazaki fragments
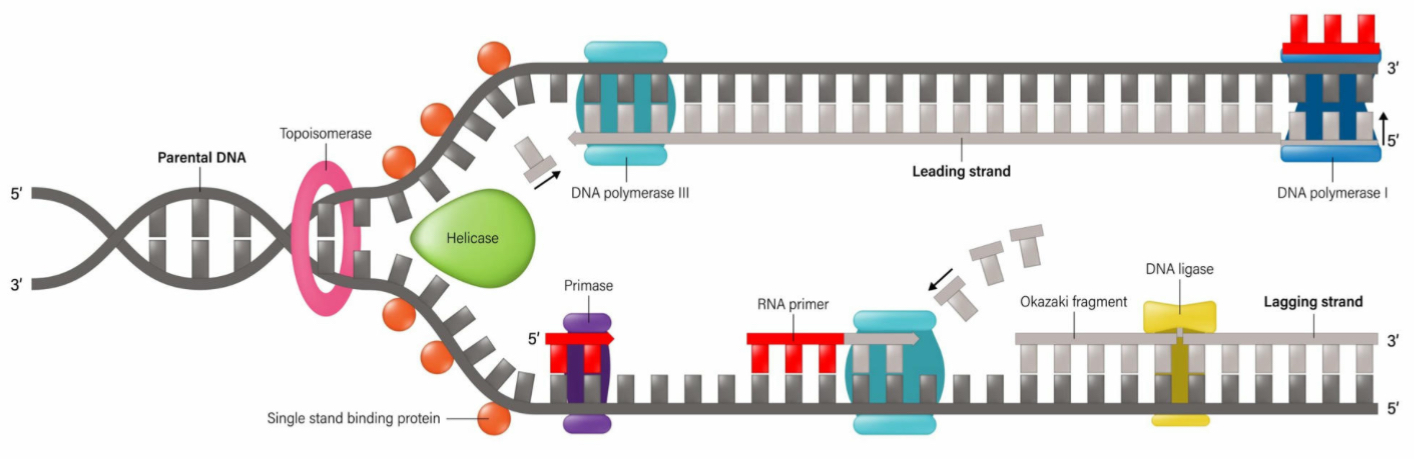
What is the role of DNA Ligase?
It joins the fragments together
What is transcription?
Where DNA is transcribed into messenger RNA (mRNA)
Where does transcription occur?
In the nucleus
What are the RNA bases?
Adenine, uracil, cytosine, guanine
What’s the difference between RNA and DNA?
RNA:
Single stranded
Contains uracil
And ribose sugar
DNA:
Double helix structure
Contains thymine
And deoxyribose sugar
What is translation?
The process where mRNA is changed into proteins
Where does translation occur?
In the ribosomes
What is a codon?
Three bases that code or a coding for an amino acid
What is an anticodon?
Where transfer RNA (tRNA) is a complementary sequence
What is an example of a start codon?
AUG (adenine, uracil, guanine)
What are Some examples of stop codons?
UAA (uracil, adenine, adenine), UAG (uracil, adenine, Guanine), UGA (uracil, guanine, adenine)
What’s the difference between ribose and deoxyribose sugars?
Deoxyribose is missing the oxygen at the 2’ carbon