Phase Transformations Exam 2
1/87
Earn XP
Description and Tags
Interfaces, Nucleation, Groth
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
88 Terms
Three types of crystal interfaces
free surfaces (solid/vapor), grain boundaries, interphase interfaces
Interfacial free energy
excess free energy of a system containing an interface
Ls
latent heat of melting and vaporization
The close-packed surface plane orientations occur at the minimum Esv (the cusp)
True
Esv
solid-vapor interface energy
Vicinal surface
a terraced surface alternating between 2 planes
Wulff plane
a plot in which the free energy of the plane is equal to the distance between the surface and the origin, used to predict the equilibrium shape of crystals
Equilibrium shapes maximize surface energy
False- they minimize excess surface energy
For Wulff construction, planes are perpendicular to the radius vector to each point closest to the origin (and these planes form the equilibrium shape)
True
The shape of crystals becomes spheroidal (instead of faceted) above the recrystallization temperature
False- this occurs above the roughening temperature
Pure tilt grain boundaries
disorientation/rotation axis is contained in the boundary plane
Pure twist grain boundaries
disorientation/rotation axis is perpendicular to the boundary plane
Low-angle GBs
structure can be described by dislocation arrays, surface energy is linearly proportional to theta
High-angle GBs
structure of a configurationally disordered condensed state with excess free volume, surface energy is independent of theta
Theta ranges for LAGB, transition regime, and HAGB
theta<10, 10<theta<15, 15<theta
LAGBs have special angles with cusps of minimized energy
False- HAGBs have these!
Special HAGB angles depend on the disorientation axis and crystal structure
True!
D (plus LAGB equation)
dislocation spacing, D = b/theta
Geometrically necessary dislocations
dislocations required to accommodate LAGB misorientation
Why is HAGB energy constant with theta?
As theta increases, the strain fields overlap and partially cancel
All HAGB have cusps in energy
False- they’re observed in coherent (symmetric tilt GB) and incoherent twins
Symmetric LA tilt GBs are made up of edge dislocations with a single Burgers vector
True
Asymmetric LA tilt GBs are made up of edge dislocations with a single Burgers vector
False- the edge dislocations have multiple Burgers vectors
Coincident site lattice
The overlapping lattice points from overlaid, rotated lattices
Sigma # (CSL) meaning
the number of original lattice sites in the CSL motif
Coherent twin boundaries have the lowest interfacial energy
True!
Asymmetric incoherent twins have terrace-ledge structures
True
CTB, SITB, AITB
coherent twin boundary, symmetric incoherent twin boundary, asymmetric incoherent twin boundary
Grain shapes reduce G by reducing curvature
True!
Grains with less than 4 sides shrink, while grains with more than 4 sides grow (4 is the stable number of sides)
False- 6 is the stable number of sides
Grain growth occurs through the migration of GB
True!
Equation for growth rate (v)
v = MF, M = mobility, F = driving force (proportional to GB energy)
GB segregation
Solute atoms exert lattice strain and concentrate in the GB
Segregation (Xb) increases as T ______ and solubility ______
decreases, decreases
Solutes at GB can exert a drag force to increase GB velocity
False- this decreases GB velocity
CSLs with large sigma values indicate preferred misorientation angles
False- low sigma values are preferred due to their decreased excess energy
In equilibrium, up to 3 grains can meet at the same edge
True- 4 or more grains will be separated by a new edge
interfacial energy of special HAGB is ______ than that of general HAGB
less
GB migration velocity of special HAGB is ______ than that of general HAGB
greater
Zener pinning effect
particles exert a restraining force on GB
Fully coherent
perfect matching of atomic planes at the interface
The interfacial energy of all interfaces is equal to the chemical energy
False- this is only true for fully coherent interfaces
Coherent interfaces can have small geometric mismatches
True- mismatch under 5% is common, and it introduces strain energy!
Semi-coherent interfaces
have a larger mismatch in lattice parameter and use dislocations to reduce strain energy
Interfacial energy for semi-coherent interfaces comes from a chemical contribution and a structural misfit contribution
True
Incoherent interfaces
have a large misfit (>25%), interfacial energy is insensitive to orientation
Fully coherent precipitate shape
matching orientation, spherical
Incoherent precipitate shape
spherical (but other shapes are possible)
Semi-coherent precipitate shape
plane
Coherency is independent of precipitate size
false- coherent precipitates become semi-coherent above the critical radius
Two types of solid-liquid interfaces
flat, diffuse
Interface control
low interface mobility, relative fast diffusion
Diffusion control
high interface mobility, local equilibrium at interface
Nucleation
creation of interface
Growth
migration of interface
Military transformations (thermal sensitivity, interface glissile?, diffusion, rate limiting step)
athermal, glissile, no diffusion, interface control
Civilian transformations (thermal sensitivity, interface glissile?, diffusion, rate limiting step)
thermally activated, non-glissile, short/long-range diffusion, interface/diffusion/mixed control)
Military transformation examples (2)
martensite, twinning
Civilian transformation examples (4)
grain growth, bainite, solidification, eutectoid
Homogeneous nucleation
uniform throughout material, needs large driving force
Heterogeneous nucleation
occurs at special locations, needs small driving force
Critical radius
The radius above which free energy decreases and the nucleus grows
delta Gv
driving force, change in free energy per precipitate volume
delta G*
energy barrier, the maximum increase in energy before decreasing to the minimum G
N
nucleation rate
Embryos exist when T is ____ than TN and r is ____ than r*
less, less
Nuclei exist when T is ____ than TN and r is ____ than r*
greater, greater
The energy barrier for homogeneous nucleation is _______ than that of heterogeneous nucleation
greater
Diffuse surface
macroscopically flat, most metals, lower bonding energy, random growth of solid into liquid (continuous growth)
Flat surface
stepped/zig-zagged features, nonmetals, higher bonding energy, growth of solid into liquid at specific sites (lateral growth)
Solid growing into a superheated liquid
heat flow against interface velocity, planar interface
Solid growing into a supercooled liquid
heat flow with interface velocity, cellular/dendritic interface
Three cases of alloy solidification
equilibrium solidification; no diffusion in solid/perfect mixing in liquid; no diffusion in solid/diffusion mixing in liquid
Equilibrium solidification
assumes straight solidus and liquidus lines, infinitely slow cooling, compositions follow phase diagram
No diffusion in solid, perfect mixing in liquid
uniform liquid composition at any time, liquid composition follows liquid line, solid composition changes with position, average solid composition is lower than the solidus line
No diffusion in solid, diffusional mixing in liquid
liquid composition decreases from liquidus line to bulk concentration, system can reach steady state with stable solidification rate, coring in solid
Zones in ingot solidification
chill zone, columnar zone, central zone
Chill zone
rapid undercooling, heterogeneous nucleation at surface defects on mold wall, high nucleation rate
Columnar zone
crystals grow in direction of heat transfer, favors tertiary dendrites to form new primary arms
Occlusion in the chill zone leads to selective growth
True
Equiaxed zone
randomly oriented grains, cavity pipe is formed in narrow freezing ranges
Macrosegregation
composition changes over distance
Inverse segregation
solute-rich liquid flows back between dendrites to compensate for shrinkage
Microsegregation
composition changes on the scale of secondary dendrite arm spacing, mitigated by homogenization heat treatment
Segregation
non-uniform distribution of impurities and alloying elements, depends on chemical composition and rate of cooling
Seeded crystal growth
growth of a crystal with the same structure as the heterogeneous interface
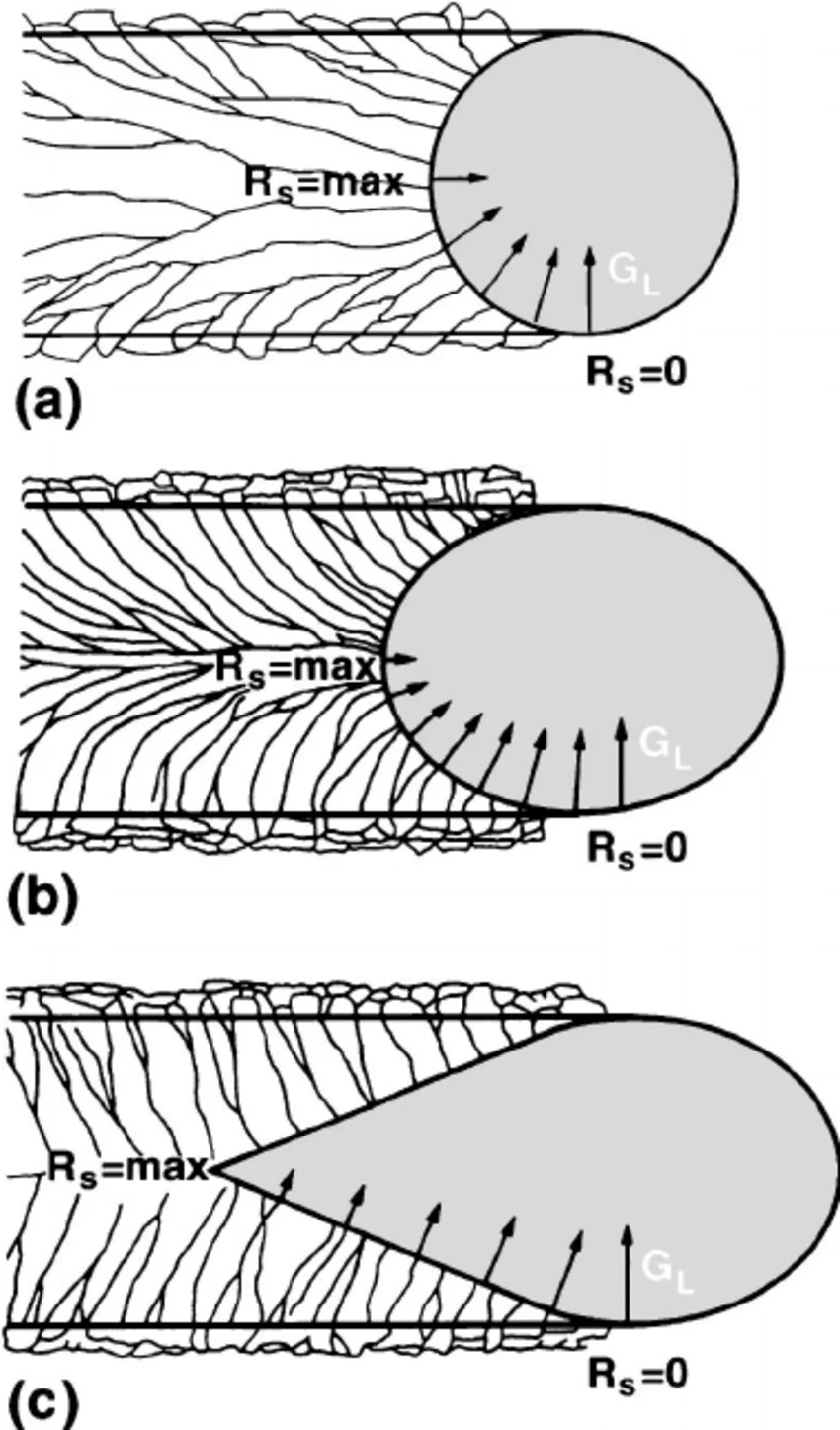
Order of increasing weld speed (tap picture to see the entire thing)
a, b, c
Ls/Tm (delta S) is around ___ for diffuse surfaces and ___ for flat surfaces
R, 4R