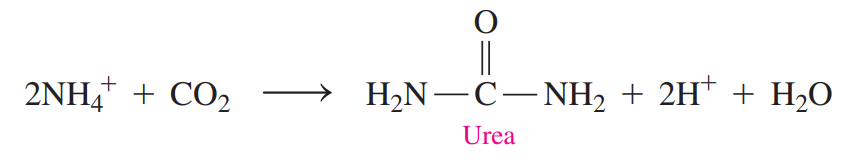Chapter 18: Metabolic Pathways and Energy Production
18.1: Metabolism
- Metabolism: All the chemical reactions that provide energy and the substances required for continued cell growth.
- Catabolic Reactions: These are complex molecules that are broken down into simpler ones with an accompanying release of energy.
- Anabolic Reactions: These utilize the energy available in the cell to build large molecules from simple ones.
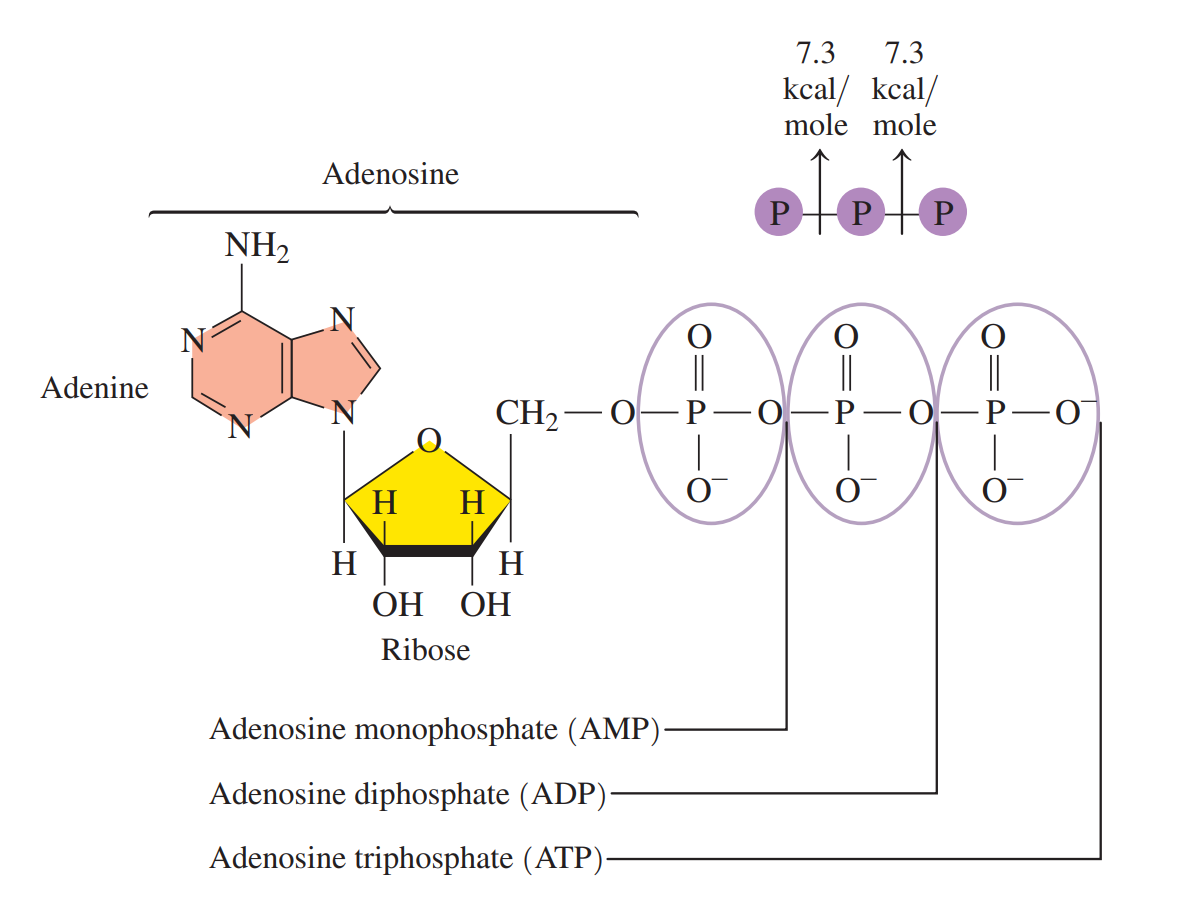
- Adenosine Triphosphate (ATP): A high-energy compound that stores energy in the cells. It consists of adenine, a ribose sugar, and three phosphate groups.
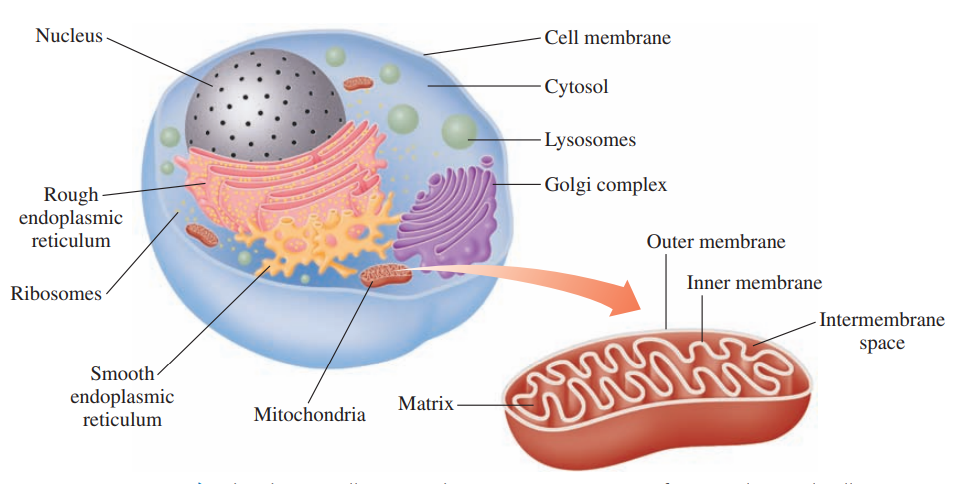
Cell Structure for Metabolism
- Cell membrane: It separates the contents of a cell from the external environment and contains structures that communicate with other cells.
- Cytoplasm: It consists of the cellular contents between the cell membrane and nucleus.
- Cytosol: It is the fluid part of the cytoplasm that contains enzymes for many of the cell’s chemical reactions.
- Endoplasmic reticulum: It is the rough type that processes proteins for secretion and synthesizes phospholipids; smooth type synthesizes fats and steroids.
- Golgi complex: It modifies and secretes proteins from the endoplasmic reticulum and synthesizes cell membranes.
- Lysosome: It contains hydrolytic enzymes that digest and recycle old cell structures.
- Mitochondrion: It contains the structures for the synthesis of ATP from energy-producing reactions.
- Nucleus: It contains genetic information for the replication of DNA and the synthesis of protein.
- Ribosome: It is the site of protein synthesis using mRNA templates.
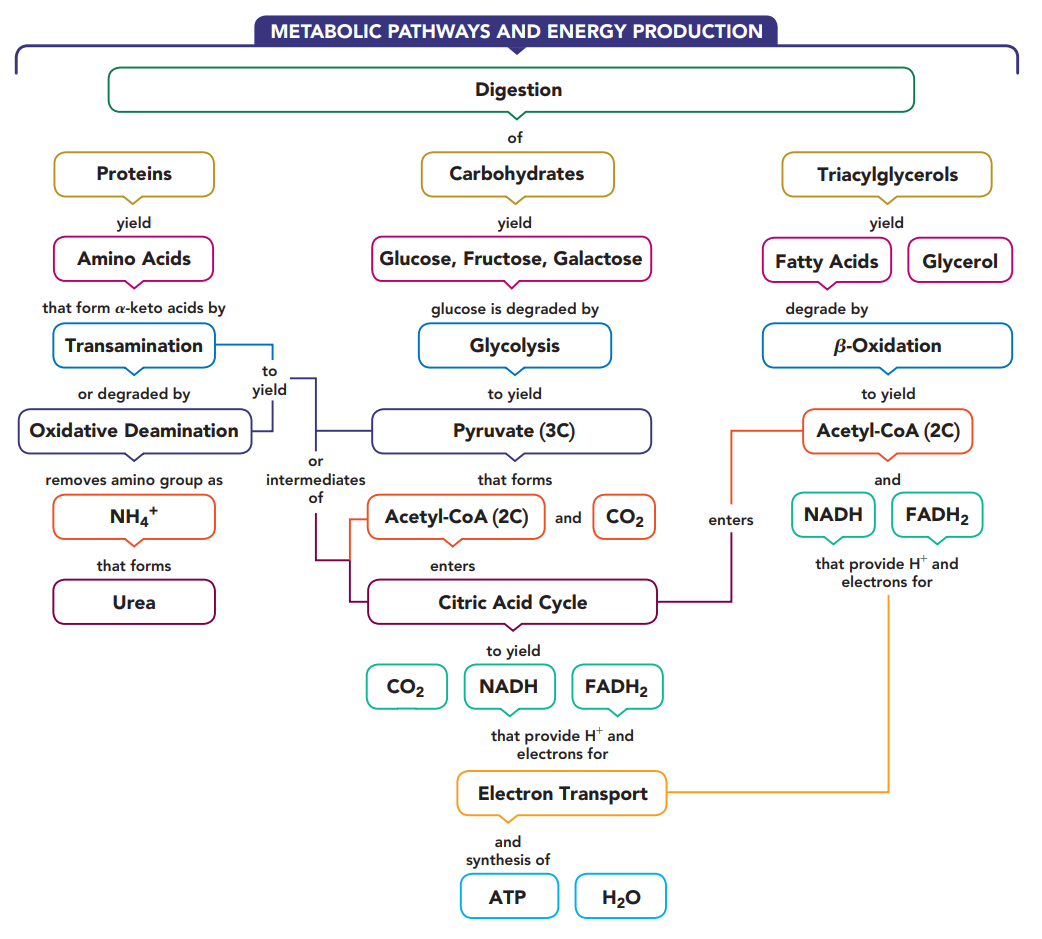
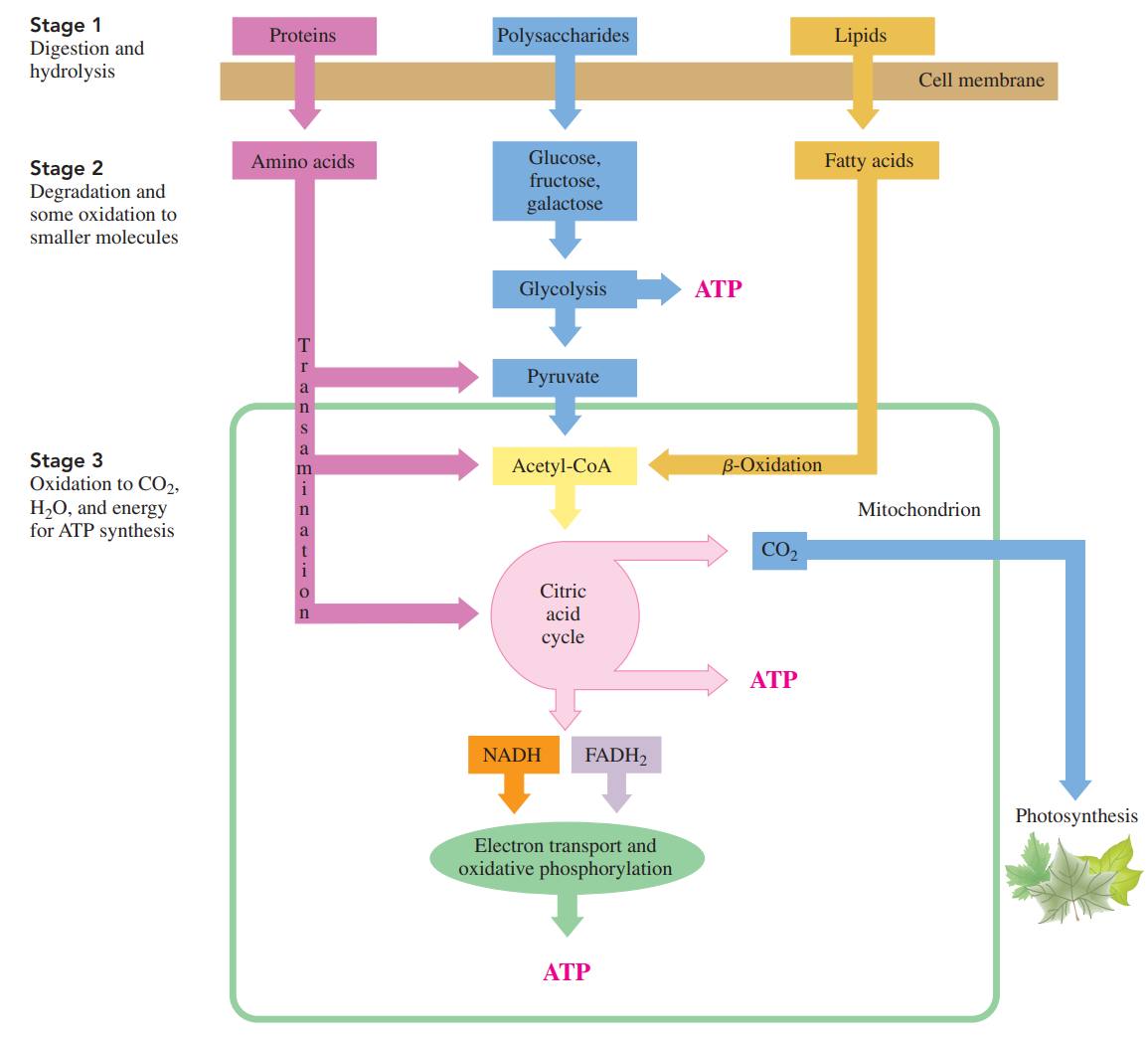
Three Stages of Catabolism
- Catabolism begins with the processes of digestion in which enzymes in the digestive tract break down large molecules into smaller ones.
- The polysaccharides break down to monosaccharides, fats break down to glycerol and fatty acids, and the proteins yield amino acids.
- These digestion products diffuse into the bloodstream for transport to cells.
- Within the cells, catabolic reactions continue as the digestion products are broken down further to yield two- and three-carbon compounds.
- The major production of energy takes place in the mitochondria, as the two-carbon acetyl group is oxidized in the citric acid cycle.
- As long as the cells have oxygen, the hydrogen ions and electrons from the reduced coenzymes are transferred to electron transport to synthesize ATP.
18.2: Digestion of Foods
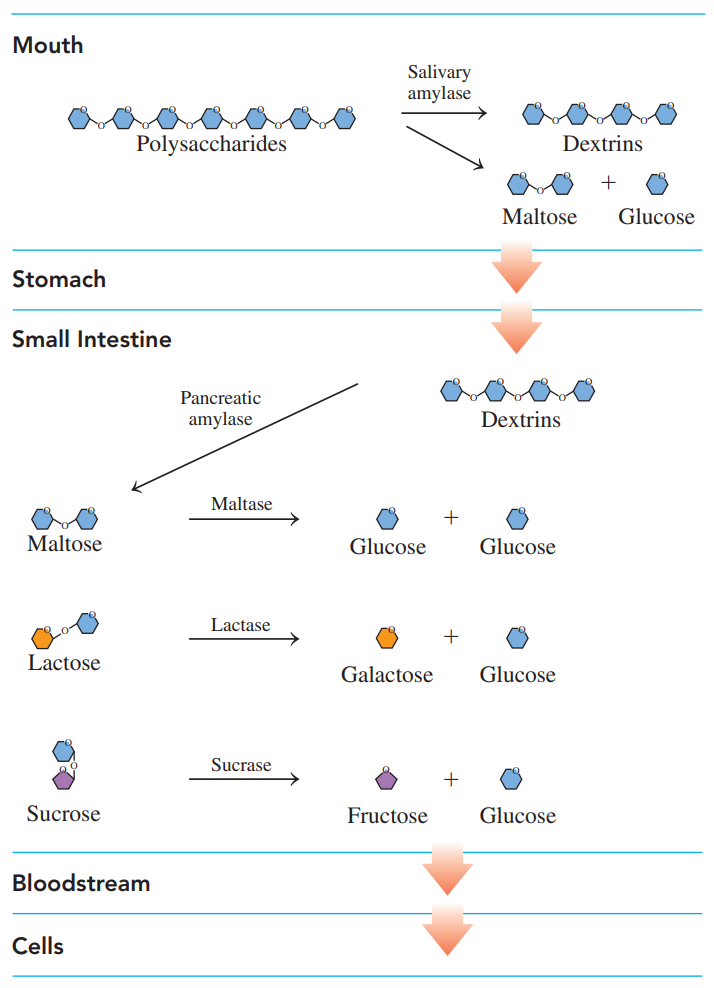
Digestion of Carbohydrates
- Enzymes produced in the salivary glands hydrolyze some of the 𝜶-glycosidic bods in amylose and amylopectin, producing maltose, glucose, and dextrins — which contain three to eight glucose units.
- After swallowing, the partially digested starches enter the acidic environment of the stomach, where the low pH stops carbohydrate digestion.
- In the small intestine, which has a pH of about 8, enzymes produced in the pancreas hydrolyze the remaining dextrins to maltose and glucose.
- Then enzymes produced in the mucosal cells that line the small intestine hydrolyze maltose as well as lactose and sucrose.
- The resulting monosaccharides are absorbed through the intestinal wall into the bloodstream, which carries them to the liver, where the hexose fructose and galactose are converted to glucose.
- Glucose is the primary energy source for muscle contractions, red blood cells, and the brain.
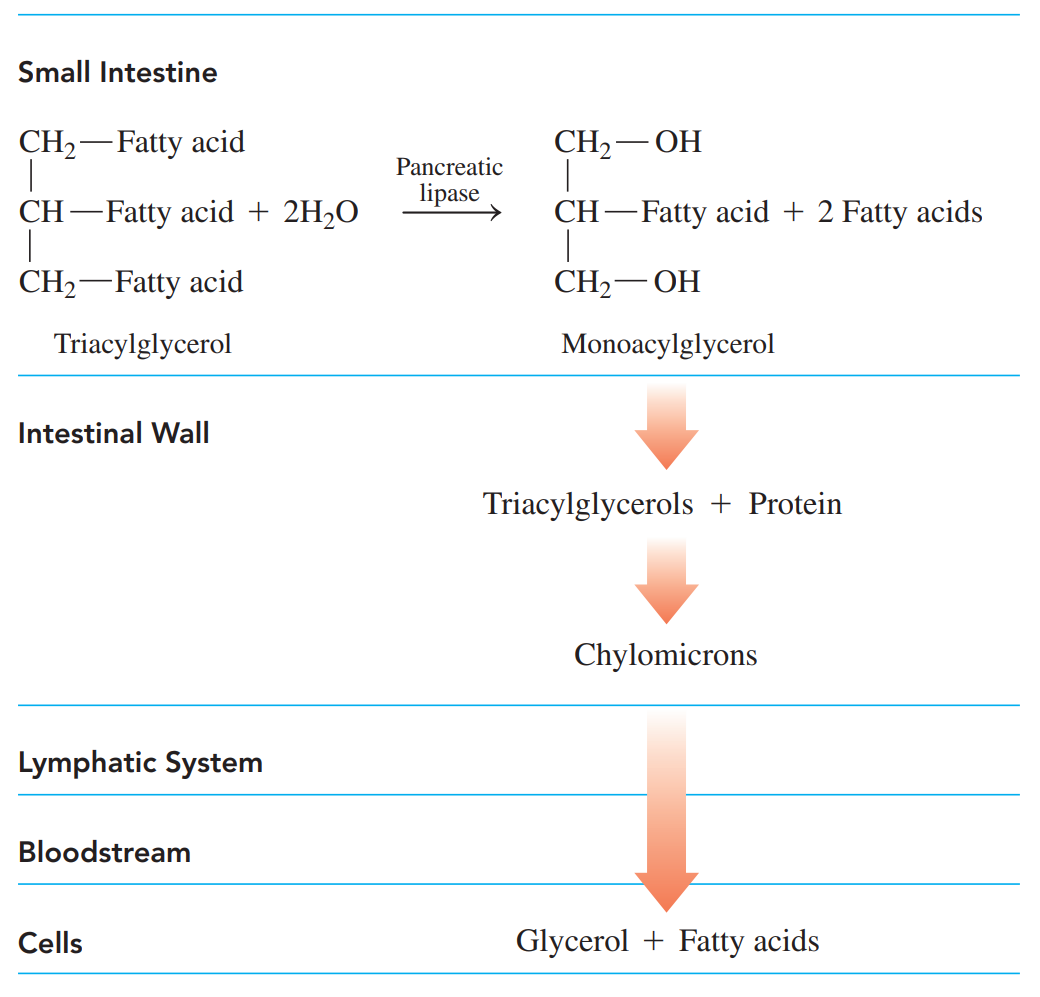
Digestion of Fats
- It begins in the small intestine when the hydrophobic fat globules mix with bile salts released from the gallbladder.
- Emulsification: A process where the bile salts break the fat globules into micelles.
- Enzymes from the pancreas hydrolyze the triacylglycerols to yield monoacylglycerols and fatty acids, which are then absorbed into the intestinal lining where they recombine to form triacylglycerols.
- Chylomicrons: The nonpolar compounds are then coated with proteins to form lipoproteins which are more polar and soluble in the aqueous environment of the lymph and bloodstream.
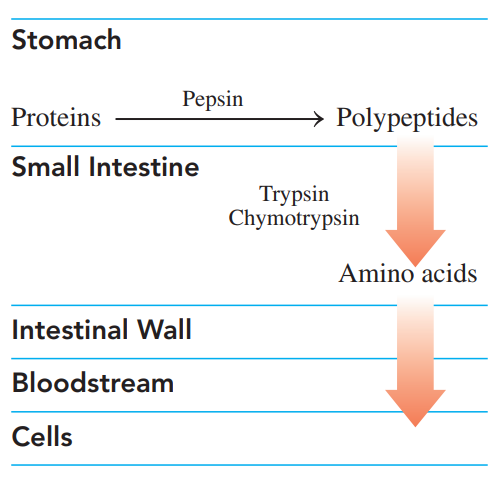
Digestion of Proteins
- It begins in the stomach, where hydrochloric acid at pH 2 denatures the proteins and activates enzymes.
- Polypeptides move out of the stomach into the small intestine, where trypsin and chymotrypsin complete the hydrolysis of the peptides to amino acids.
- The amino acids are absorbed through the intestinal walls into the bloodstream for transport to the cells.
18.3: Coenzymes in Metabolic Pathways
- Oxidation: A reaction that involves the loss of hydrogen or electrons by a substance, or an increase in the number of bonds to oxygen.
- Reduction: A reaction that involves the gain of hydrogen ions and electrons or a decrease in the number of bonds to oxygen.
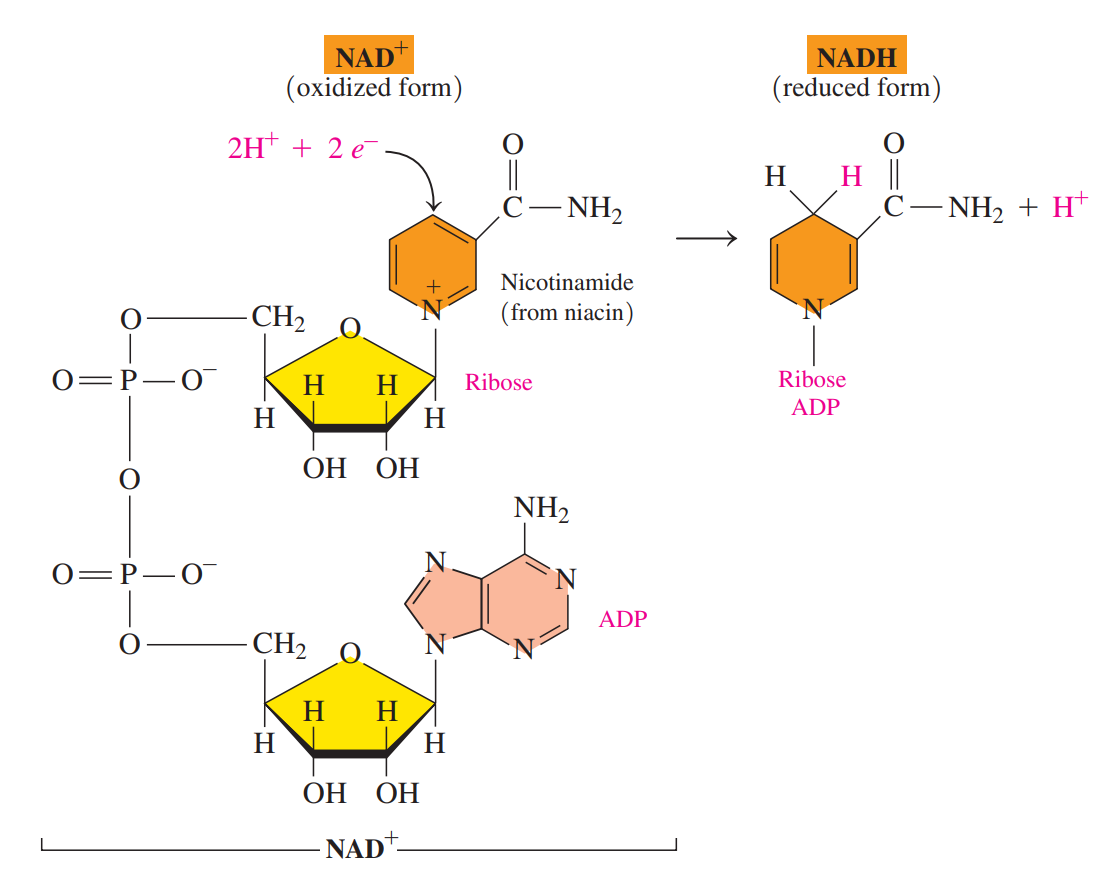
- Nicotinamide adenine dinucleotide (NAD+)
- An important coenzyme in which the vitamin niacin provides the nicotinamide group, which is bonded to ribose and ADP.
- The oxidized NAD+ undergoes reduction when carbon in the nicotinamide ring reacts with 2H, leaving one H+.
- The NAD+ coenzyme is required for metabolic reactions that produce carbon–oxygen double bonds.
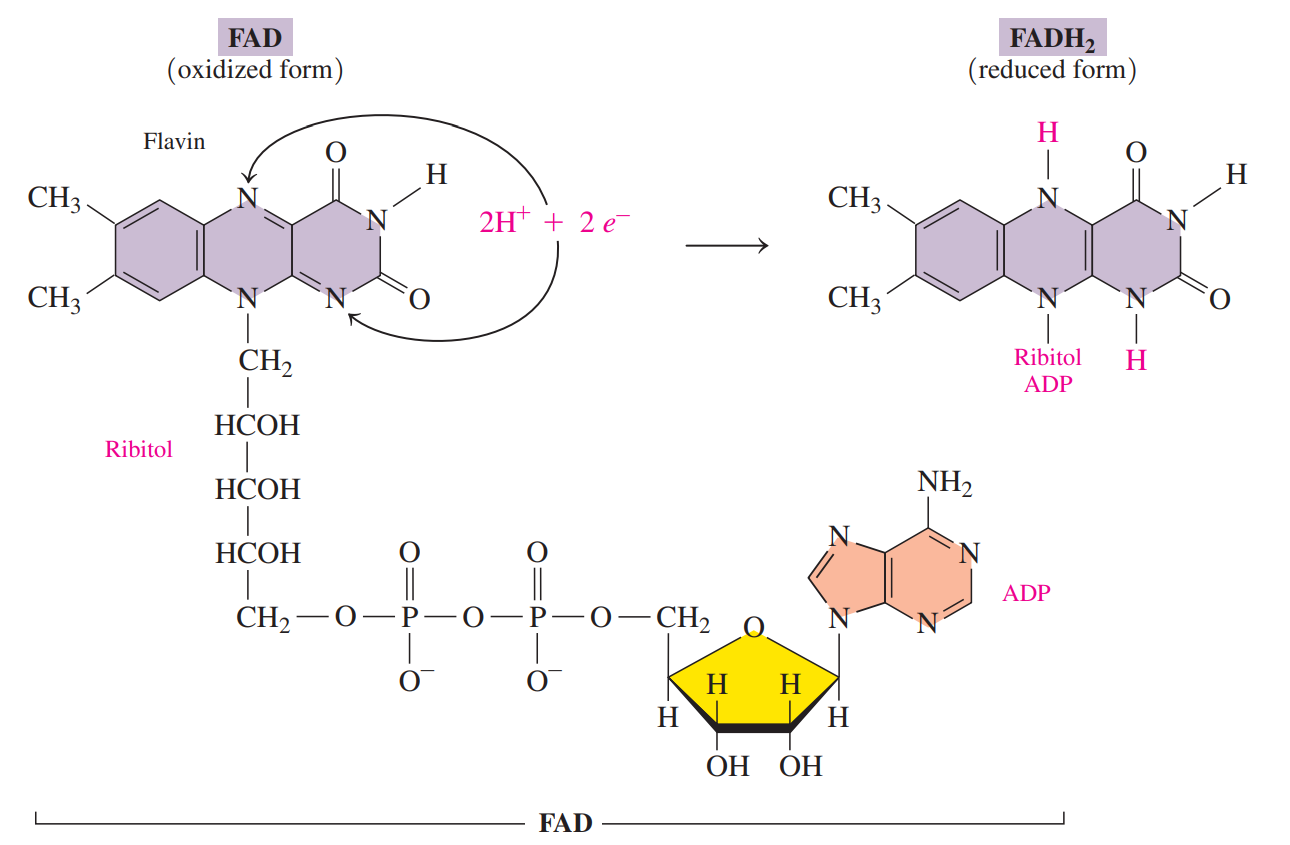
- Flavin adenine dinucleotide (FAD)
- A coenzyme that contains ADP and riboflavin.
- Riboflavin: Also known as Vitamin B2, consists of ribitol and flavin.
- The oxidized form of FAD undergoes reduction when the two nitrogen atoms in the flavin part of the FAD coenzyme react with 2H reducing FAD to FADH2.
- It is used as a coenzyme when an oxidation reaction converts a carbon–carbon single bond to a carbon–carbon double bond.
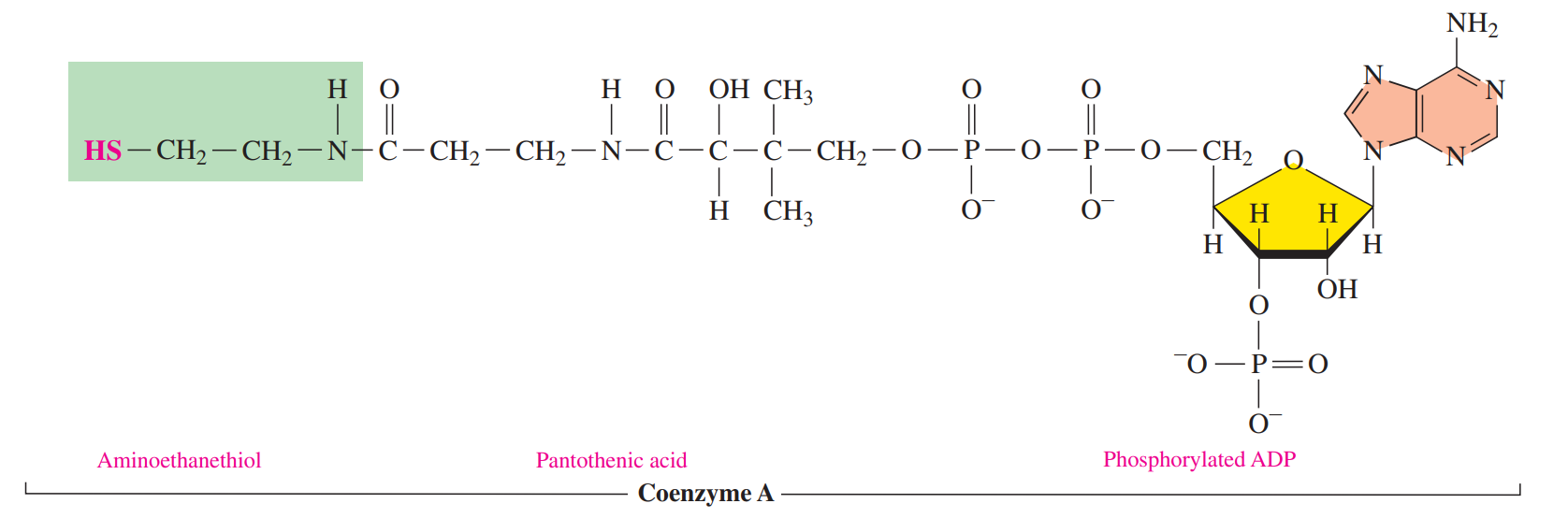
- Coenzyme A
- Its function is to prepare small acyl groups for reactions with enzymes.
- The reactive feature of coenzyme A is the thiol group which bonds to a two-carbon acetyl group to produce the energy-rich thioester acetyl-CoA.
18.4: Glycolysis: Oxidation of Glucose
- Glycolysis
- A pathway wherein the glucose in the bloodstream enters our cells where it undergoes degradation.
- It is an anaerobic process; no oxygen is required.
- A six-carbon glucose molecule is broken down to two molecules of three-carbon pyruvate.
- All the reactions in glycolysis take place in the cytoplasm of the cell.
- Energy-investing phase: The energy is obtained from the hydrolysis of two ATP, which is needed to form sugar phosphates; the first five reactions.
- In reactions 4 and 5, a six-carbon sugar phosphate is split to yield two molecules of three-carbon sugar phosphate.
- Energy-generating phase: The energy is obtained from the hydrolysis of the energy-rich phosphate compounds and used to synthesize four ATP; the last five reactions (6-10).
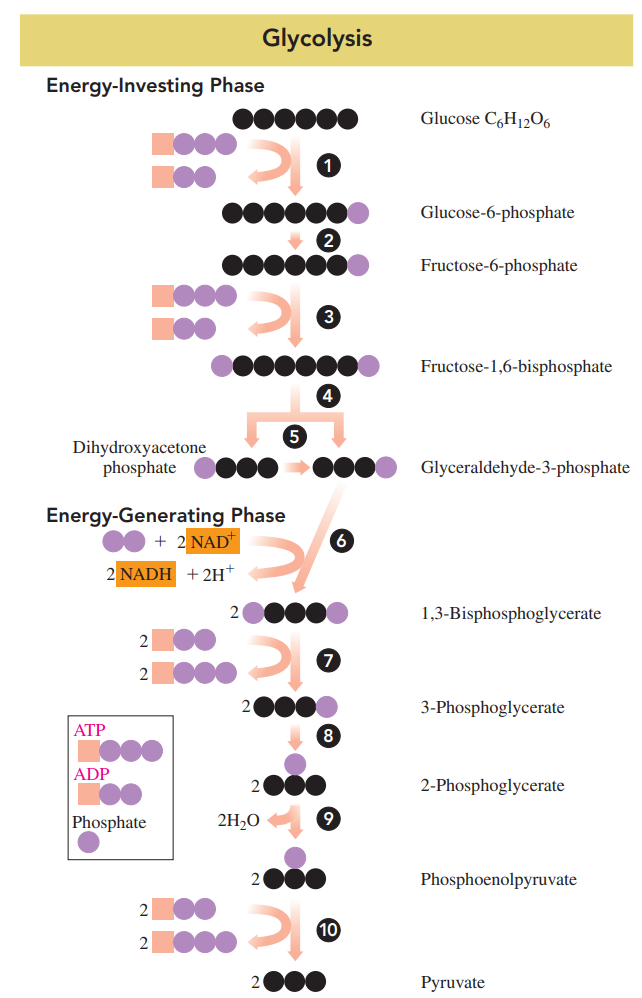
Energy-Investing Reactions 1 to 5
- Reaction 1: Phosphorylation
- In the initial reaction, a phosphate group from ATP is added to glucose to form glucose6-phosphate and ADP.
- Reaction 2: Isomerization
- The glucose-6-phosphate, the aldose from reaction 1, undergoes isomerization to fructose6-phosphate, which is a ketose.
- Reaction 3: Phosphorylation
- The hydrolysis of another ATP provides a second phosphate group, which converts fructose-6-phosphate to fructose-1,6-bisphosphate.
- Reaction 4: Cleavage
- Fructose-1,6-bisphosphate is split into two three-carbon phosphate isomers: dihydroxyacetone phosphate and glyceraldehyde-3-phosphate.
- Reaction 5: Isomerization
- Because dihydroxyacetone phosphate is a ketone, it cannot react further. However, it undergoes isomerization to provide a second molecule of glyceraldehyde-3-phosphate, which can be oxidized.
Energy-Generating Reactions 6 to 10
- Reaction 6: Oxidation and Phosphorylation
- The aldehyde group of each glyceraldehyde-3-phosphate is oxidized to a carboxyl group by the coenzyme NAD+, which is reduced to NADH and H+.
- A phosphate group adds to each of the new carboxyl groups to form two molecules of the high-energy compound, 1,3-bisphosphoglycerate.
- Reaction 7: Phosphate Transfer
- Phosphorylation transfers a phosphate group from each 1,3-bisphosphoglycerate to ADP to produce two molecules of the high-energy compound ATP.
- At this point in glycolysis, two ATP are produced, which balance the two ATP consumed in reactions 1 and 3.
- Reaction 8: Isomerization
- Two 3-phosphoglycerate molecules undergo isomerization, which moves the phosphate group from carbon 3 to carbon 2 yielding two molecules of 2-phosphoglycerate.
- Reaction 9: Dehydration
- Each of the phosphoglycerate molecules undergoes dehydration 1loss of water2 to give two high-energy molecules of phosphoenolpyruvate.
- Reaction 10: Phosphate Transfer
- In a second direct phosphorylation, phosphate groups from two phosphoenolpyruvate are transferred to two ADPs to form two pyruvate and two ATP.
Pathways for Pyruvate
- The pyruvate produced from glucose can now enter pathways that continue to extract energy.
- Aerobic Conditions
- In glycolysis, two ATP were generated when one glucose molecule was converted to two pyruvates.
- Under these conditions, pyruvate moves from the cytoplasm into the mitochondria to be oxidized further.
- In a complex reaction, pyruvate is oxidized, and a carbon atom is removed from pyruvate as CO2.
- The coenzyme NAD+ is reduced during oxidation.
- The resulting two-carbon acetyl compound is attached to CoA, producing acetyl-CoA, an important intermediate in many metabolic pathways
- Anaerobic Conditions
- When we engage in strenuous exercise, the oxygen stored in our muscle cells is quickly depleted.
- Under these conditions, pyruvate remains in the cytoplasm where it is reduced to lactate.
- NAD+ is produced and is used to oxidize more glyceraldehyde3-phosphate in the glycolysis pathway, which produces a small but needed amount of ATP.
18.5: The Citric Acid Cycle
- Citric Acid Cycle: A series of reactions connects the intermediate acetyl-CoA from the metabolic pathways in stages 1 and 2 with electron transport and the synthesis of ATP in stage 3.
- It is also known as the tricarboxylic acid (TCA) cycle or the Krebs cycle.
- Citric Acid: A tricarboxylic acid, forms in the first reaction.
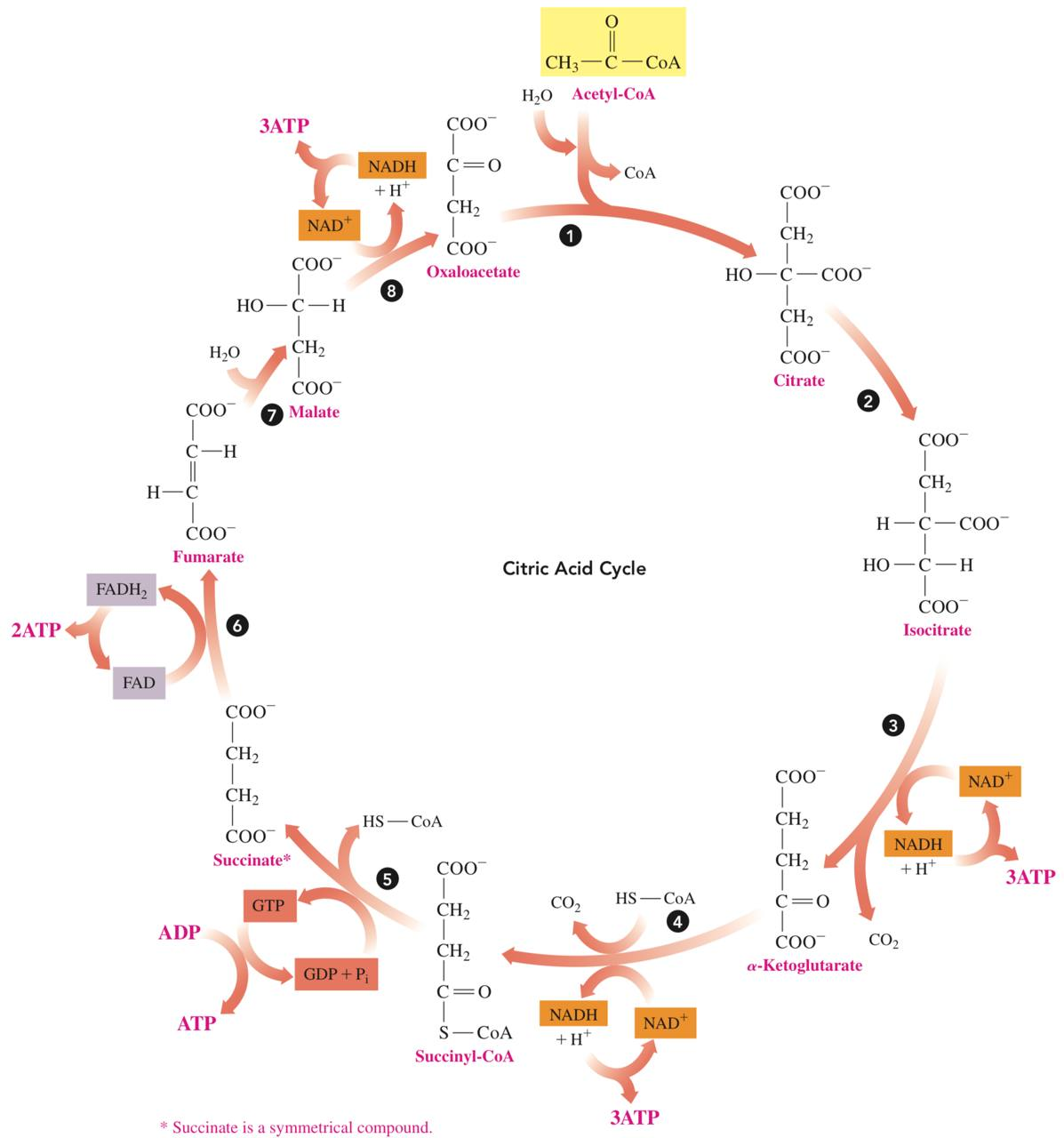
The Cycle
- Reaction 1: Formation of Citrate
- In the first reaction of the citric acid cycle, the acetyl group from acetyl-CoA bonds with oxaloacetate to yield citrate.
- Reaction 2: Isomerization
- The citrate produced in reaction 1 contains a tertiary alcohol group that cannot be oxidized further
- The citrate undergoes isomerization to yield its isomer isocitrate, which provides a secondary alcohol group that can be oxidized in the next reaction.
- Reaction 3: Oxidation and Decarboxylation
- The secondary alcohol group in isocitrate is oxidized to a ketone.
- A decarboxylation converts a carboxylate group to a CO2 molecule producing 𝜶-ketoglutarate.
- The oxidation reaction also produces hydrogen ions and electrons that reduce NAD+ to NADH and H+.
- This reduced coenzyme NADH will be important in the energy-producing reactions we will discuss in electron transport
- Reaction 4: Oxidation and Decarboxylation
- 𝜶-ketoglutarate undergoes oxidation and decarboxylation to produce a four-carbon group that combines with CoA to form succinyl-CoA
- Reaction 5: Hydrolysis
- Succinyl-CoA undergoes hydrolysis to succinate and CoA. The energy released is used to add a phosphate group to GDP which yields GTP.
- Reaction 6: Oxidation
- Hydrogen is removed from each of two carbon atoms in succinate, which produces fumarate, a compound with a trans double bond.
- Reaction 7: Hydration
- Hydration adds water to the double bond of fumarate to yield malate, which is a secondary alcohol.
- Reaction 8: Oxidation
- The last step of the citric acid cycle, the secondary alcohol group in malate is oxidized to oxaloacetate, which has a ketone group.
18.6: Electron Transport and Oxidative Phosphorylation
- In electron transport, hydrogen ions and electrons from NADH and FADH2 are passed from one electron carrier to the next until they combine with oxygen to form H2O.
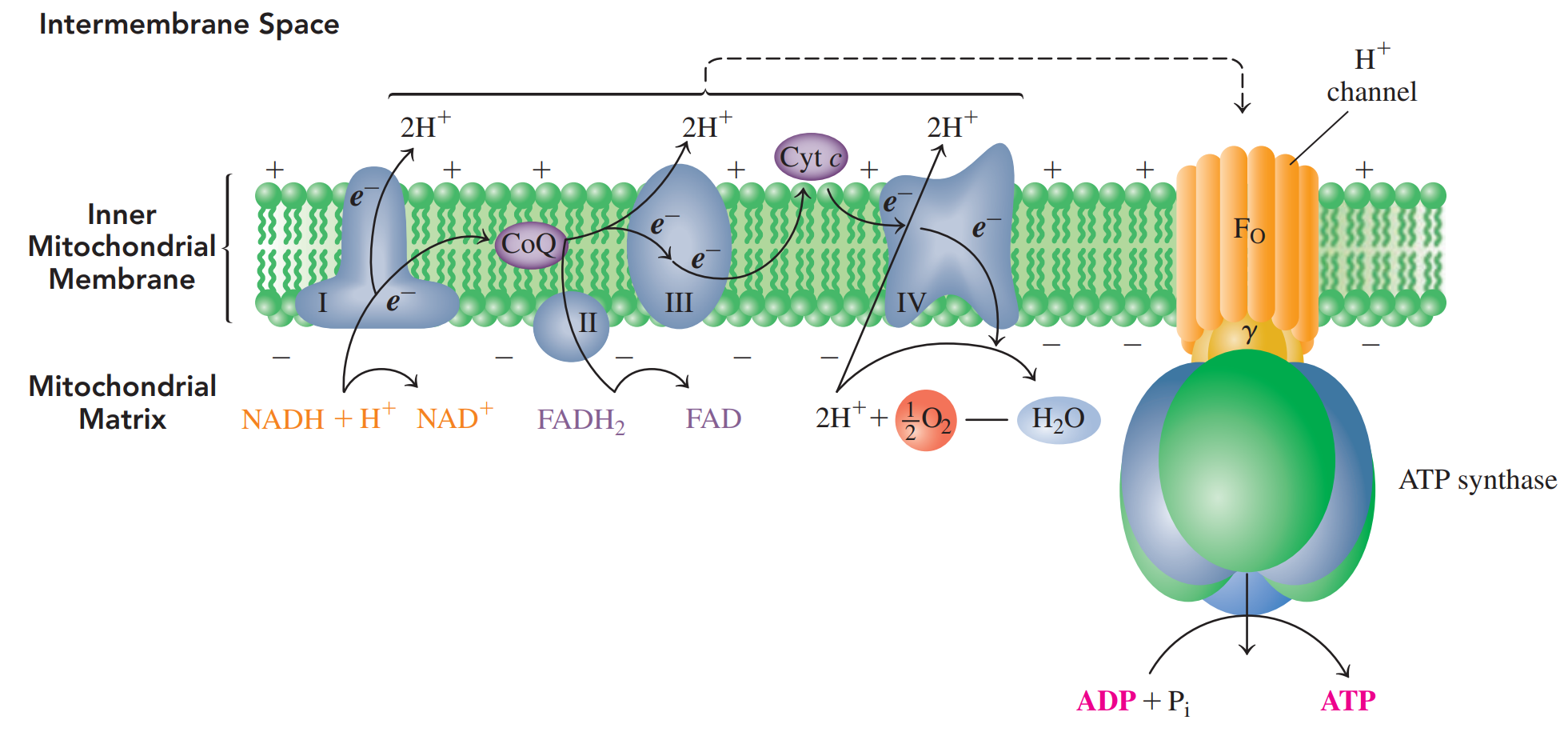
- Oxidative phosphorylation: The energy released during electron transport is used to synthesize ATP from ADP and Pi.
- Chemiosmotic model: Links the energy from electron transport to a H+ gradient that drives the synthesis of ATP.
- ATP Synthesis: An enzyme complex that uses the energy released by H+ ions returning to the matrix to synthesize ATP from ADP and Pi .
- ATP from Glycolysis
- In glycolysis, the oxidation of glucose stores energy in two NADH molecules as well as two ATP from direct phosphate transfer.
- However, glycolysis occurs in the cytoplasm, and the NADH produced cannot pass through the mitochondrial membrane.
- ATP from the Oxidation of Two Pyruvate
- Under aerobic conditions, pyruvate enters the mitochondria, where it is oxidized to give acetyl-CoA, CO2, and NADH.
- Because glucose yields two pyruvates, two NADH enter electron transport, where the oxidation of two pyruvate leads to the production of six ATP.
- ATP from the Citric Acid Cycle: One turn of the citric acid cycle produces two CO2, three NADH, one FADH2, and one ATP by direct phosphate transfer.
- ATP from the Complete Oxidation of Glucose: The total ATP for the complete oxidation of glucose is calculated by combining the ATP produced from glycolysis, the oxidation of pyruvate, and the citric acid cycle.
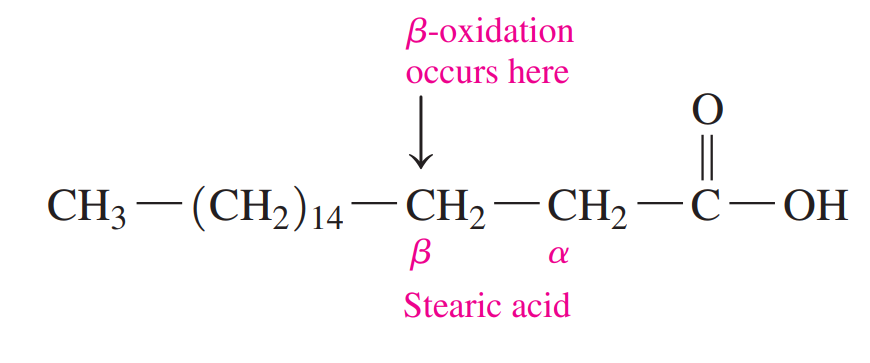
18.7: Oxidation of Fatty Acids
- A large amount of energy is obtained when fatty acids undergo oxidation in the mitochondria to yield acetyl-CoA.
- Beta-oxidation: This is where fatty acids undergo the removal of two-carbon segments, one at a time, from the carboxyl end.
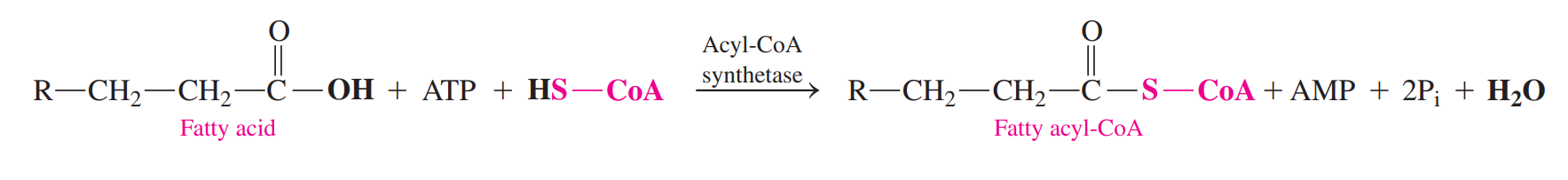
- Fatty Acid Activation: It combines fatty acid with coenzyme A to yield fatty acyl-CoA.
- The energy for the activation is obtained from the hydrolysis of ATP to give AMP and two inorganic phosphates.
- Ketone Bodies: The products of ketogenesis: are acetoacetate, 𝜷-hydroxybutyrate, and acetone.
- Ketosis: A condition of the accumulation of ketone bodies; which occurs in severe diabetes, diets high in fat and low in carbohydrates, alcoholism, and starvation.
18.8: Degradation of Amino Acids
- Transamination
- An 𝜶-amino group is transferred from an amino acid to an a-keto acid, usually a-ketoglutarate.
- A new amino acid and a new 𝜶-keto acid.
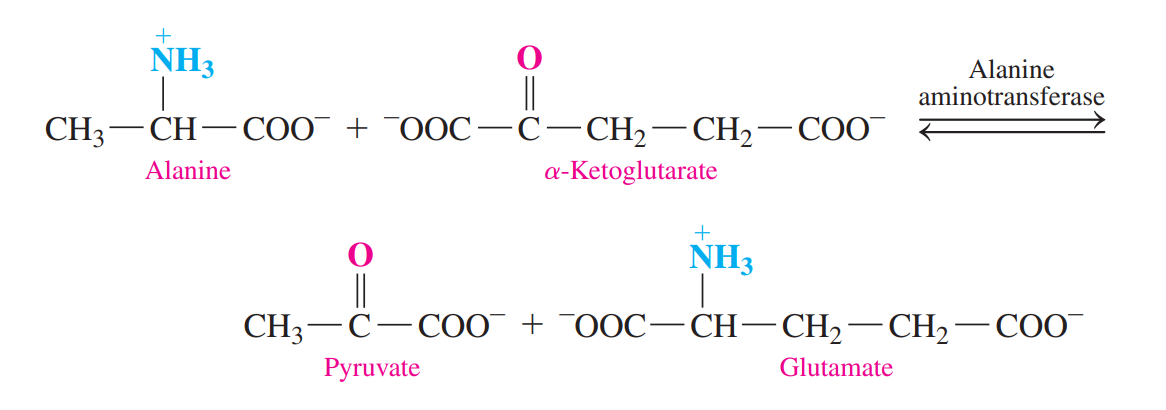
- Oxidative Deamination: The ammonium group in glutamate is removed as an ammonium ion.
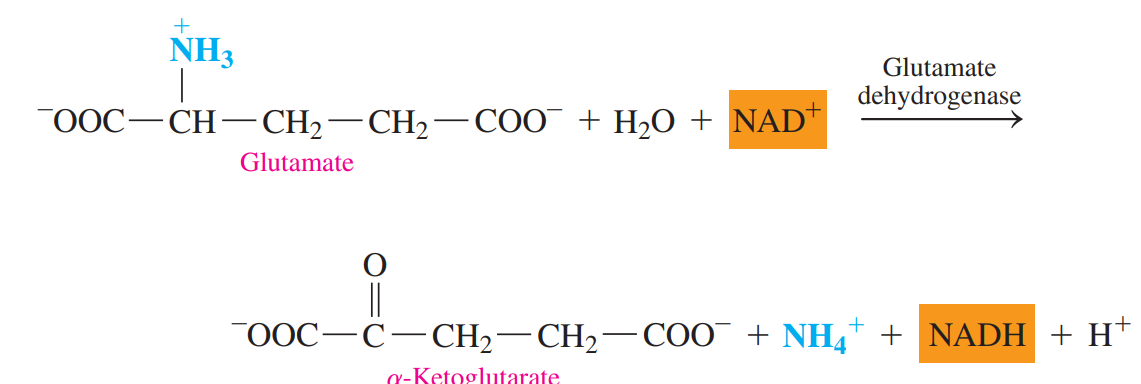
- Urea Cycle: A series of reactions that detoxifies ammonium ions by forming urea.
- The ammonium ion, which is the end product of amino acid degradation, is toxic if it is allowed to accumulate.