3.1 REDOX AND STANDARD ELECTRODE POTENTIAL
1/36
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
37 Terms
Role of platinum electrode
Inert electrode carry the charge
How to measure volume of gas produced
Gas syringe measure volume of gas produced at specified times
Which way do electrons flow in a cell
The more positive of system is the oxidising agent
Accepting electrons via external circuit
Vanadium ions and colours
VO2 + (+5) yellow
VO 2+ (+4) blue
V 3+ (+3) green
V 2+ (+2) purple
Young boys grow powerful
Anode and cathode identifying
Anode - Oxidised (more negative electrode potential)
Cathode - Reduction (more positive)
Standard cell potential formula
Ecell∘ =Ecathode∘ −Eanode∘
Postitve - more negative
Oxidising agent
Accepts electrons from substance ( reduced )
Reducing agent
Donates electrons (oxidised)
Half-equation definition
Shows oxidation or reduction tracks electron transfer
Half cells
Metal or electrolyte combination(oxidation or reduction)
Standard electrode potential definition
“The standard electrode potential (E°) is the e.m.f. of a half-cell compared with the Standard Hydrogen Electrode under standard conditions.”
298 K (25 °C)
Solutions at 1 mol dm⁻³
Gases at 100 kPa pressure
Electrode potentials?
Oxidising agent - more positive
Reducing more negative
Standard hydrogen electrode
E= 0 provide a baseline potential
Set up - Platinum electrode (coated platinum black increase surface area)
When is platinum electrode used in cells
No solid metal present to act as the electrode
Inert, does not react, conducts electrons and provide surface for redox reaction
Pt electrode purpose
Provide electrical connection when no solid metal present
Why is platinum used as electrode
Chemical inert
Good conductor of electricity
Provide surface for electron transferr
When do you not need a platinum electrode
When solid metal present that can act as electrode
Only use when ions or gases in soloution
Standard conditions
Concentration 1moldm-3
Temperature 298k
Pressure (gas) 1atm
Manganate reaction
MnO4 - + 8H+ + 5e- → Mn2+ + 4H2O
Cell EMF (electromotive force) definition
Potential difference (voltage ) between 2 half cells under Standard conditions
Represent driving force for electron flow in cell
Calculating cell EMF
Ecell = E reduction(cathode) - E oxidation (anode)
Significance of cell EMF (feasibility)
The sign of Ecell determines whether a reaction is feasible
Positive Ecell
Reaction is feasible
Electrons will flow naturally
Cell produce electrical energy
Negative Ecell
Not feasible
Only occur if external energy is supplied
Zero
System at equilibrium
Why does EMF indicate feasibility
Electrons flow from
More negative electrode potential →positive electrode potential
Reducing agent → Oxidising agent
Reaction is feasible if
The oxidising agent has a higher E than the reducing agent
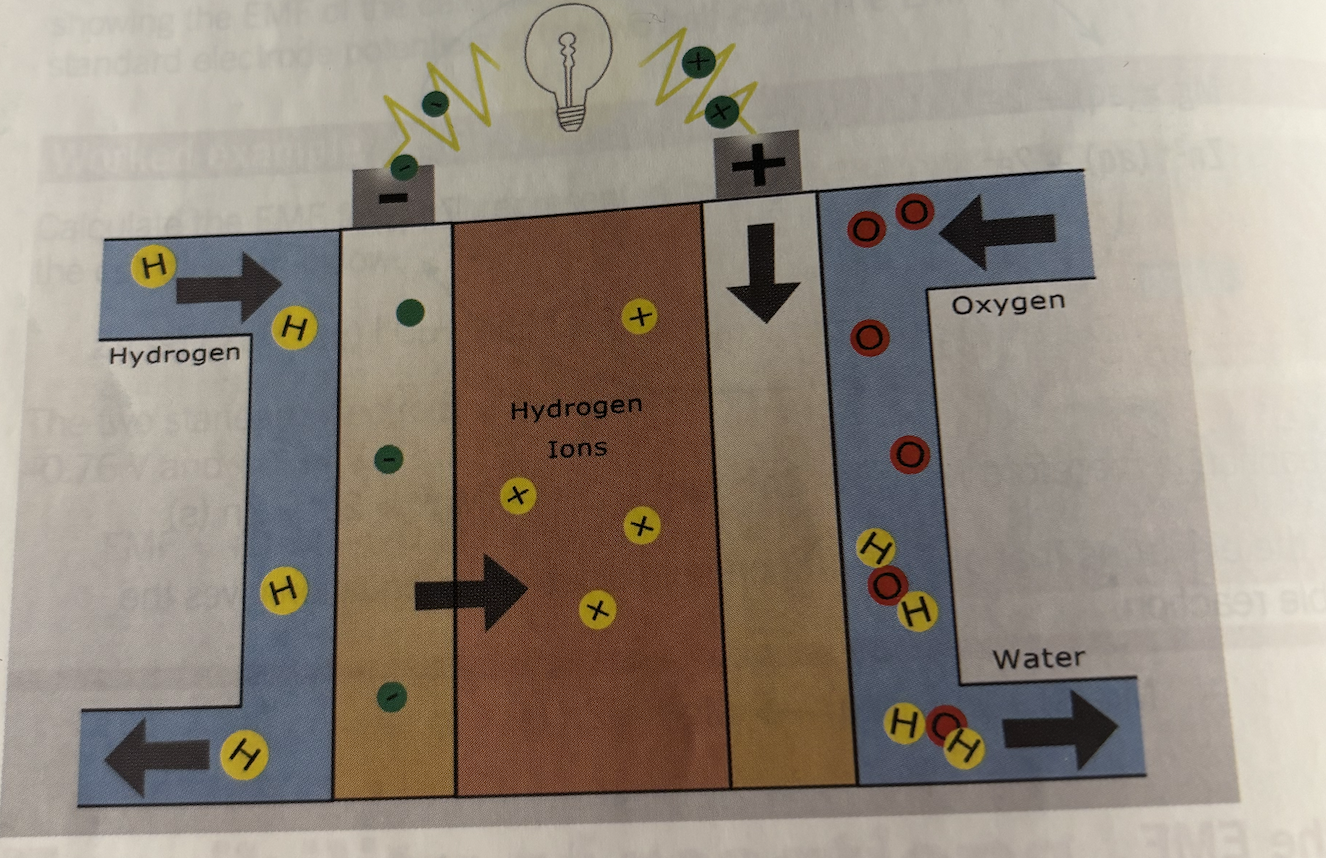
Hydrogen fuel cells
A hydrogen fuel cell converts chemical energy from hydrogen and oxygen into electrical energy using redox reactions
Reactants continuously supplied
Products continuously removed
2H2 + O2 → 2H2O
Half reactions in hydrogen fuel cells
Anode (oxidation)
Hydrogen is oxidised
H2→ 2H+ + 2e-
Hydrogen lose electrons produce protons
Cathode (reduction)
Hydrogen is reduced
O2 +4H+ +4e- → 2H20
Standard electrode potential definition
EMF measured on high resistance voltmeter when half cell containing-
1moldm-3 conc and all gases 1atm 298K
Standard hydrogen electrode definition
Inert platinum electrode
1moldm-3 Soloution of H+
Hydrogen gass pressure 1atm bubbled over temp 298k
Structure of hydrogen fuel cell
Anode (hydrogen electrode)
Cathode (oxygen electrode)
Electrolyte membrane
Catalyst Platinum
Functions
Electrodes allow electron transfer
Electrolyte allow ion movement
External circuit
Benefits of hydrogen fuell cells
Convenient store and release energy
High energy efficiency convert chemical → electrical energy avoid energy loss via combustion
Continuous operations - operate as long as fuel is supplied
Water product - no Co2
Disadvantages of fuel cells
Hydrogen production (not a primary energy source)
Electrolysis of water os steam reform methane (produces CO2)
Hydrogen gas hard to store (low density high pressure low temp)
Catalyst cost platinum rare and expensive
Ammeter
Measure current (flow of electrons) A
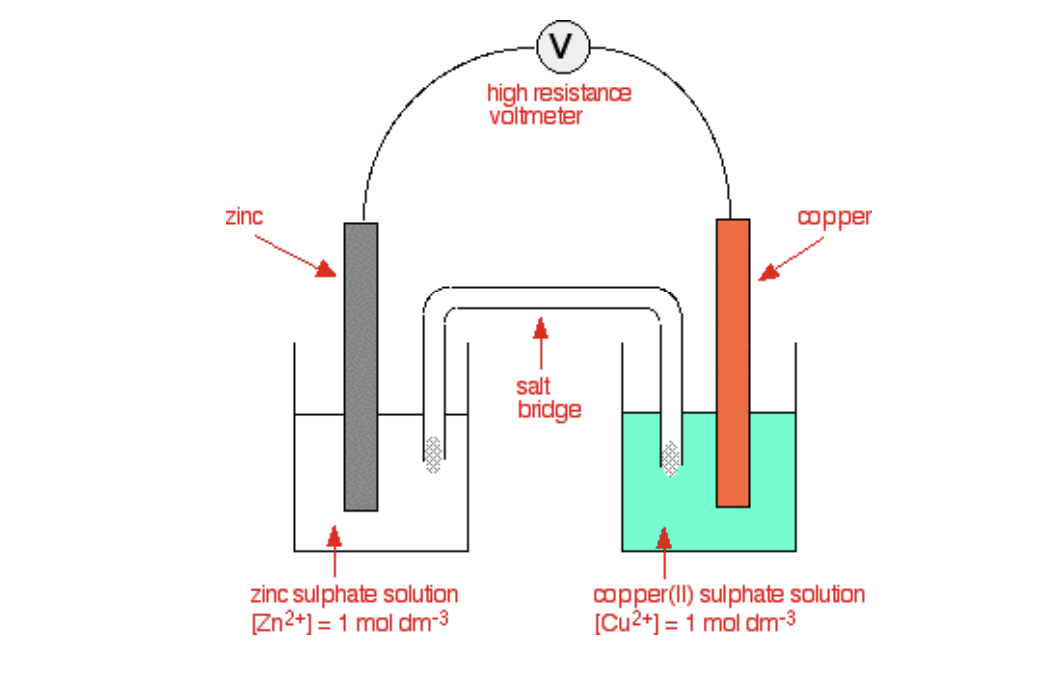
Half cells structure
1moldm-3 of soloutions
Electrode
Slat bridge
Wire
High resistance voltmeter
Wire and high resistance voltmeter function in half cells
Allow electrons flow
High resistance voltmeter measure potential difference
Salt bridge function
Complete circuit ion flow without mixing
Gel soaked KNO3
Corrosion
Oxidation occur
So electrode potential of metal must be lower than acid for metal to be oxidised
Half cells method of Cu2+ and Zn2+
Measure 50cm of Cu(SO4) and ZnSO4 in separate beakers
Place copper foil if CuSO4 soloution and zinc foil in ZnSO4 soloution
Connect zinc foil strip and Cu foil strip in ammeter
Connect voltmeter across foils
Place 2 beakers next to each other
Soak filter paper in saturated KNO3
Measure potential difference