ANS 10 - drug design p3 - pharmocokinetics
1/13
Earn XP
Description and Tags
improving pharmacokinetics,properties of agonist ,antagonists
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Why is altering pharmacokinetics important?
• Reduce dosing levels
• Improved duration of action
• Better mode of application
What is lipinski’s rule of 5 ?
The ideal characteristics of a drug for oral delivery
If you want it to be orally bioavailable :
• Molecular weight ≤ 500
• log P ≤5 - partition coefficient
• ≤10 H - bond accepting groups
• ≤5 H - bond donating groups
What combination of molecules are acetylcholine receptors made up from?
nicotinic, muscarinic
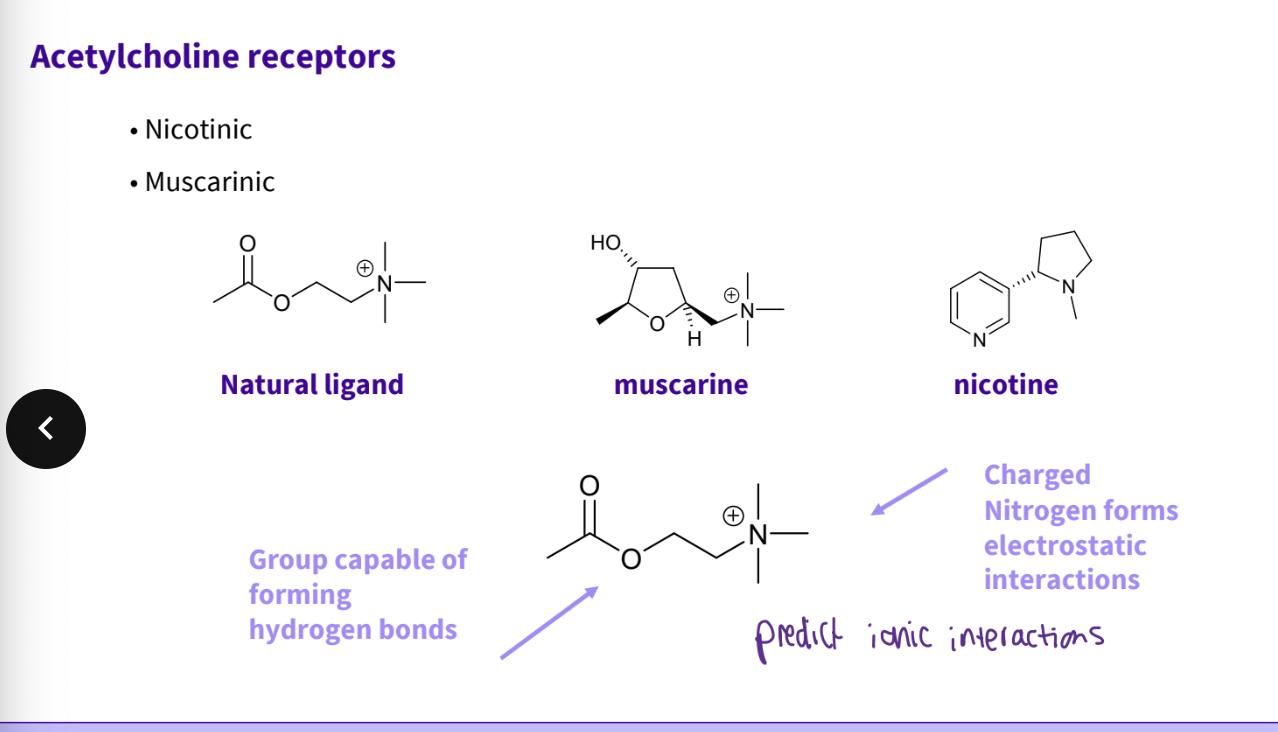
What are some acetylcholine receptors properties /structures ?
group capable of forming hydrogen bonds
charged nitrogen forms electrostatic interactions
ester site
anionic site
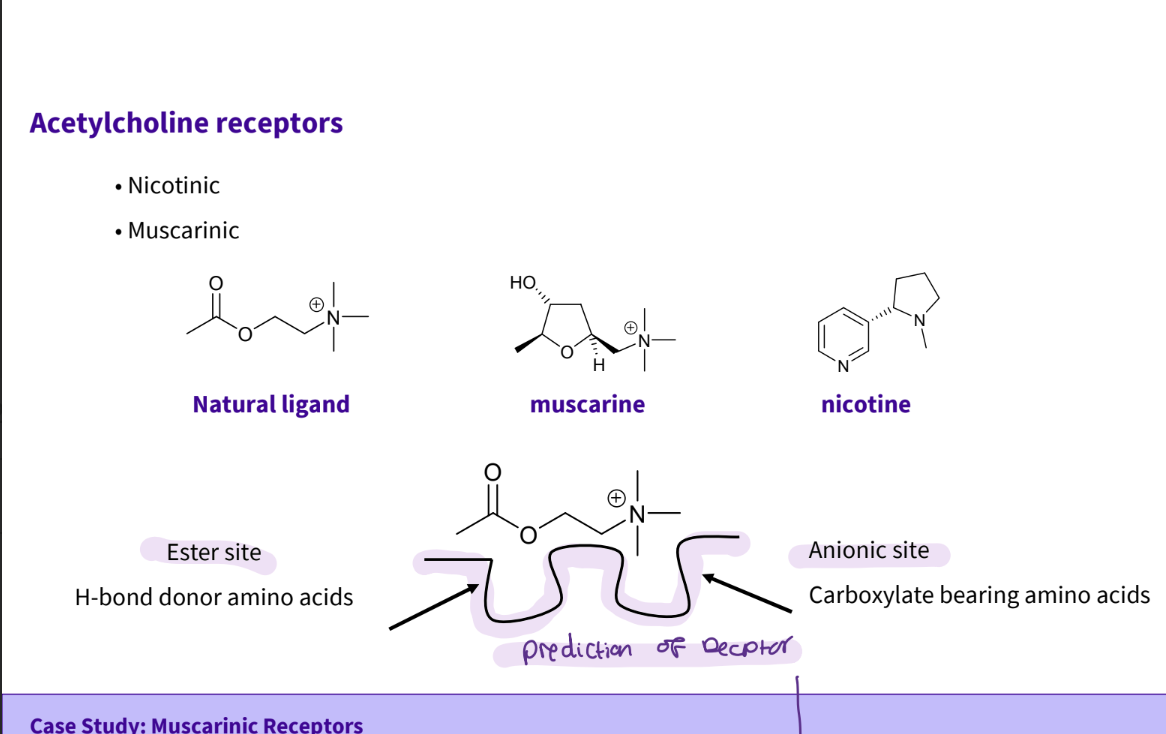
What factors do you need to consider for the receptor interaction site?
Distance between the two interacting sites
Substitution pattern on nitrogen
Substitution at ester
Chirality (acetylcholine is not but.....)
Why does acetylcholine not have ideal drug characteristics?
• Quaternary ammonium group makes it difficult for the molecule to pass
across the cell membranes
• Ester group is easily hydrolysed by acetylcholine esterase
• Not specific – interacts with all muscarinic and nicotinic receptors
What are the key steps/rules to determine agonist activity in a molecule?
Note - need to be similar to acetylcholine
need to have quaternary ammonium - with only one group no bigger than an ethyl
2. Look at the ings rule the distance between the atoms - no more than 5 atoms between the charge and the h atom
3. Does it have an ester /ether - at the right position - 2 carbons away - substituent capable of hydrogen bonding
4. Does it have chirality - if so it needs to be s
What doe sthe length affect ?
increasing the length between the charge and The last atom decreases activity
increasing the length between the charge and the last atom decrease s activity
Why is chirality important?
if on the b- carbon increasing potency , increasing selectivity for muscarinic over nicotinic,reduces ester hydrolysis
if on the a- carbon - decrease potency ,increases selectivity for nicotinic
s for - is equipotent with acetylcholine,more active
summary - agonist properties
1. Quaternary ammonium group ESSENTIAL for activity
2. Substitution pattern at nitrogen is CRUCIAL
3. Ings Rule of five
4. Substitution larger than methyl between nitrogen and oxygen reduces activity
5. A substituent capable of HYDROGEN BONDING is ESSENTIAL for binding
What are the properties required for antagonist behaviour ?
tropane scaffold
tertiary nitrogen not charged
etehr for h- bonf formation
ings rule of 5
Describe the structural properties of an antagonist
Ideally, nitrogen should be quaternary
Non quaternary are believed to be protonated by receptor before binding
Changing substituents to ethyl, isopropyl, etc enables additional positive interactions
Complex skeleton around nitrogen is not required –
simple alkyl OK
What makes the best antagonists?
H should be replaced by a carbo or heterocyclic unit
The best antagonists have one aromatic and one saturated cycle
Aromatics should not be too large e.g. naphthalene
Cycles can be fused to give a tricyclic
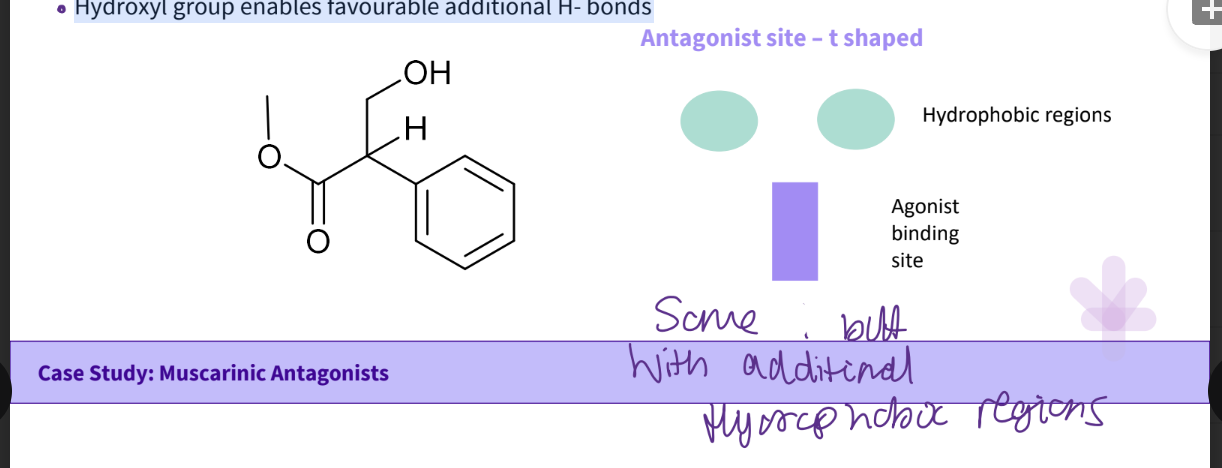
The hydroxyl group enables favourable additional H- bonds
What is a binding theory diagram ?
the squares and circles