CHE 2B CH 16- FINAL
1/95
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
96 Terms
Viscosity
Resistance to flow (due to cohesive forces) (also used to describe how thick/thin a liquid is). Think oil, honey, syrup- they all have higher viscosity, bc they don’t want to move around
solubility vs concentration
Solubility refers to the maximum amount of solute that can dissolve in a solvent at a given temperature, while concentration measures how much solute is present in a solution relative to the volume of solvent.

how can you have LDFs, even on non polar molecules, where it theoretically shouldn’t be possible for them to have an intananous dipole?

Network covalent solids
A type of solid where atoms are bonded by a continuous network of covalent bonds, resulting in very hard materials like diamond and silica.
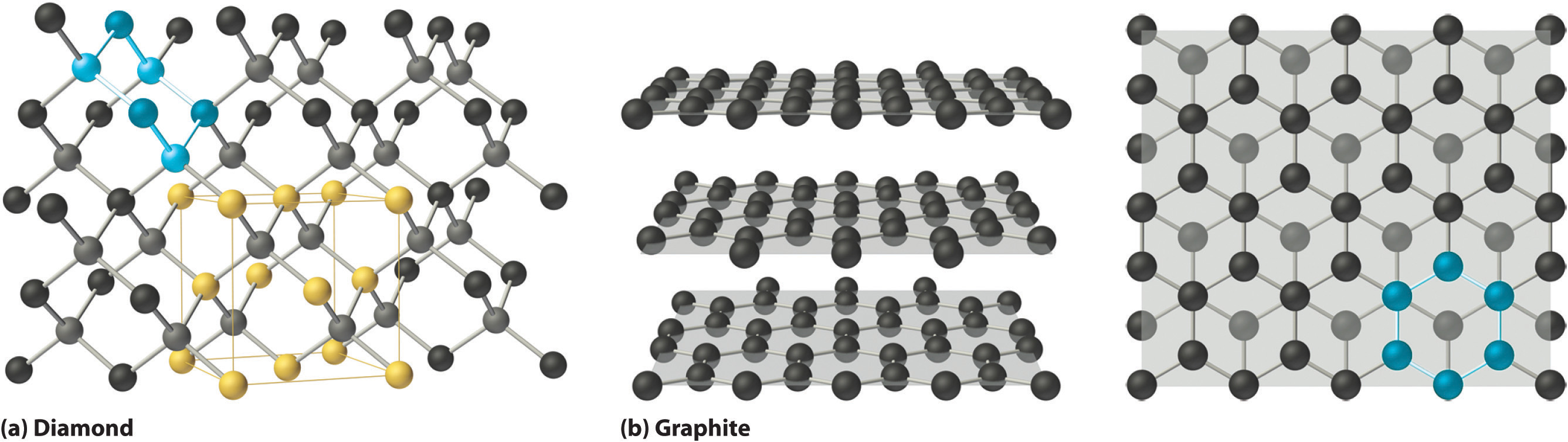
what are the 6 different types of Classification of Crystalline Solids, and what do their compositions and forces look like?
View table (the descriptions and examples are just bonus info)
crystalline solid
A solid whose atoms are arranged in an orderly, repeating pattern, resulting in distinct geometric shapes and defined melting points.
cohesive forces
forces between LIKE molecules that cause them to attract each other.
Surface tension- what is it, what is it defined as

relationship between surface tension and temperature, and what that means of KE and IMFs
Surface tension decreases with increasing temperature because higher temperature means higher kinetic energy. Higher kinetic energy results in more movement of molecules, which decreases the potential for attractive forces between molecules. With lower attractive forces, the surface tension will decrease.
what are some examples of surface tension irl- cleaning, material used for tents

Capillary action and what are some examples
the ability of a liquid to flow in narrow spaces without the assistance of external forces, due to adhesive forces between the liquid’s surfaces.
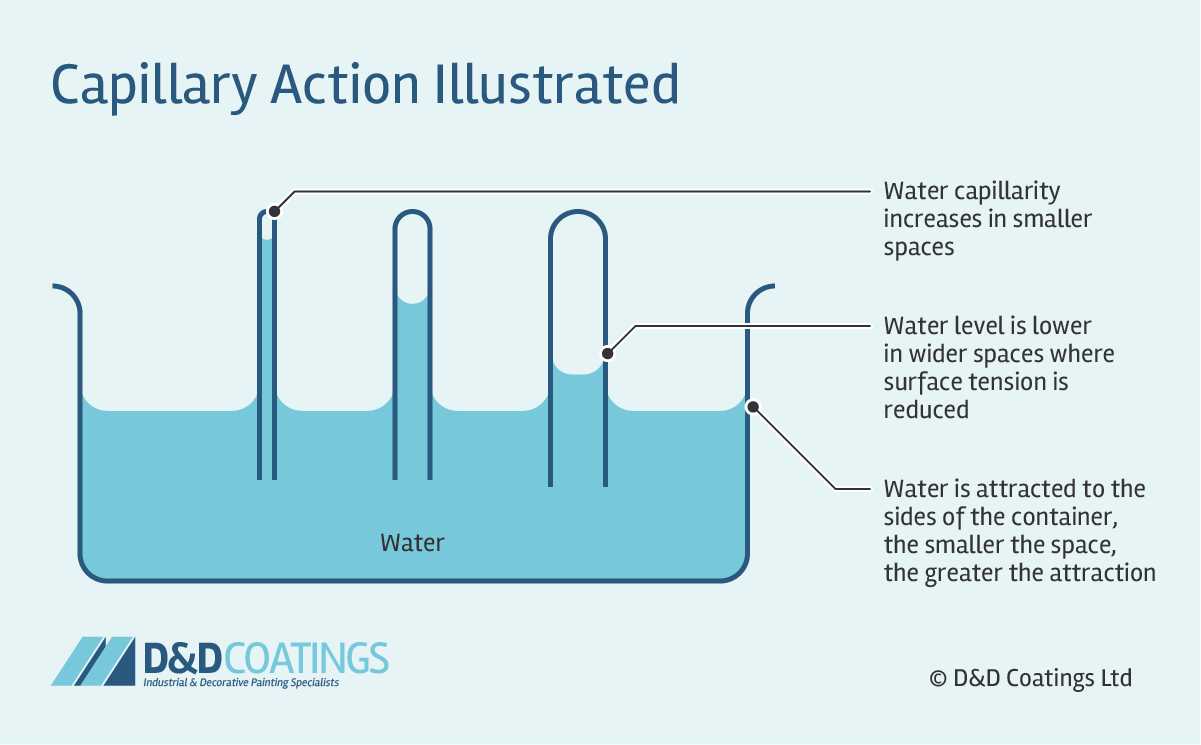
adhesive force
the attraction between UNLIKE molecules that causes them to stick to different surfaces.
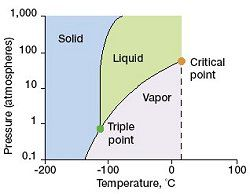
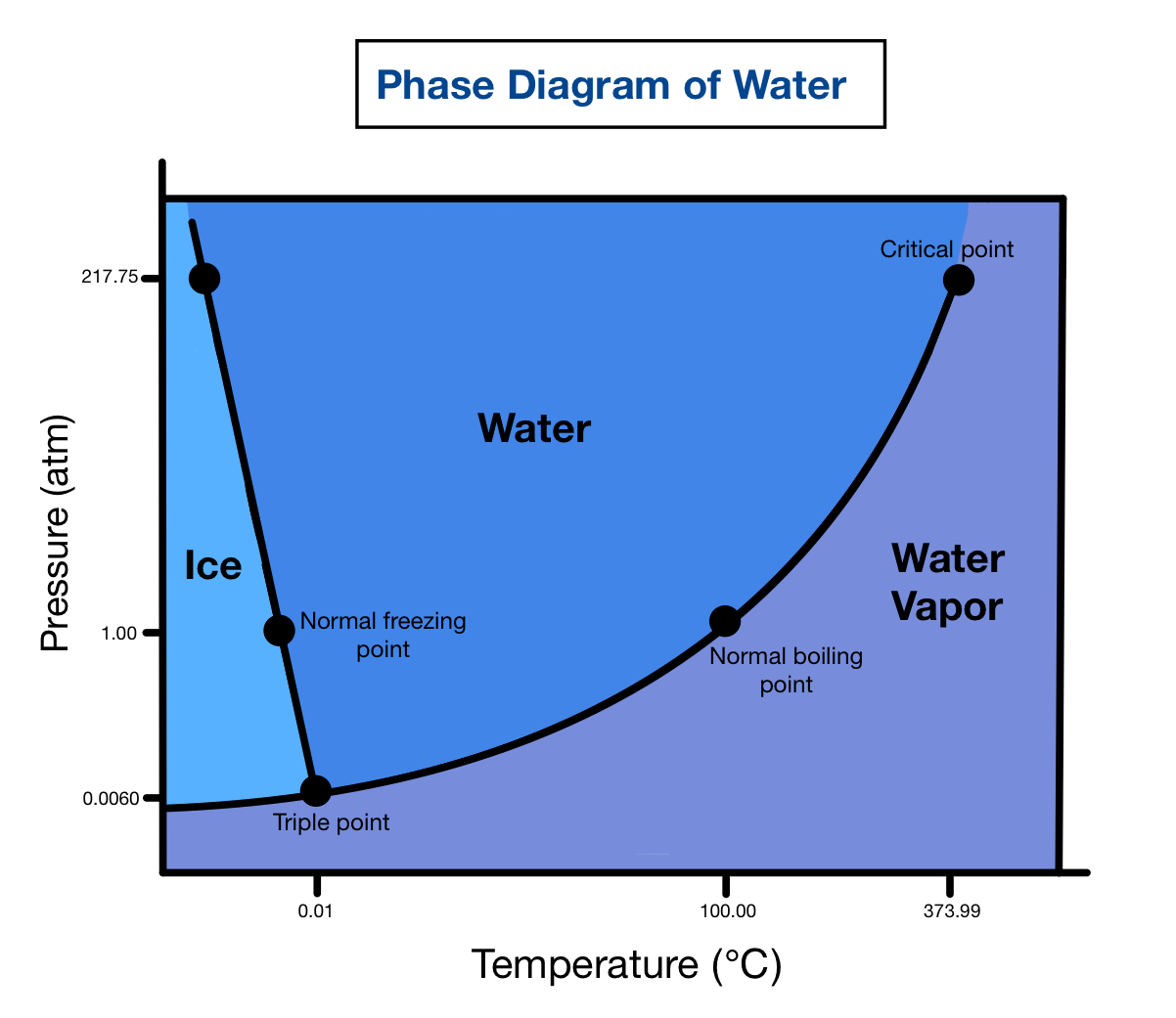
what is the critical point on a phase diagram
The temperature and pressure at which the liquid and gas phases of a substance becomes indistinguishable from one another
what is the triple point on a phase diagram
The unique set of conditions at which all three phases of a substance (solid, liquid, and gas) coexist in equilibrium.
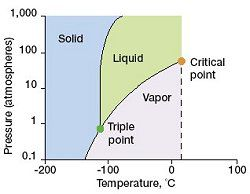
Isotherm
constant T (but P can change, vertical line on graph)
Isobar
Constant P (but T can change, horizontal line on graph)
ideal gas law vs Clausius- what info are you looking for in each one?
Ideal gas law: 1 compound is given, P will be vapor pressure of the liquid, you might also use n=m/M
Clausius-Clapeyron: 1 compound given, along with 2 states (P and T for both), maybe ΔH for the compound. PLUG AND CHUG
best way to compute a C-C equation problem
1.) Solve the right side
2.) put both sides as the power of e (to cancel out the ln)
3.) Solve entirely
common properties of solids
Solids maintain definite shape and definite volume.
• Solids can be crystalline or non-crystalline.
• Solids do not effectively compress or expand.
• The density of a solid is greater than that of a liquid or gas.
• Solids do not mix.
common properties of liquids
Liquids have an indefinite shape but a fixed volume.
• Liquids flow readily depending on their viscosity.
• Liquids do not readily undergo compression or expansion.
• The density of a liquid is greater than that of a gas.
• Soluble liquids mix uniformly.
Mass percent equation
mass %= [(ml - mg)/ml] × 100
energy of evaporation equation
endothermic (for example, when you have water on your skin, you feel colder bc the water is taking energy from your body to evaporate!)
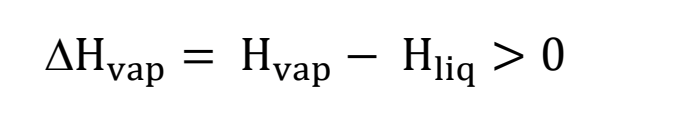
energy of condensation equation
(CAN ALSO BE ΔHvap = -ΔHcondensation) exothermic (explains steam burn- when it condenses onto your skin, the energy goes into you)
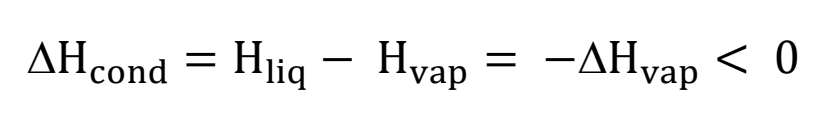
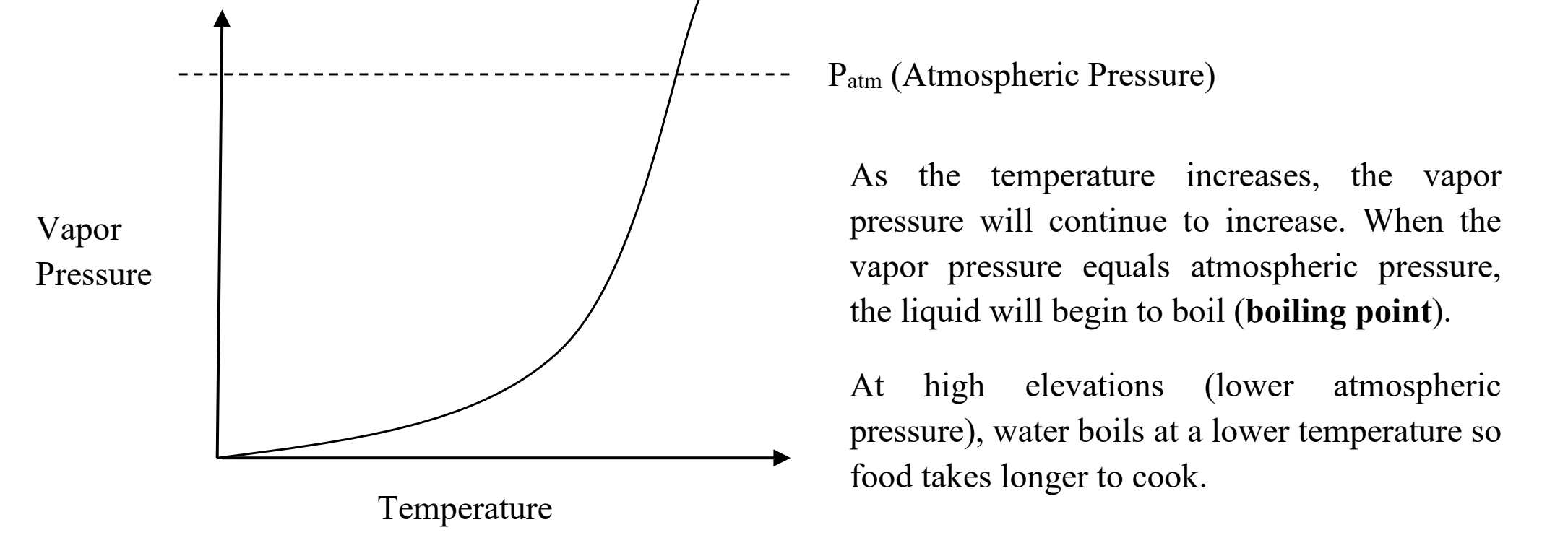
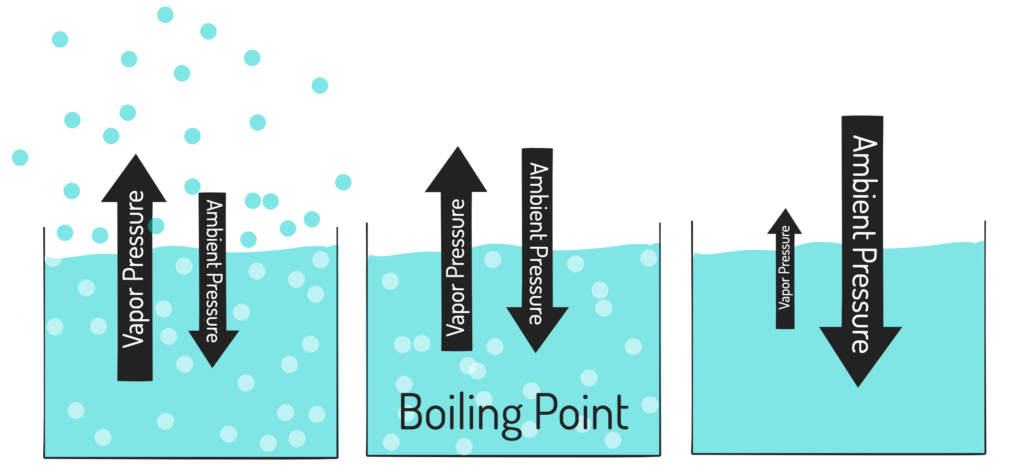
when the Pvapor = Patm =
BOILING POINT
sublimation
solid → gas
deposition
gas → solid
general equation for phase change ΔH:
(b/c u have the same start/end point)
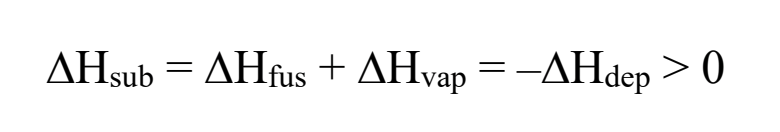
how is the enthalpy of melting referred to?
∆Hfus
(common exam question) if you are given a heating curve with all the q values of each phase change/∆T, how do you find the qtotal of the curve?
all the q’s together (q1+q2+q3+q4…)
why is ∆Hvap> ∆Hfus ?
To melt a substance, the molecules are heated to the point where the molecules can move freely, which reduces the forces of attraction. To boil a substance, the molecules are heated to a point where the forces of attraction approach zero (in an ideal sense). Thus, vaporization requires more energy to overcome the attractive forces.
supercooled liquid
A cooled liquid that has no place for crystals to form. (liquid below freezing point, but no freezing)
superheated liquid
state where liquid is heated above its BP without turning into a gas. It is very unstable and can erupt into gas if disturbed.
Supercritical Fluid
substance at a P + T above its critical point, where there is no distiniction between liquid and gas
Heating Curve vs. Phase Diagram
In contrast to a heating curve, a phase diagram is not at constant pressure. Phase diagrams depict phases at various pressures. If one takes a cross-section of a phase diagram along an isobar, it represent the heating curve. For example, consider the phase diagram of water (see Google images). Now, consider only the phases along the isobar of atmospheric pressure, or 1 atm. That isobar represents the heating curve of water between its solid, liquid, and gas phases.
does temperature change during a phase change?
NO!!!!!!!!
how would you calculate q during a phase change?
use q=nΔH
how would you calculate the value of q when there IS a ΔT?
q=mCpΔT
NORMAL boiling point vs boiling point
The normal boiling point is the temperature at which a substance boils at a pressure of 1 atmosphere, whereas the boiling point can vary based on atmospheric pressure.
what are IMFs
they are what 2 molecules in proximity exert on each other. it will affect the physical properties of a substance. Even some ‘nonreactive’ elements experience it
Dipole-Dipole forces
exists when a molecule has a permanent dipole (it’s just inherent within its structure). THIS HAPPENS when there’s an imbalance in e- density, and when there’s atoms w/ differing EN values in the molecule
London dispersion forces (LDF)
are weak intermolecular forces that arise from instantaneous dipoles in molecules. They occur in all molecules, regardless of polarity, and are generally stronger in larger, more polarizable atoms or molecules.
Relation between molar mass and LDF, and why it happens
as MM inc, LDF inc, because of polarizability (the ability for an atom/molecule to form instant dipoles)
hydrogen-bonds- what conditions does do they form?
1.) there’s 1 molecule w/ a highly EN atom
2.) there’s another molecule with an H atom directly bonded to an EN element (like N, O, or F)
IMF strength ranking, biggest strength to smallest
IONIC, H-BOND, DIPOLE, LONDON (tho it is the weakest, it inc as molecule inc in size)
how is VISCOSITY, SURFACE TENSION, BP, Pvap, ΔHvap affected when there are a lot of IMFs? (answer is either high or low)
viscosity- high (liquid will be thicker bc the molecules. are more stuck together)
surface tension- high (liquid will want to stick together more)
BP- high - (more bonds will need to be broken, requiring more energy)
Pvap- low (bc more energy will be needed for evaporating the liquid, it will be stubborn and not want to vaporize, leading to less vapor in the air)
ΔHvap - high (bc more energy will be needed to evaporate, it’ll be very endothermic)
intermolecular vs intramolecular
Intermolecular- attractions between molecules
intramolecular- within 1 particular molecule- like a bond!
what is the equation for MOLARITY (M)
amount of solute (moles) / volume of solution (liters)
What are the 3 different types of ways to express concentration w/ percents?
a.) Mass percent= mass of solute/mass of solution x 100
b.) Volume percent= volume of solute/volume of solution x 100
c.) Mass Volume Percent= mass of solute (grams) / volume of solution (mL) x 100
What are parts per units usually used for?
They are used for very low concentrations
what are the 3 parts per units?
pink- what to use if it asks “ppx per weight”
blue- what to use if it asks “ppx per volume”

MOLE FRACTION (Xi) & PERCENT- what is it and what is the equation?
Mole fraction (Xi) is the ratio of the number of moles of a component to the total number of moles in a mixture.
xi= moles i /total moles
(and to get the percent, just multiply xi by 100)
Molality (m)- what’s the advantage of using this over molarity and what’s the equation for it?
Unlike molarity, molality isn’t affected by temperature (b/c with molarity, when the temp changes, that causes the volume to also change)
m= amount of solute (moles)/mass of solvent (kg)
Normality (N)= what is it and what is the equation
essentially, it’s the molarity multiplied by the number of equivalents (something that takes into account of the number of ions in solution per source). This unit is usually used with acids/bases, redox, and precipitation.
N= (# of equivalents) (molarity)
Solubility
solubility= grams of solute dissolved/ given mass or volume of solvent
in concentration problems, are masses additive?
YES, because mass in conserved, masses are additive in solution when no reaction occurs, so:
msoln = msolute + msolvent
in concentration problems, are volumes additive?
SOMETIMES- in general they are UNLESS there is a stated volume change or there is a density change. Therefore:
Vsoln = Vsolute + Vsolvent
(sometimes)
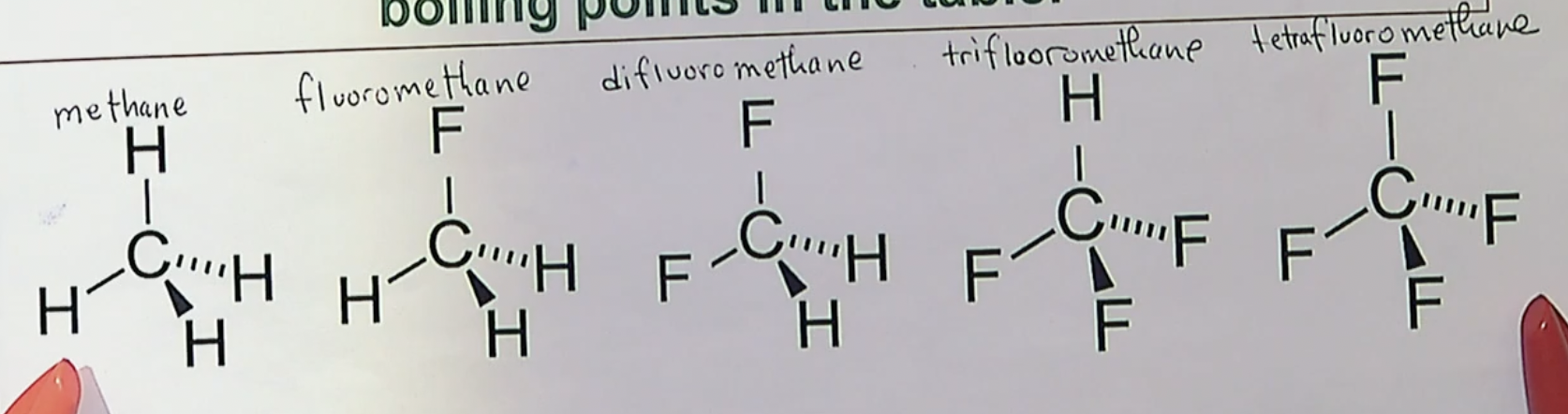
which one of these will have the strongest bond/highest dipole?
the one in the middle. The one on the left of it only has 1 F, so its dipole isn’t as big. The one of the right of it has 3 F’s, but having that extra F will slightly cancel out how strong the pull is on the other F’s, making the molecule with the 2 F have the highest dipole.
what does miscibility mean?
its the ability of 2 liquids to homogeneously mix in a solution
homogeneous
mixture of two or more substances that are evenly distributed throughout.

heterogenous
mixture of two or more substances that are not evenly distributed throughout.

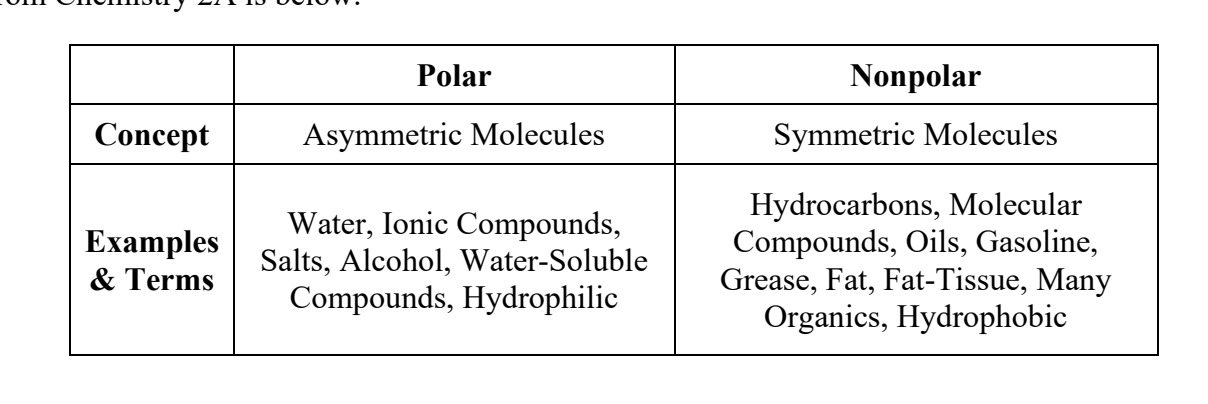
What does the term “like dissolves like”? (describe it using the word miscible)
“Like” refers to polarity. In other words, polar molecules will dissolve (or are miscible with) other polar molecules, and nonpolar molecules will dissolve (or are miscible with) other nonpolar molecules. This process occurs because the dipoles of polar molecules can interact with dipoles of other polar molecules. They are attracted like magnets.
What are some examples of polar and non-polar molecules?
In general, polar molecules are going to be water-based, and non-polar molecules are going to be organic, oil-based substances.
why should you drink milk after eating spicy food? Why doesn’t water really help?

why isn’t the organic compound DDT (non-polar) used to kill mosquitos anymore?

how can it be ok for something toxic to humans (Ba+2) (polar) to be safe to swallow?

why don’t spiders get stuck in their own webs?
what does the ∆Hsoln refer to?
since mixing a solute and solvent to form a solution takes energy, the ∆Hsoln is the ENTHALPY OF SOULTION
What would a ± value of ∆Hsoln mean?
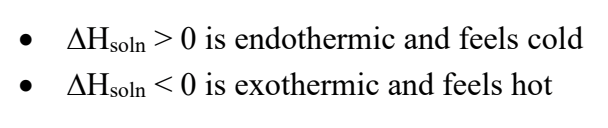
What are the 3 factors that contribute to ∆Hsoln? and because of that, what is ∆Hsoln equal to?
∆Hsoln = ∆Hsolvent (1) + ∆Hsolute(2) + ∆Hmixing(3)

What does it mean if ∆Hsoln=0?
the solution would be called an ideal solution. This can happen with some organic mixtures
when separating a solute/solvent of a nonpolar solution, will that take a small or large amount of energy, and why?
SMALL. np solutions have weaker bonds, because they have no dipoles, and only LDFs, the weakest bond. This means that it will take a small amount of energy to separate them.
when separating a solute/solvent of a polar solution, will that take a small or large amount of energy, and why?
LARGE. p solutions have stronger bonds, because the have dipoles, and are a lot more attracted to each other than np soln’s.
if you mix a polar and a np solution together, will they be attracted to each other?
hell no
if the ∆Hsoln= a large, positive number, what does that mean and will a solution form?
a positive number means making the solution is an ENDOthermic process, and since you will need a lot of energy to make the solution, it will not happen (unless heat/energy is added)
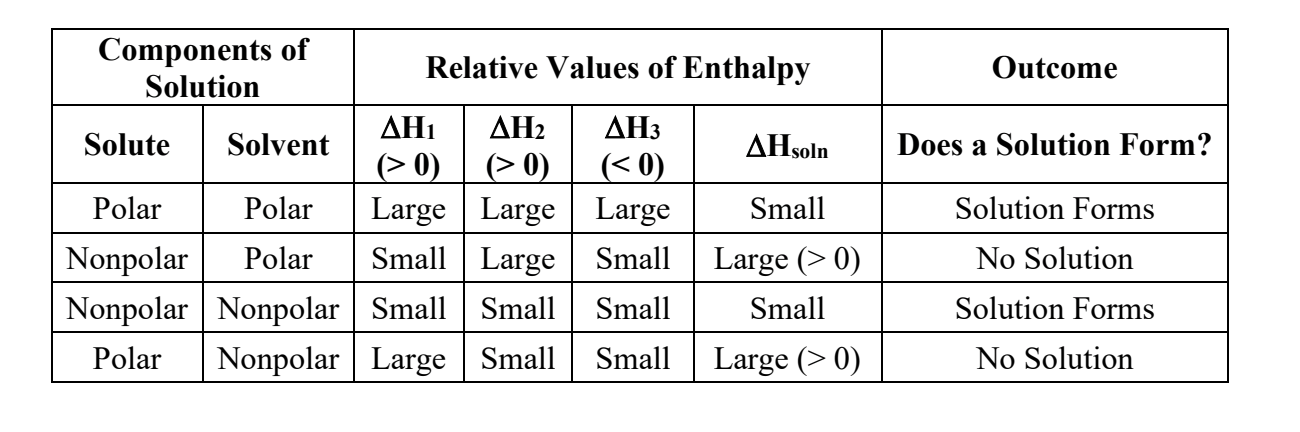
if the ∆Hsoln= a small, positive/negative number (ON IN GENERAL, WHEN IT’S CLOSE TO ZERO), what does that mean and will a solution form?
it would mean either the solution requires a little energy to form (in the case of a small +) (which is fine), OR forming the solution will release energy (with a - number AKA exothermic)
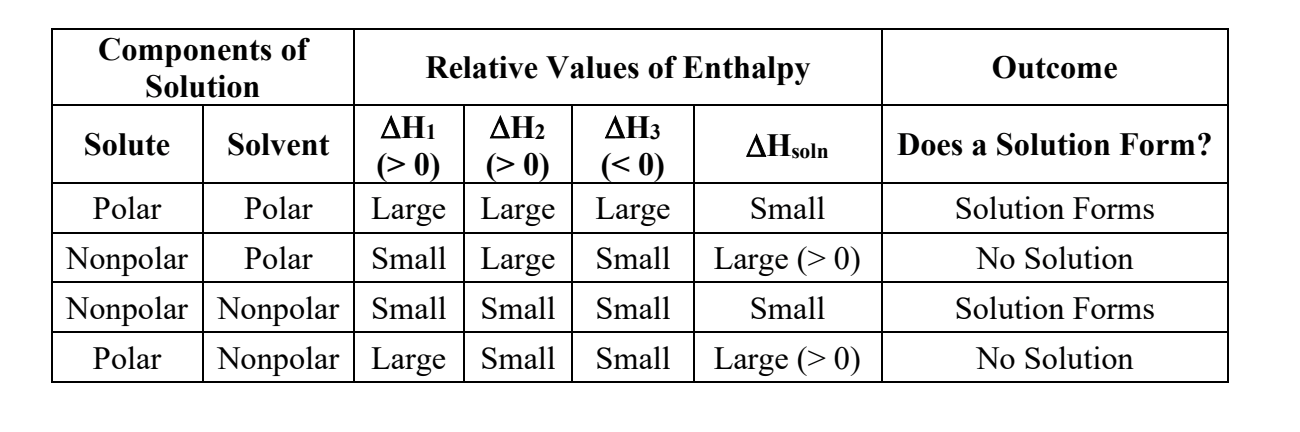

Which combos will form a solution?
1.) NO, becasue sol 1 is P, and sol 2 is NP.
2.) YES, bc sol 1 is P (ionic as hell) and sol 2 is P (bc of the dipole from OH)
3.) YES, bc sol is P (T-shaped!) and sol 2 is P (the Cl’s are NASTY and will steal the e'-s' from the H’s)
how would you tell what something’s vapor pressure will be like?
it can be determined by the strength of the IMFs (because the more the molecules stick together, the least likely they’ll want to evaporate).
so, if there are strong imf= the vapor pressure will be low
and if there are weak imf= Vp will be higher
Relationship between solubility and temperature
Solubility inc with temperature (AKA more solute will dissolve. This is why you can dissolve more sugar in hot coffee than iced coffee)
Exceptions to the relationship between solubility and temperature
Sulfates and phosphates
What does saturated mean
The saturated line represents the point where dissolution and crystallization are both occurring and in equilibrium with each other
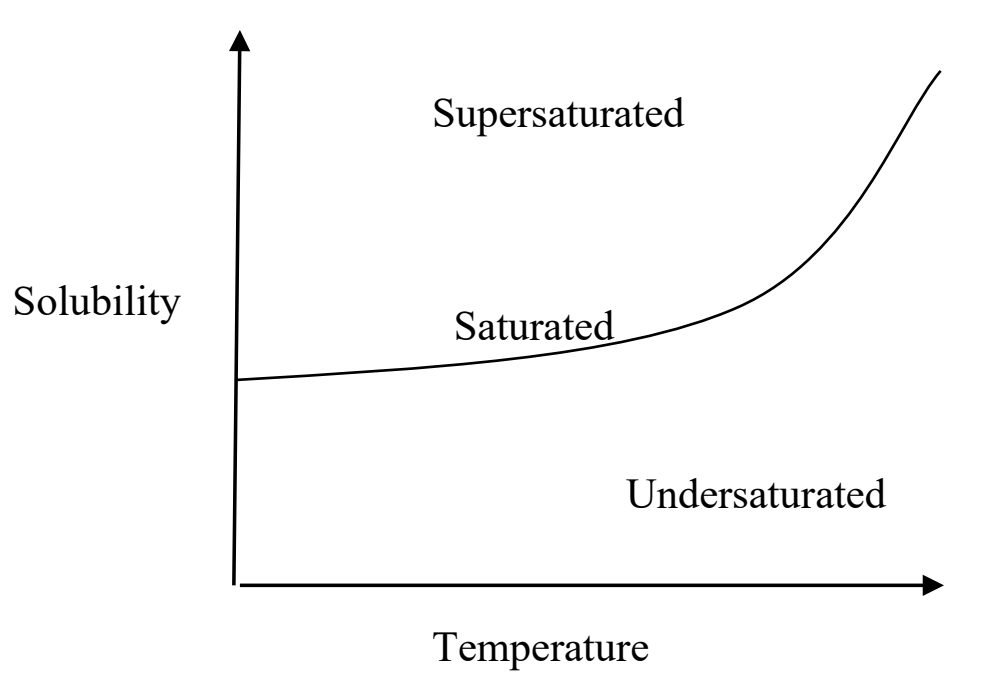
What does undersaturated mean
This region on the graph occurs when the solvent can dissolve more solute than is available.
(ex, u put less sugar in your coffee than it can potentially hold)
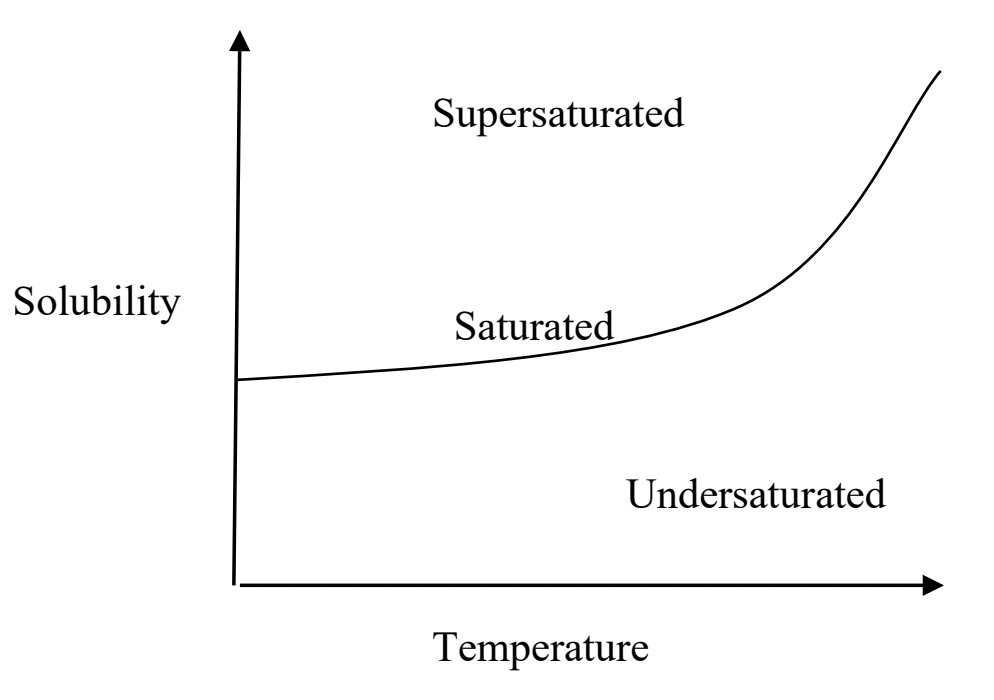
What does supersaturated mean
This region on the graph occurs when the solution has more solute in solutions than equilibrium allows. This scenario is unstable and any perturbation could cause the excess solute to crystallize.
(ex, u put more sugar if your coffee than it can hold, and it starts to collect on the bottom)
What are some examples of solubility/temperature- concerning washing dishing and showering

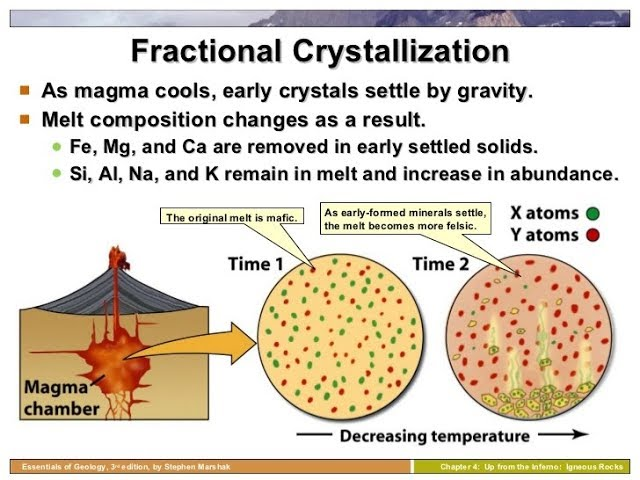
What is fractional crystallization?
Fractional crystallization- what is it?
it uses the concept of solubility to separate solutes. Consider an impure solid consisting of mostly A and some B. Dissolve this solid in a solvent at high temperature. Then cool the solution and each (A and B) will crystallize at different times making separation easier.
What are the 3 factors affecting solubility?
Structure, Temperature, Pressure
How does structure affect solubility?
It depends on whether a molecule is fat-soluble (np) and water-soluble (p), which will influence the interactions between the molecules
What is a fat-soluble compound, and what does it look like?
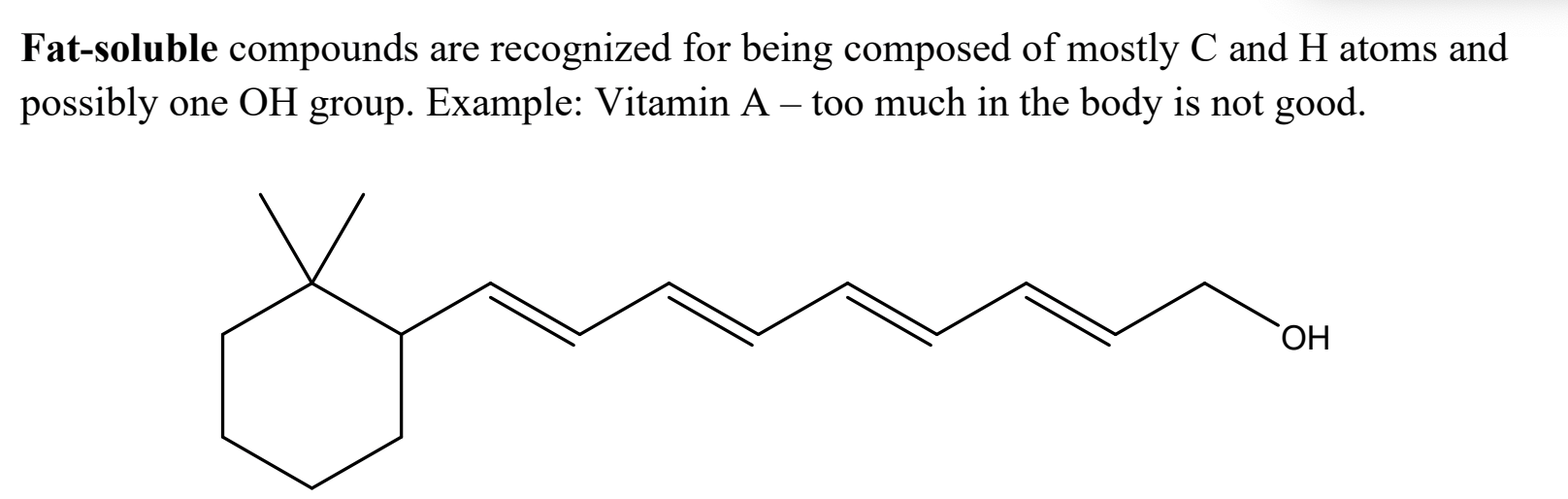
What is a water-soluble compound, and what does it look like?
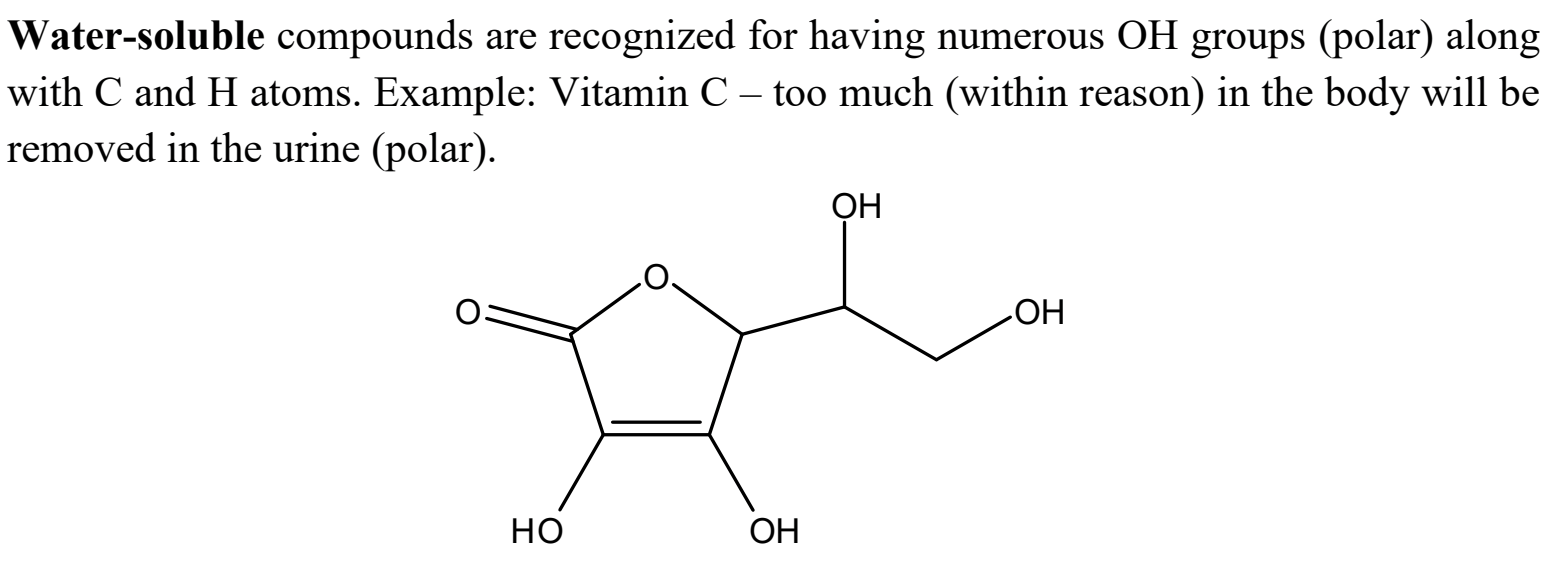
How does temperature affect solubility? Also, what is an example, specifically with soda?
If you open a warm soda, there will be much more fizz and a loss of carbonation as compared to a cold soda. Keeping soda cool is much preferred because it keeps the carbonation and the taste! (since the KE of the gas will be smaller with the lower temp, so they will be less likely to escape)

How does pressure affect solubility? What does this have to do with why soda is under higher pressure?
When the pressure inc, the solubility of a gas in a liquid inc.
Soda is under relatively high pressure bc it allows for more carbonation in the liquid (bc more CO2 will be dissolved)
Which equation describes pressure and solubility’s relationship?
HENRY’S LAW!!
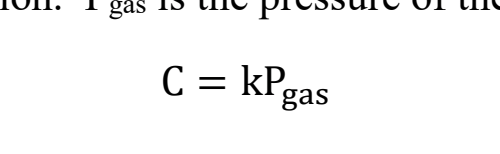
What is what in Henry’s Law?

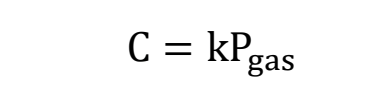
how can henry’s law be written if there is more than one pressure and concentration?
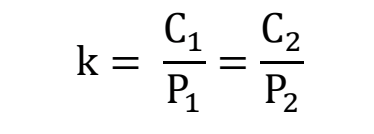
What is the relationship between pressure and altitude?

negligible
so small its unimportant