Chemistry - Intermolecular Forces and Properties
0.0(0)
Studied by 13 people0%Unit Mastery
0%Exam Mastery
Build your Mastery score
Supplemental Materials
Card Sorting
1/51
Earn XP
Description and Tags
Last updated 7:25 PM on 9/27/22
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
52 Terms
1
New cards
Condensed phase
Solid and liquid phases. Particles in a solid or liquid are fairly close together compared with those of a gas.
2
New cards
Gas
Particles far apart; possess complete freedom of motion.
3
New cards
Liquid
Particle are closely packed but randomly oriented; retain freedom of motion; rapidly change neighbors.
4
New cards
Solid
Particles are closely packed in an ordered array; positions are essentially fixed.
5
New cards
Intermolecular force
Attraction between molecules; generally not as strong as intramolecular forces.
6
New cards
Intramolecular force
Attraction between atoms within a molecule. (Ionic, metallic, or covalent bonds)
7
New cards
Dispersion force "London dispersion force"
The motions of electrons in one atom influence the motions of electrons in its neighbors. The instantaneous dipole on one atom can induce an instantaneous dipole on an adjacent atom, causing the atoms to be attracted to each other. The strength of the dispersion force depends on the polarizability of a molecule.
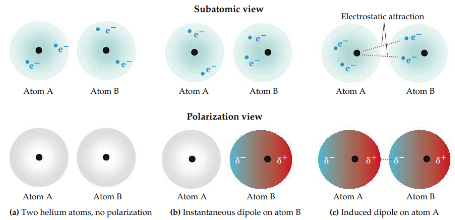
8
New cards
Polarizability
The molecule's ease with which the charge distribution is distorted to induce an instantaneous dipole. The greater the polarizability, the more easily the electron cloud can be distorted to give an instantaneous dipole. Therefore, more polarizable molecules have larger dispersion forces.
9
New cards
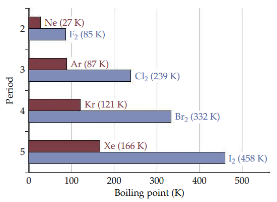
Molecular weight vs. Dispersion
The polarizability increases as the number of electrons in an atom or molecules increase. The strength of dispersion forces therefore tends to increase with increasing atomic or molecular size. Because molecular size and mass generally parallel each other, dispersion forces tend to increase in strength with increasing molecular weight.
10
New cards
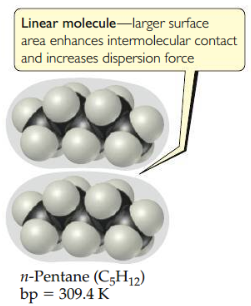
Molecular shape vs. Dispersion
Intermolecular attraction is greater for a linear molecule because the molecules can come in contact over the entire length of the long, somewhat cylindrical molecules. Less contact is possible between the more compact and nearly spherical molecules.
11
New cards
Linear molecule
Larger surface area enhances intermolecular contact and increases dispersion force.
12
New cards
Spherical molecule
Smaller surface area diminishes intermolecular contact and decreases dispersion force.

13
New cards
Instantaneous dipole
Temporary dipole that occurs for a brief moment in time when the electrons of an atom or molecule are distributed asymmetrically.
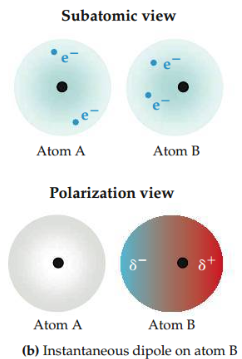
14
New cards
Induced dipole
A temporarily uneven distribution of electrons in an otherwise nonpolar atom or molecule.
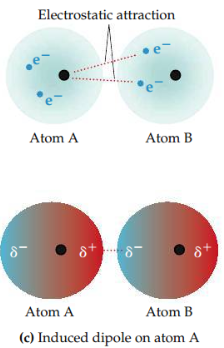
15
New cards
Permanent dipole
These occur when two atoms in a molecule have substantially different electronegativity.
16
New cards
Dipole-Dipole force
Result of a permanent dipole moment in polar molecules. Electrostatic attractions between the partially positive end of one molecule an d the partially negative end of a neighboring molecule. Repulsion can also occur when the positive (or negative) ends of two molecules are in close proximity.
17
New cards
Polarity vs Dipole-Dipole
For molecules of approximately equal mass and size, the strength of intermolecular attractions increases with increasing polarity. (Boiling point increases as the dipole moment increases)
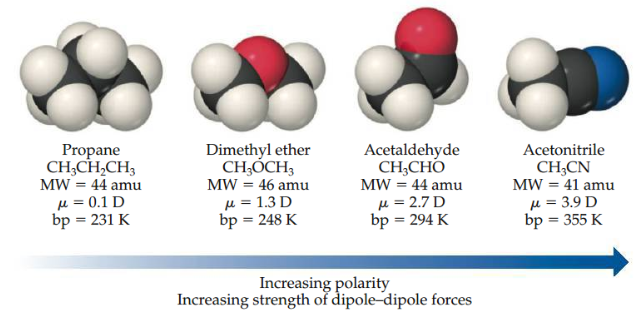
18
New cards
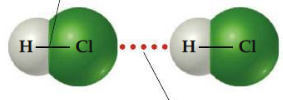
Hydrogen bond
Attraction between a hydrogen atom attached to a highly electronegative atom (usually F, O, or N) and a nearby small electronegative atom in another molecule or chemical group. Because the electron-poor hydrogen is so small, it can approach an electronegative atom very closely and, thus, interact strongly with it.
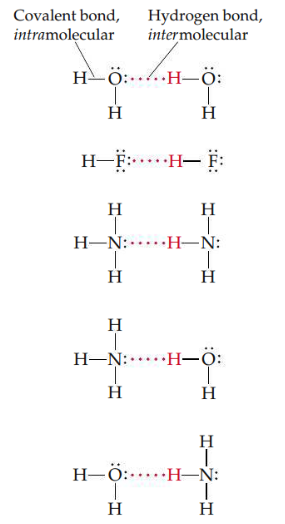
19
New cards
Ion-Dipole force
The attraction between an ion and a polar molecule. Cations are attracted to the negative end of a dipole, and anions are attracted to the positive end. The strength of the attraction increases as either the ionic charge or the magnitude of the dipole moment increases.
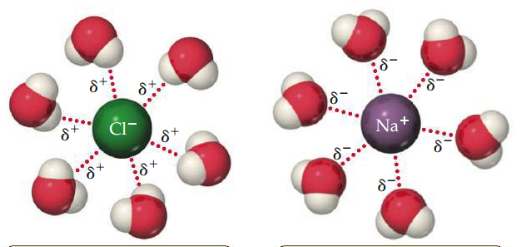
20
New cards
Comparing intermolecular forces
-When the molecules of two substances have comparable molecular weights and shapes, dispersion forces are approximately equal in the two substances. (generally use dipole-dipole forces)
-When the molecules of two substances differ widely in molecular weights, and there is no hydrogen bonding. (generally use dispersion forces)
-When the molecules of two substances differ widely in molecular weights, and there is no hydrogen bonding. (generally use dispersion forces)
21
New cards
Viscosity
A measure of the resistance of fluids to flow. The ease with which the molecules of the liquid can move relative to one another depend on the attractive forces between molecules and on whether the shapes and flexibility of the molecules are such that they tend to become entangled. (long molecules can become tangled like spaghetti)

22
New cards
Surface tension
The intermolecular, cohesive attraction that cause a liquid to minimize its surface area. The energy required to increase the surface area of a liquid by a unit amount.
23
New cards
Adhesive force
Intermolecular forces that bind a substance to a surface.
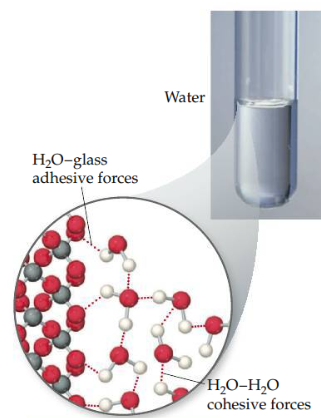
24
New cards
Cohesive force
Intermolecular forces that bind similar molecules to one another, such as the hydrogen bonding in water.
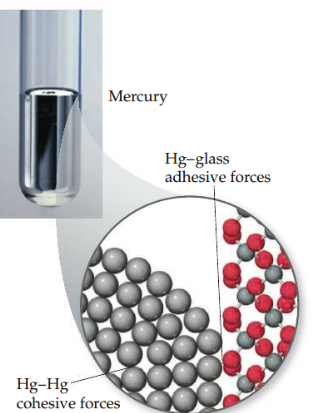
25
New cards
Capillary action
The process by which a liquid rises in a tube because of a combination of adhesion to the walls of the tube and cohesion between liquid particles.
26
New cards
Phase changes
The conversion of a substance from one state of matter to another. The phase changes we consider are melting and freezing (solid ⇌ liquid), sublimation and deposition, and vaporization and condensation (liquid ⇌ gas).
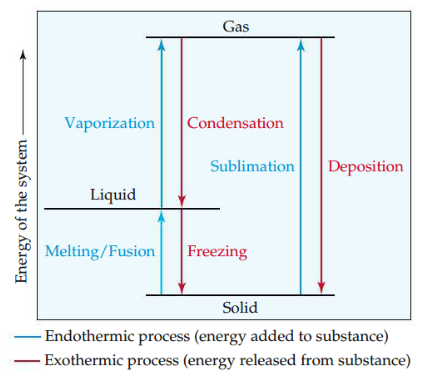
27
New cards
Dynamic equilibrium
A state of balance in which opposing processes occur at the same rate.
28
New cards
heat of fusion
The enthalpy change, ∆Hfus for melting a solid. (~6.01Kj/mol - water)
29
New cards
heat of vaporization
The enthalpy change, ∆Hvap for vaporization of a liquid. (~40.7Kj/mol - water)
30
New cards
heat of sublimation
The enthalpy change, ∆Hsub for vaporization of a solid. The sum of heat of fusion and heat of vaporization. (~47Kj/mol - water)
31
New cards
Normal melting point
The melting point at 1 atm pressure. (0 °C - water)
32
New cards
Boiling point
The temperature at which its vapor pressure equals the external pressure, acting on the liquid surface. At this temperature, the thermal energy of the molecules is great enough for the molecules in the interior of the liquid to break free from their neighbors and enter the gas phase. The boiling point increases as the external pressure increases.
33
New cards
Normal boiling point
The boiling point of a liquid at 1 atm (760 torr) pressure.
34
New cards
Intermolecular vs Heat of vap/fus
Stronger intermolecular forces means higher heat of vap/fus.
35
New cards
Supercooling
Heat is removed so rapidly that the molecules have no time to assume the ordered structure of a sold. A supercooled liquid is unstable; particles of dust entering the solution or gently stirring is often sufficient to cause the substances to solidify quickly.
36
New cards
Calculating ∆H for temperature and phase changes
Specific heat: (moles)(grams/mol)(J/g-K)(∆K) = Joules/1000 = Kilojoules
Heat of phase change: (moles)(Kj/mol) = Kilojoules
Heat of phase change: (moles)(Kj/mol) = Kilojoules
37
New cards
Critical pressure
The pressure at which a gas at its critical temperature is converted to a liquid state.
38
New cards
Critical temperature
The highest temperature at which it is possible to convert the gaseous form of a substance to a liquid. Increases with an increase in the magnitude of intermolecular forces.
39
New cards
1 atm is equal to
101,325 Pascals
760 Torr
14.6959 PSI
760 Torr
14.6959 PSI
40
New cards
Clausius-Clapeyron Equation
The natural log of the vapor pressure of a liquid is inversely proportional to its temperature.
ln P = (-∆Hvap)/(R) * 1/(T) + C
y = m x + B
P - pressure
T - temperature in kelvin
R - ideal gas constant
∆Hvap - molar enthalpy of vaporization
C - constant
ln P = (-∆Hvap)/(R) * 1/(T) + C
y = m x + B
P - pressure
T - temperature in kelvin
R - ideal gas constant
∆Hvap - molar enthalpy of vaporization
C - constant
41
New cards
Ideal gas constant
62.36L⋅mmHg/K⋅mol
0.08206L⋅atm/K⋅mol
8.314J/K⋅mol
0.08206L⋅atm/K⋅mol
8.314J/K⋅mol
42
New cards
Enthalpy of vaporization of a substance
∆Hvap = -slope * R
slope = -∆Hvap/R
slope = -∆Hvap/R
43
New cards
Vapor pressure
The pressure of air when evaporation and condensation are at equilibrium. At any temperature some liquid molecules have enough energy to escape the surface and become a gas. As temperature rises the fraction of molecules that have enough energy to break free increases.
44
New cards
Ionic solid
Held together by the mutual electrostatic attraction between cations and anions. Differences between ionic and metallic bonding make the electrical and mechanical properties of ionic solids very different from those of metals: Ionic solids do not conduct electricity well and are brittle.
- Soluble in water
- High melting point
- High lattice energy
- Soluble in water
- High melting point
- High lattice energy
45
New cards
Metallic solid
Held together by a delocalized “sea” of collectively shared valence electrons. This form of bonding allows metals to conduct electricity. It is also responsible for the fact that most metals are relatively strong without being brittle
- Malleability
- Ductility
- Malleability
- Ductility
46
New cards
Alloy
Material that contains more than one element and has the characteristic
properties of a metal.
properties of a metal.
47
New cards
Substitutional alloy
When atoms of the solute in a solid solution occupy positions normally occupied by a solvent atom. Atoms of solute around the same size of solvent atoms. Formed when the two metallic components have similar
atomic radii and chemical-bonding characteristics.
atomic radii and chemical-bonding characteristics.
48
New cards
Interstitial alloy
When the solute atoms occupy interstitial positions in the “holes” between solvent atoms. The solute atoms must have a much smaller bonding atomic radius than the solvent atoms
49
New cards
Hetergeneous alloy
Components not dispersed uniformity.
50
New cards
Molecular solid
Molecular solids are held together by the intermolecular forces: dispersion forces, dipole–dipole interactions, and hydrogen bonds. Because these forces are relatively weak, molecular solids tend to be soft and have low melting points.
51
New cards
Network covalent solid
Held together by an extended network of covalent bonds. This type of bonding can result in materials that are extremely hard, like diamond, and it is also responsible for the unique properties of semiconductors
- Tend to have higher melting and boiling points
- Tend to have higher melting and boiling points
52
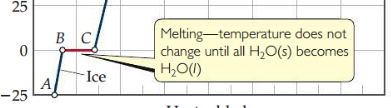
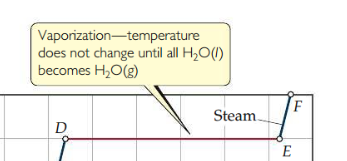
New cards
Heating curve
Plot of heat vs. temperature. Within a phase, heat is the product of specific heat, sample mass, and temp change. The temperature of a substance does not change during a phase change.