PHYS C2
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
Radiation Def
process of emitting energy as waves or particles
Types:
Ionising - capable of ionisation (removing atom’s electrons) as they pass through matter
Non-Ionising
Types Ionising Radiation
Directly Ionising - Charged particles interact directly with atomic electrons through Coulombic forces (likes repel, opposites attract) - electrons, positrons, protons
Indirectly Ionising - Electrically neutral particles or rays do not interact strongly with matter. Ionisation mainly secondary (occurs after primary ionisation event) - neutrons, gamma rays, x-rays
Photon
A quantum of electromagnetic energy
Behaves as wave and particle
Fundamental particle
No mass or electric charge
Wave-Particle Duality
fundamental particles exhibit properties of both waves (like interference) when propagating and particles (like momentum) when interacting
α-particle
Helium nucleus (2 protons, 2 neutrons, +2 charge)
Highly ionizing - serious tissue damage
Slower, less penetrating, smaller range than β-particle
β-particle
Mass and charge of electron
Different to electron as it originates from nucleus
Less ionising, faster, more penetrating, larger range than α-particle
Lose large fraction of energy in collisions
easily deflected
neutrons
give rise to protons and α-particles via spontaneous radioactive decay and other nuclear reactions (fission, fusion)
Similar mass to proton
Stopping power
gradual loss of energy of charged particle or wave as it penetrates an absorbing medium
Range of a charged particle
total distance travelled through a medium until it comes to rest
Depends on:
-Particle charge
-Particle energy
-Particle mass (lighter = further)
-Medium density
Electron Excitation Formula
Emitted photon has energy: ΔE = E2 – E1
Nuclear binding energy
energy required for a stable atom to separate it into its constituent parts
negative as the separated condition is given an arbitrary energy of 0
different for isotopes
Electronic binding energy
energy needed to remove an electron from an atom
Radioactive Decay
Transition from the quantum state of the original nuclide (parent) to a quantum state of the product nuclide (daughter)
Daughter nuclei may decay further
Radioactive decay energy
release of nuclear energy from an unstable parent nucleus mostly in the form of particles and electromagnetic radiation
causes daughter nucleus to have less energy that parent
average decay rate formula
N - number radioactive atoms
λ - decay constant (probability of decay per unit time) unit: 1/s
e.g λ = 0.05 s^-1 an average of 5% percent of the atoms decay each second
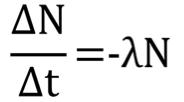
Decay Rate
Expressed as negative as the number of atoms is decreasing
Constant, characteristic value for each radionuclide
Independent of the atom’s age, physical conditions
(temp, pressure), and chemical state of atom’s
environment
Activity
number of nuclei decays per unit time, denotes how quickly a source will shed mass or energy (to reach a more stable binding energy per nucleon)
depends on how many nuclei remain
Activity Formula
Activity (A, Bq)
N(t) - number of atoms at time t
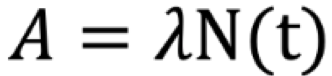
Exponential Decay
models how the number of radioactive atoms in a sample decreases over time at a rate proportional to the amount present
Initially falls steeply then rate slows due to constant half life and decay constant
Exponential Decay with Decay Constant Formula
N(t) - number atoms remaining
N(0) - number initial atoms
Can be swapped for Activity (A(t) and A(0) )
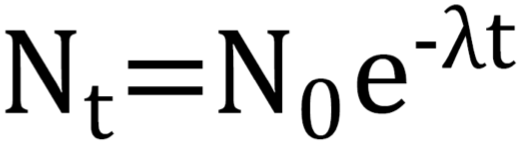
Exponential Decay with Linear Attenuation Coefficient Formula
D(x) - Dose after passing through material
D(o) - initial dose
Can be swapped for Intensity (I(x) and I(o) )

(Physical) Half-Life
time required to reduce initial activity by half (50%, 25%, 12.5%)
if t = half life, N(t) = 0.5N(0)
Physical Half-Life Formula
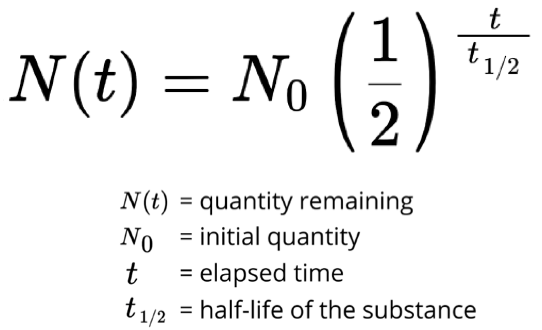
Biological half-life def + formula
λ(b): time required for half of radiopharmaceutical to be lost from biological system
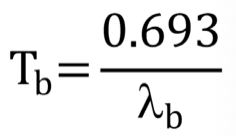
Effective rate of radioactivity loss def and formula
Radiopharmaceutical loss is due to biological half-life (via faecal and urinary excretion, perspiration) and physical decay (λ(p)) of radionuclide
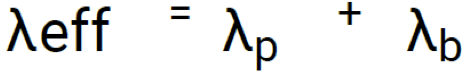
Effective half-life formula and purpose
Useful assess radiation risk and planning safe doses and timing for nuclear medicine procedures
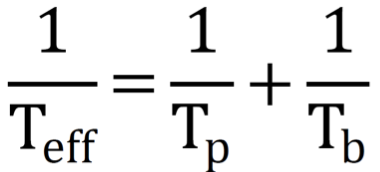
α decay
Nucleus emits α-particle and often some gamma rays
Alpha particle gets absorbed by matter quickly
E.g: radium-226 to radon-222
Electron Capture
Neutron deficient nucleus captures orbital electron which combines with a proton to form a neutron
Neutrino and x-ray emitted
Electrons from higher energy levels can drop down to the new vacancy, releasing photons in process
Daughter is often in excited state so may release gamma rays
Decay scheme is to the left since Z decreases
E.g: 125-I decay
β+ decay
Occurs in neutron-deficient radionuclide
Proton converts to neutron, positron, and neutrino
Additional gamma rays can be emitted
E.g: decay of 15-O
Neutrinos
most abundant particles that has mass (although very small) in the universe
Chargeless elementary particle
In lepton family
interacts through weak nuclear force and gravity
released in some nuclear reactions to conserve energy, momentum, and lepton number laws
Metastable State
Nucleus in a high energy state with an unusually long lifetime but will eventually decay into stabler, lower energy state
Isomeric Transition
Metastable daughter decays by emission of a γ-ray
E.g: 99m-Tc decays to 99-Mo
β – Decay
Occurs when neutron rich radionuclide converts a neutron into a proton, and emits a beta particle and an antineutrino
Decay scheme is to the right since Z increases
E.g: C-14 decays to C-12
Everyday Sources of Radiation
Cosmic rays
Radioactive substances in earth’s crust
Radioactive elements in air, food, and water
Radiation Indoors
uranium and thorium radioactive decay in bricks, tiles, concrete
Bore water and poor ventilation increases radon
High altitude = less shielding of atmosphere from cosmic rays
Artificial Radiation Sources
Nuclear medicine procedures
Industry pollution
Air travel
Australia average annual radiation dose from natural background radiation
1.5 to 2 mSv
Radiation Risk
Risk for developing cancer increases per Sv
Hereditary effects much more unlikely