Electrode Potentials and Cells
1/61
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
62 Terms
What is voltage?
The amount of energy carried by the electrons
What is current?
The flow of electrons in a circuit that carries the energy
What is potential difference?
Difference in voltage
What is electromotive force (EMF)?
The potential difference between two half cells
What is an electrode potential?
The potential difference between two half cells that make up a cell
How are half cells written in formulae?
As half equations
How are complete cells written in formulae?
As a full equation
What is the most simple half cell we can have?
A piece of metal dipped into a solution of that same metal’s ions
For example, a zinc metal strip dipped in zinc sulfate solution
Are half cells always written as reduction or oxidation?
Reduction
Therefore the electrons must always be on the left hand side
What are the standard conditions for the simplest half cell?
The solution of metal ions must be 1 moldm-3
For the simplest half cells, what is determining the voltage?
The position of equilibrium determine the voltage that can flow or potential difference between the metal strip and the solution of metal
The more negative the electrode potential of a half cell/a half cell equation is, the more likely the electrons are to be released
This means that the direction of the half cell equation goes in reverse
X+(aq) + e- ⇌ X(s) is more likely to go in reverse the lower the electrode potential is
What is an electrochemical cell?
When two half cells are joined together
Symbol for electrode potential?
Eo
What is the Daniell Cell?
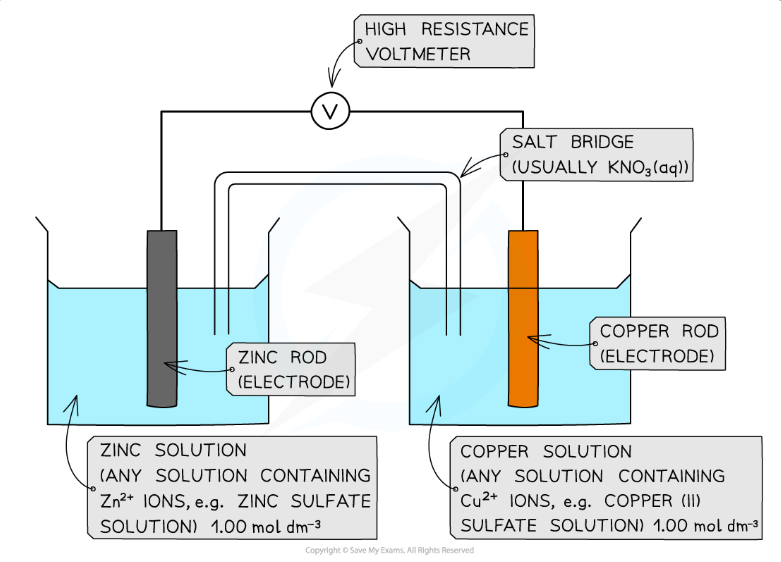
How does the Daniell Cell work?
Zinc half cell: Zn2+(aq) + 2e− → Zn(s) and E
o= -0.76Copper half cell: Cu²⁺(aq) + 2e⁻ → Cu(s) and its E
o= -+0.34The zinc half cell equation is more likely to go backwards and therefore the half cell/zinc atoms releases electrons
The copper half cell equation is more likely to go forwards as it has a higher electrode potential and therefore the half cell/copper ions will accept electrons to become copper atoms
This creates a flow of electrons around our cell- a current
What is required for electrons to flow between two half cells?
The half cells must have a potential difference
One species must release electrons, one species must accept electrons
What is the purpose of the salt bridge?
It completes the circuit by allowing the movement of ions between half cells
It neutralises charges in each half cell by allowing ions within the salt bridge to flow and neutralise any built up charges
How does a salt bridge work and why is it important for a Daniell Cell?
If the zinc keeps releasing electrons and the copper keeps accepting electrons, eventually the solutions will become positively and negatively charged as Zn2+ is continually released into it’s solution and Cu2+ is used up, only leaving negative SO42- ions
This will cause the electrons to be attracted to the zinc solution and repelled by the copper solution, preventing the flow of electrons meaning we have no current
Salt bridges normally containing KNO3 release K+ ions and NO3- ions into each solution
The K+ ions will react with the SO42- ions to neutralise the negative charge in the copper half cell
The NO3- ions will react with the Zn2+ ions to neutralise the positive charge in the zinc half cell
This means that each solution is essentially neutrally charged, maintaining the flow of electrons
Why is KNO3 used in a salt bridge?
K+ and NO3- are unreactive with the electrodes and the solutions
They will not form precipitates that would block the salt bridge- all nitrates are soluble
If you had a KCl salt bridge and a solution with Ag+ ions, the chloride ions would react with silver ions to from an insoluble white precipitate
How do we write an overall equation for an electrochemical cell (of zinc and copper for example)?
The copper half cell is more likely to go in the forward direction so its written as Cu²⁺(aq) + 2e⁻ → Cu(s)
The zinc half cell is more likely to go in the backwards direction so it is written as Zn(s) → Zn²⁺(aq) + 2e⁻
Cancel out the electrons and any other species like you would for a redox equation
Overall cell: Zn(s) + Cu²⁺(aq) → Zn²⁺(aq) + Cu(s)
How do we calculate the electrode potential of a cell?
Eo cell of EMF of the cell = Eo pos - Eo neg
Keep in mind that both cells may have positive electrode potentials or they may both be negative
In that case you take away the more negative from the less negative/less positive from more positive
Remember if you mine a negative you’re adding the number
What way way does equilibrium shift for half cells with increasingly negative electrode potentials and what is their reducing ability?
These half cell equations are more likely to go backward
Reducing agents are always on the right hand side of the equations
Since the electrons are on the left hand side of the equations, the species releases electrons when moving in the backward direction, meaning it has been oxidised, causing another species to become reduced
The half cell with the lowest electrode potential is the best reducing agent and the worst oxidising agent
What way way does equilibrium shift for half cells with increasingly positive electrode potentials and what is their oxidising ability?
These half cell equations are more likely to go forward
Oxidising agents are always on the left hand side of the equations
Since the electrons are on the left hand side of the equations, the species accepts electrons when moving in the forward direction, meaning it has been reduced, causing another species to become oxidised
The half cell with the lowest electrode potential is the best reducing agent and the worst oxidising agent
Why does a Daniell cell contain a high-resistance voltmeter?
Prevents electrons from travelling continuously from one half cell to another, therefore keeping the current low which is safer
The voltmeter is also there to have a steady flow of electrons so the cell won’t run out of power quickly
How would we power an appliance with a Daniell cell?
We’d just place the appliance in the place we’d put the voltmeter
How do we draw out cell diagrams for a Daniell cell?
Zn(s) | Zn²⁺(aq) || Cu²⁺(aq) | Cu(s)
A line (|) is used to show state changes
Two lines (||) is used to show the salt bridge
The most reduced species should be on the outside, the most oxidised should be on the inside/closer to the salt bridge
The species with the most negative electrode potential goes on the left
If a half cell does not include a state change we use a comma instead of the line
If a half cell does not include a state change or the species is not capable of conducting electricity we also need a platinum electrode so we write Pt(s) on the outside
For example: Pt(s) | Fe²⁺(aq), Fe³⁺(aq) || Cu²⁺(aq) | Cu(s) - The iron ions are aqueous and do not conduct electricity by themselves
or Pt(s) | H₂(g) | H⁺(aq) || Cu²⁺(aq) | Cu(s) - hydrogen and hydrogen ions cannot conduct electricity but there is still a state change so we include the line instead of a comma
What do we use as a reference to measure the electrode potential of all half cells?
Standard hydrogen electrode (SHE)
Used as a reference for the electrode potential of all electrodes
The electrode potential o the SHE is 0.00 V
How is the electrode potential of a half cell measured?
By connecting them to a standard hydrogen electrode
What is the standard hydrogen electrode?
The H+ ions can be provided by an acid like hydrochloric acid
If you were using sulfuric acid you would you a 0.5 moldm-3 solution as it is a diprotic acid
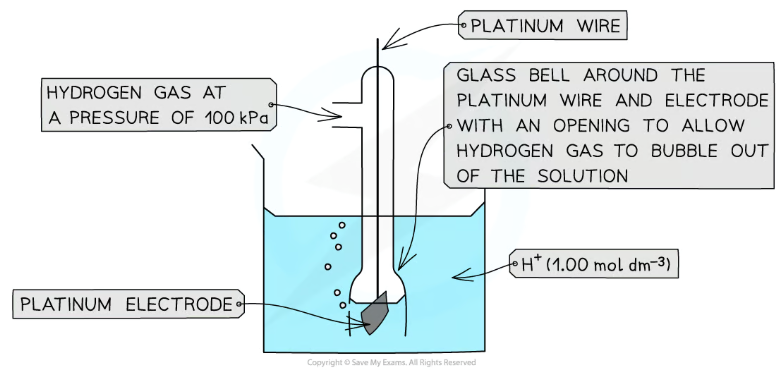
What is the Half equation for the standard hydrogen electrode?
2H⁺(aq) + 2e⁻ ⇌ H₂(g)
This is (probably) in your electrochemical series in your booklet
What does a cell diagram look like for the standard hydrogen electrode?
Pt(s) | H₂(g) | H⁺(aq) || _ | _
The platinum is needed as hydrogen or hydrogen ions cannot conduct electricity by themselves
The empty spaces with _ is where another half cell could be placed
What conditions are needed to measure standard electrode potentials using a SHE?
Concentration of 1 moldm-3
Pressure of 100 KPa
Temperature of 298K
Why is platinum suitable to be used as an electrode in the SHE or in half cells where the species are in the same state/are incapable of conducting electricity?
Good electrical conductor
Inert/doesn’t react with the electrolyte (ions in solution)
How can we work out the electrode potential of a half cell with only the electrode potential of another half cell, the EMF of the whole cell and the cell diagram? Use silver and and tin half cells for this
The electrode potential of the tin half cell is -0.14
The cell diagram is Sn(s) | Sn²⁺(aq) || Ag⁺(aq) | Ag(s)
The EMF of the cell is +0.94
Since the half cell with the most negative electrode potential is always on the left of the salt bridge, we know the negative electrode potential so:
E
ocell = Eopos - Eoneg0.94V = E
opos - (-0.14V)E
opos = +0.80 V = Electrode potential of the silver half cell
What does a reaction being spontaneous or feasible mean?
A reaction that can occur in standard conditions without a continuous external energy input
How an we deduce if a reaction is feasible?
Lets take silver reducing zinc (II) ions
To reduce zinc, silver needs a more negative electrode potential that zinc as electrodes with lower electrode potentials are better reducing agents as they are more likely to have their equilibrium move backwards and release electrons for the other species to accept
However, the electrode potential of a silver half cell is less negative than a zinc half cell (+0.80V versus -0.76V)
This means that the reaction is not feasible
How can concentration effect electrode potential values for half cells?
Take this half cell for example: Zn²⁺(aq) + 2e⁻ ⇌ Zn(s)
Increasing the concentration of zinc ions would shift equilibrium right
This would also cause electrons to be used up so the electrode potential becomes more positive
Increasing the concentration of zinc atoms would shift equilibrium left, causing electrons to be release and therefore the electrode potential value decreases
How can concentration affect the electrode potential of a full cell?
Take this cell for example: Zn(s) + 2Ag⁺(aq) → Zn²⁺(aq) + 2Ag(s)
Zinc half cell: Zn²⁺(aq) + 2e⁻ ⇌ Zn(s)
Silver half cell: 2Ag⁺(aq) + 2e⁻ ⇌ 2Ag(s)
Decreasing the concentration of zinc ions causes equilibrium to shift left so electrons are released
The electrode potential of the half cell becomes more negative
When calculating E
oCell, taking a more negative number away from a positive will give us a greater electrode potential value for the cell
What is a battery and how do they work?
Several cells joined together
Redox reactions can be used to generate electrical current
This is the basis of batteries and fuel cells
What are the two types of battery?
Non-rechargeable batteries (sometimes called primary cells)
Rechargeable batteries (sometimes called secondary cells)
What is the Lithium-Ion cell used for?
Used in mobile phones and laptops
What are the positive and negative electrodes in lithium-ion cell?
Positive: lithium cobalt oxide - Li+[CoO2]-
Negative: Carbon (graphite)
What are the equations occurring at the positive and negative electrodes of a lithium-ion cell?
Positive: Li+(aq) + CoO2(aq) + e- ⇌ Li+[CoO2]- (s)
Negative: Li+(aq) + e- ⇌ Li(s)
What is the overall equation for a lithium-ion cell? How does this show how rechargeable batteries work? (The electrode potential of the lithium half cell is more negative than the lithium cobalt oxide)
Li(s) + CoO2(s) ⇌ Li+[CoO2]-(s)
This shows the discharge equation which provides our device with energy
External voltage (using a charger) reverses the reaction- this is called the charging equation
What is the reducing agent in the Lithium-ion cell and how can we see this?
Lithium
Cobalt is reduced from +4 ion CoO2 to +3 ion Li+[CoO2]-
Aside from the lithium ion cell, what are some other examples of rechargeable batteries?
Zinc-carbon dry cell
Nickel-Cadmium cell
How do fuel cells work?
A fuel cell converts chemical energy into electrical energy in a similar fashion to a battery
Fuel cells are open systems- reactants flow in and products flow out rather than being stored in the cell
Where are fuel cells used?
In spacecraft and hydrogen powered cars
What are hydrogen and hydrogen-oxygen fuel cells?
A fuel cell that pumps hydrogen and oxygen in and pumps water out
The hydrogen fuel cell operates in acidic conditions while the hydrogen-oxygen fuel cells operates in alkaline conditions
Commercial cells use porous platinum electrodes in contact with concentrated aqueous potassium hydroxide
The pores increase the surface area
How does a hydrogen-oxygen fuel cell work?
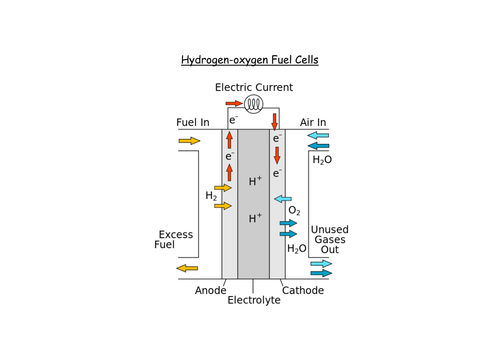
Give the equations for what occurs in a hydrogen-oxygen fuel cell and the overall equation?
Positive: O2(g) + 2H2O(l) + 4e- ⇌ 4OH-(aq)
Negative: 2H2O(l) + 2e- ⇌ 2OH-(aq) + H2(g)
Overall: O2 (g) + 2H2(g) → 2H2O(l)
What is the conventional cell diagram for a hydrogen-oxygen fuel cell?
Pt(S) | H2(g) | H2O(l) || O2(g) | H2O(l) | Pt(s)
How are hydrogen and hydrogen-oxygen fuel cells similar?
They both have the same overall equation: O2 (g) + 2H2(g) → 2H2O(l)
They both have the same cell electrode potential
They both have the same conventional cell diagram
Why may hydrogen fuel cells cause and increase is CO2 emissions even though they only release water?
Hydrogen for the fuel cell is provided in two ways:
Electrolysis of water splits water into H+ and OH- ions but the electricity needed is produced by burning fossil fuels at power stations
The reaction of hydrocarbons with steam produces hydrogen but it also produces carbon dioxide
CH4(g) + 2H2O(l) → CO2 + 4H2(g)
What are some limitations of hydrogen fuel cells?
Production of hydrogen relies on either the burning of hydrogen to produce electricity for electrolysis of water or reacting hydrocarbons with steam, both of which release carbon dioxide
Hydrogen is flammable
Storage of fuel- the infrastructure is not available for things like hydrogen gas pumps at power stations
Platinum mining is expensive and also destroys habitats
Fossil fuels also needed to produce energy for mining
Expensive as they contain platinum
What are organic fuel cells?
Other fuels could be used like glucose, methanol, ethanol etc. to produce electricity
What is the overall equation for an organic fuel cell always the same as?
The combustion equation of the organic compound
In organic fuel cells, how can we find the equation at one half cell when only given the equation for the other half cell? Use the reaction of methanol reacting with water producing CO2 and H+ at the negative electrode.
At the negative electrode: CH₃OH + H₂O → CO₂ + 6H⁺ + 6e⁻
The overall equation is: CH₃OH + 1.5 O₂ → CO₂ + 2H₂O - we know this because the overall equation is the same as the combustion equation
At the positive electrode we need to find what is missing from negative electrode equation to give us the overall
On the left hand side of the positive electrode equation we have to have 6H+ and 6e- because they do not appear in the overall equation and well as 1.5 moles of oxygen as this appears in the overall equation but not the negative
On the right hand side we have to have 3 moles of H2O because it would cancel out with the 1 mole in the negative equation to give 2H2O
So, the equation at the positive electrode is: 6e- + 6H+ + 1.5O2 → 3H2O
What is the cell diagram for this cell and why? he negative electrode is carbon and the positive is platinum
CH₃OH + 1.5 O₂ → CO₂ + 2H₂O
C(s) | CH₃OH(aq) | CO₂(g) || O₂(g) | H2O(l) | Pt(s)
The carbon is bonded to an oxygen meaning it must have been oxidised
The oxidation state of carbon in methanol is -2 and then its +4 in carbon dioxide so it has been oxidised
The oxidation state of oxygen goes from 0 to -2 in both carbon dioxide and water meaning it must have been reduced
What is a problem with organic fuel cells?
They all produce carbon dioxide
What are the purposes of a platinum electrode?
Provides a reaction surface
Allows the transfer of electrons
Economic and environmental disadvantage of producing hydrogen with the hydration of hydrocarbons with steam?
Economic
Cost of production
High temperature
Use of methane/hydrocarbons