Topic 2a: bonding
1/43
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
44 Terms
ionic bond definition
electrostatic attraction between two oppositely charged ions which form when electron transfer from the outershell of one atom to another
cations and anions
cations: positive ions
anions: negative ions
ionic bond explaination (charge)
the larger the charge → stronger attraction
ionic bond explaination (ionic radius)
ionic radius depends on the number of shells and the number of protons in the nucleus
more shells → bigger ions
more protons → force of attraction on the electrons is stronger → smaller ions
isoelectronic ions
same electron configuration but different number of protons
properties of ionic compounds
because they contain giant lattices f ions strongly bonded together by electrostatic attraction
hard, brittle
high melting and boiling point
soluble in water
do not conduct electricity in solid but conduct in liquid (ions are free to move)
migration of ions experiment
using electrolysis and coloured ions (CuCr2O7) → orange negative ions move towards the positive electrode (Cr2O7 2-) and blue positive ions (Cu 2+) move towards the negative electrode
covalent bonding
strong electrostatic atraction between two nucleui and the shared pair of electrons
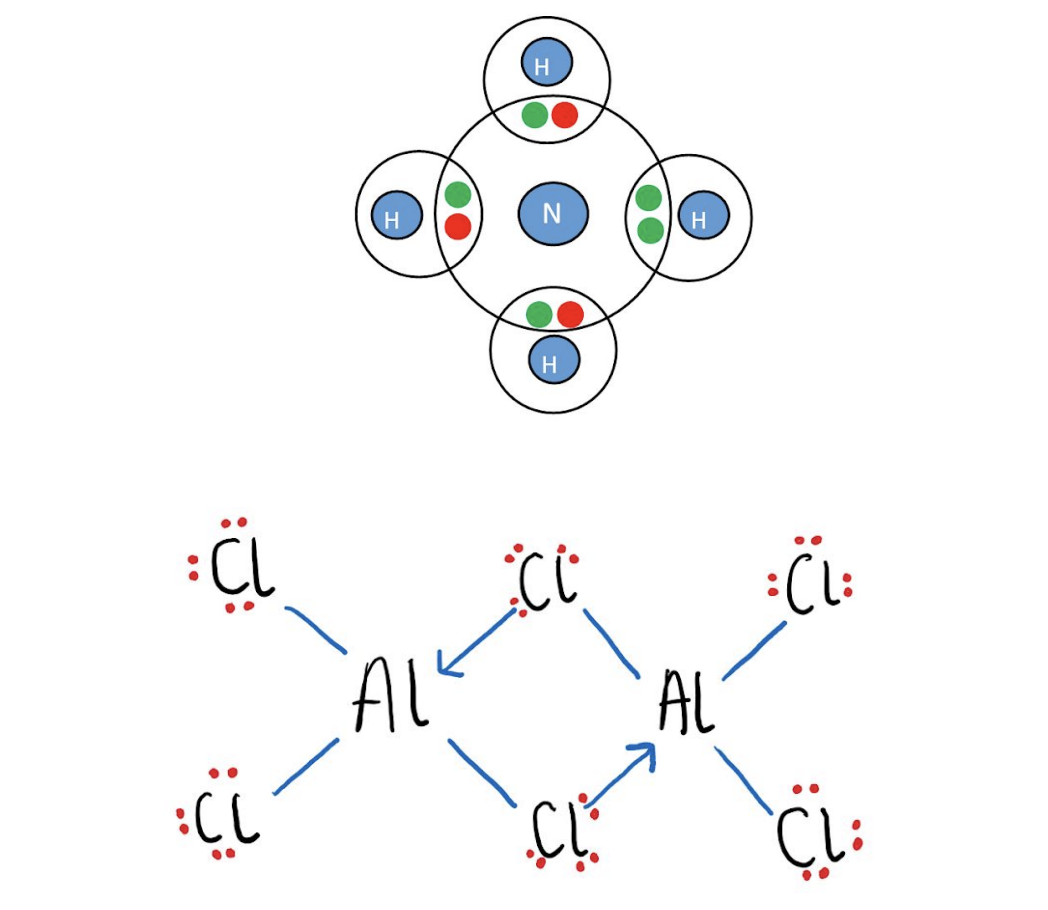
dative covalent bond
sharing of electrons is uneven → both shared electrons come from the same atom
example of dative covalent bond (Al2Cl6)
bond lengths and bond size of covalent bonds
bond length: shorter bonds are stronger → shared pair of electrons are closer to the oppositely charged nucleui → force of attraction is stronger
bond size: the smaller the atoms → stronger the bond → less shileding electrons between the nucleus and the outershell
metallic bonding
the strong electrostatic attraction between a lattice of positive metal ions and the sea of delocalised electrons
malleability and ductility of metals
malleable → can be hammered into shape
ductile → can be drawn out into a wire
conductivity of metals
can conduct heat in solid and liquid → because they have delocalised electrons that are free to move throughout the lattice
predicting the shapes of molecules
counting the pairs of electrons around the central atom
(pairs of electron repel each other so they stay as far as way as possible to minimise electron repulsion)
2 electron pairs
linear, 180° bond angle,
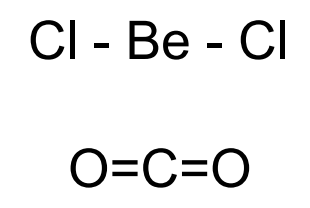
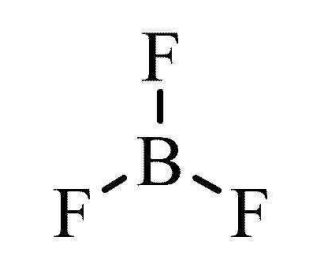
3 electron pairs
trigonal planar
bent/v shaped
trigonal planar
120° bond angle
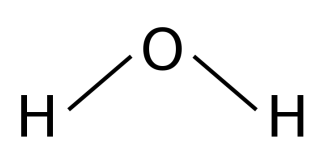
bent/ v shaped
2 bonding pair, 2 lone pair, 104.5° bond angle
4 electron pairs
tetrahedral
trigonal pyramidal
bent
tetrahedral
4 bonding pair, 0 lone pair, 109.5° bond angle
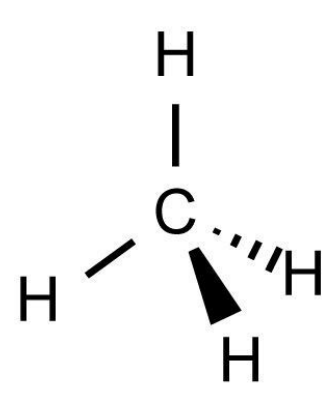
trigonal pyramidal
3 bonding pair, 1 lone pair, 107° bond angle
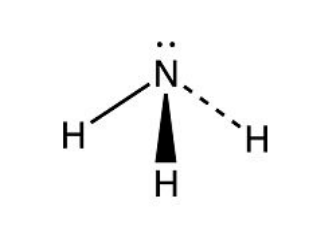
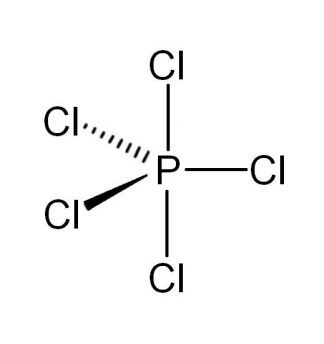
5 electron pair
trigonal bipyramidal
5 bonding pair, 0 lone pair, 120° and 90° bond angle
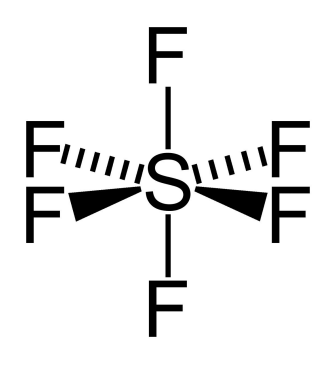
6 electron pairs
octahedral
6 bonding pair, 0 lone pair, 90° bond angle
non bonding pair space taken up…
space taken up by non bonding pair (lone pair) is greater than space taken up by bonding pair, this is because lone pairs repel lone pairs more than lone pairs repel bonding pairs
(general rule: subtract 2.5 in bond angle for each lone pair present)
answer strucutre for if no lone pair or have lone pair present
no lone pair: the shape is ___ because there are __ bonding pairs and no lone pairs around the central atom which arrange themselves to a position of minimul repulsion
electronegativity
ability of an atom to attract the bonding electrons in a covalent bond
highest electronegativity
F has the highest electronegativity in the periodic table so it has the strongest attraction for those bonding electrons
electronegativity to tell types of bonding
ionic: large difference in electronegativity (>1.7)
ionic with covalent character: large electronegativity difference (>1)
polar covalent: different electronegativities
covalent: identical or very similar electronegatrivities
polar covalent bonds
usually bonded with highly electroegative atoms like F,O,N,Cl,S,P
polar molecules
uneven distribution of electrons caused by the difference in electronegativity within the bonded atoms
eg. if the molecule is symmetrical and polar → the bond shape and the charge usually cancels out which makes it a non polar molecule
importance of intermolecular forces
melting temperature
boiling temperature
density
solubility
intermolecular forces explaination (strength)
the stronger the intermolecular forces → the more energy it requries to seperate molecules
(intermolecular forces are never strong tho)
3 types of intermolecular forces and strong or weak
london forces (weakest) → between all molecules
permanent dipole forces → only between polar molecules
hydrogen bonds (strongest) → only between molecules containing hydrogen bonded to NOF
permanant dipole - permanant dipole attraction
the slightly positive end on one molecule attracts the slightly negative end of another
instataneous dipole - instantaneous dipole attraction (london forces)
(neither contains a permanent dipole)
at any moment, the distribution of electrons in a molecule could be momentarily uneven with greater electron density in one area → this produces instantaneous dipole
the instantaneous dipole has the effect of inducing a second dipole on a neighboring molecule resulting in attraction between the molecules
(the more electrons in a molecule → more likely to have momentarily distributed unevenly electrons → stronger the london forces)
strength of london force…
depends on the number of electrons in a molecule
hydrogen bonding explaination
because when hydrogen is bonded, it only has one shell so it results in a poorly shielded proton → this means when its attracted to the lone pair of electrons on a electronegative atom of another molecule → produces strong [permanent dipole permanent dipole]
hydrogen bonding definition
electrostatic force of attraction between the poorly shielded proton of a hydrogen atom bonded to a small highly electronegative atoms such as NOF and a lone pair of electrons on a NOF atom of a neighbouring molecule
hydrogen bond bond angle
ALWAYS 180!!!!
water’s density is not 0…?
water is actually less dense below 4°C because of the open latticestructure of the ice
water’s boiling point is higher despite having less electrons??
this is because water forms hydrogen bonds and this makes its BP higher than others as it takes more energy to pull the water molecules away from their neighbours