Intermolecular Interaction between Chemical Species
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
13 Terms
Intermolecular interaction between chemical species
Between WHAT
Due to interactions between WHAT regions of different WHAT
Intermolecular interaction between chemical species
Between MOLECULES
Due to interactions between OPPOSING regions of different POLARITY (𝛿+ , 𝛿- , 𝑏𝑜𝑛𝑑 𝑑𝑖𝑝𝑜𝑙𝑒, 𝑚𝑜𝑙𝑒𝑐𝑢𝑙𝑎𝑟 𝑑𝑖𝑝𝑜𝑙𝑒)
INTRAmolecular interactions in chemical bonds WHAT one molecules WHAT interaction
INTRAmolecular interactions in chemical bonds WITHIN one molecules STRONG interaction
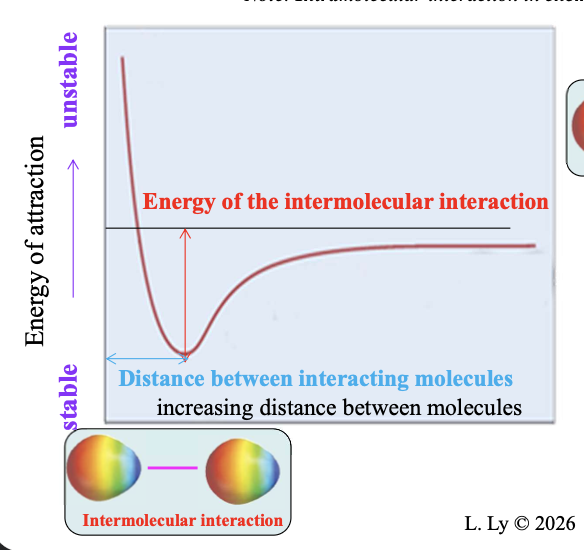
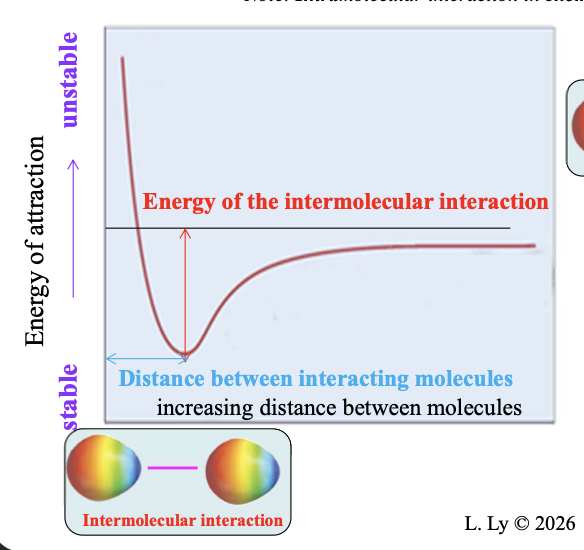
Two polar species at an infinite distance from one another have WHAT of attraction
Two polar species at an infinite distance from one another have ZERO ENERGY of attraction
TRUE Non-polar species:
Although true non-polar species (e.g., hexane, H2) with uniform electron density where 𝛿- , 𝛿+ not assigned, due to inherent movement of electron , WHAT regions of polarity exist
TRUE Non-polar species:
Although true non-polar species (e.g., hexane, H2) with uniform electron density where 𝛿- , 𝛿+ not assigned, due to inherent movement of electron , TEMPORARY regions of polarity exists.
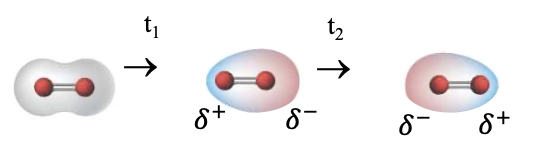
Non-polar species with significant bond dipoles/Polar species/ions:
Regions of WHAT polarity
Movement of WHAT also present
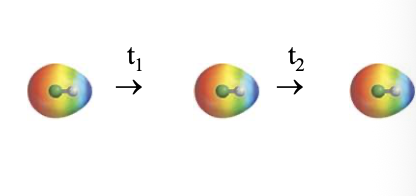
Non-polar species with significant bond dipoles/Polar species/ions:
Regions of PERMANENT polarity
Movement of ELECTRONS also present
Regions of permanent polarity are due to:
WHAT
WHAT
WHAT
Regions of permanent polarity are due to:
Differing electronegativity
Regions of larger numbers of electrons
Formal charges

Intermolecular interactions between chemical species
WHAT
WHAT
WHAT
WHAT
WHAT
Intermolecular interactions between chemical species
Ion-Dipole
Dipole-Dipole
Hydrogen bonding
Dipole-induces Dipole
Induced Dipole-Induced Dipole London Dispersion Forces (LDF)
Ion-dipole

Dipole-Dipole
Between WHAT species
Due to WHAT regions of polarity
WHAT attracted to WHAT
𝛿- (high 𝑒 density) WHAT each other
𝛿+ (low 𝑒 density) WHAT each other
Dipole-Dipole
Between POLAR species
Due to PERMANENT regions of polarity
𝛿- attracted to 𝛿+
𝛿- (high 𝑒 density) REPELLING each other
𝛿+ (low 𝑒 density) REPELLING each other
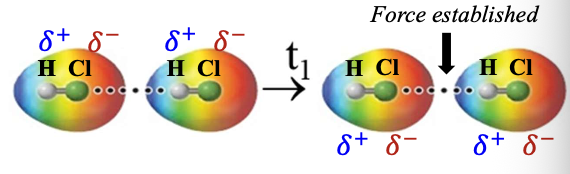
Hydrogen bonding
Extreme form of WHAT
H” bonded to a very WHAT atom
o N, O, F in NH3, H2O, HF
Interaction between “WHAT on these molecules” and “a WHAT” on another molecule
“electronegative atom – H – lone electron pair” MUST be in WHAT
Hydrogen bonding
Extreme form of DIPOLE-DIPOLE
“H” bonded to a very ELECTRONEGATIVE atom
o N, O, F in NH3, H2O, HF
Interaction between “H on these molecules” and “a lone electron pair” on another molecule
“electronegative atom – H – lone electron pair” MUST be in LINE

Dipole-induced dipole
Between WHAT and WHAT species
In mixture: as polar molecule approaching non- polar molecule, the electron cloud distorted, inducing WHAT dipole (i.e., WHAT dipole inducing WHAT dipole)
Dipole-induced dipole
Between POLAR and NON-POLAR species
In mixture: as polar molecule approaching non- polar molecule, the electron cloud distorted, inducing TEMPORARY dipole (i.e., PERMENANT dipole inducing TEMPORARY dipole)

Dipole-induced dipole
In pure species: As WHAT region approaches the WHAT region of the SAME molecule
Dipole-induced dipole
In pure species: As POLAR region approaches the NON-POLAR region of the SAME molecule
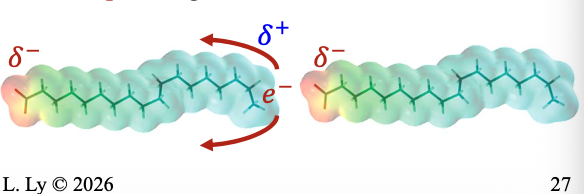
Induced Dipole-Induced Dipole London Dispersion Force (LDF)
Between WHAT species
Due to WHAT regions of polarity
Two non-polar molecules approaching each other can WHAT and have their electron cloud distorted to give WHAT dipoles
Induced Dipole-Induced Dipole London Dispersion Force (LDF)
Between NON-POLAR species
Due to TEMPORARY regions of polarity
Two non-polar molecules approaching each other can POLARIZE and have their electron cloud distorted to give TEMPORARY dipoles
